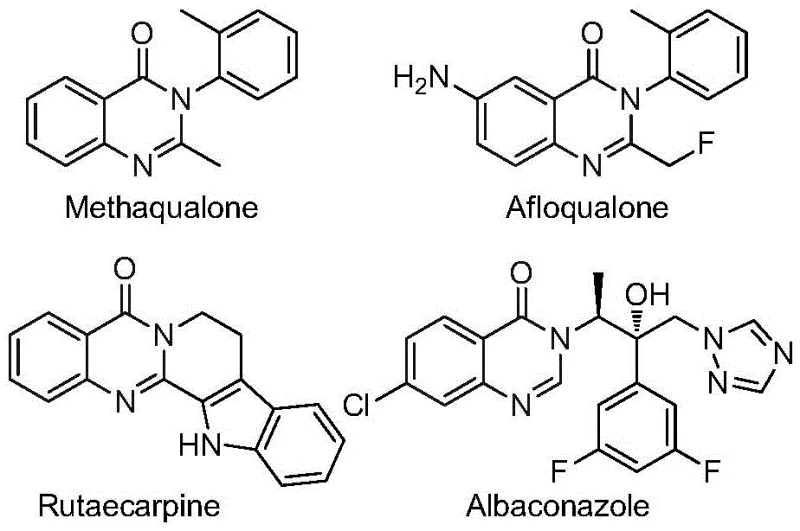
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This class of compounds is renowned for its broad spectrum of biological activities, ranging from antifungal and antiviral properties to potent anticancer effects. The introduction of the trifluoromethyl group is particularly strategic, as it enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these derivatives highly desirable candidates for modern drug development pipelines.
The significance of this technology extends beyond mere academic interest; it addresses critical bottlenecks in the manufacturing of complex nitrogen-containing heterocycles. Traditional methods often suffer from harsh conditions or limited substrate scope, hindering their adoption in large-scale commercial settings. By leveraging a palladium-catalyzed carbonylation cascade, this novel approach offers a streamlined pathway that aligns perfectly with the needs of a reliable pharmaceutical intermediate supplier aiming to deliver high-quality materials with consistent purity profiles. The ability to construct these privileged scaffolds from inexpensive starting materials marks a pivotal shift towards more sustainable and economically viable production strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose significant logistical and economic burdens on manufacturers. Conventional synthetic routes frequently necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols to mitigate the risks associated with handling toxic gases. Furthermore, many established protocols depend on precious metal catalysts such as ruthenium or platinum, which not only drive up the raw material costs but also introduce challenges in removing trace metal impurities to meet stringent regulatory standards for pharmaceutical products. Additionally, traditional approaches often require pre-activated substrates, such as benzamides or acid anhydrides, which involve additional synthetic steps, thereby increasing waste generation and reducing the overall atom economy of the process.
Another critical drawback of legacy methods is their narrow substrate tolerance. Many existing protocols fail to accommodate diverse functional groups, limiting the chemical space accessible to medicinal chemists during lead optimization. Low yields and the need for harsh reaction conditions further exacerbate these issues, making scale-up problematic. For procurement teams, these inefficiencies translate into higher costs of goods sold (COGS) and potential supply chain vulnerabilities due to the reliance on specialized reagents and equipment. Consequently, there is a pressing demand for a methodology that can overcome these limitations while maintaining high reaction efficiency and operational simplicity.
The Novel Approach
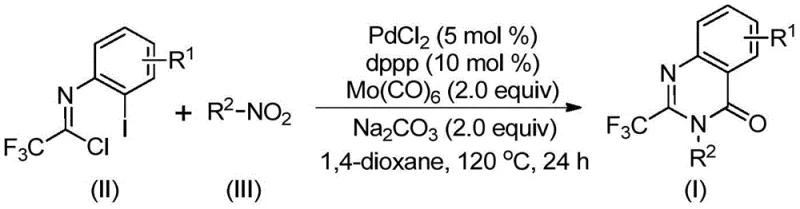
The method described in patent CN112480015B represents a paradigm shift by utilizing a palladium-catalyzed multi-component coupling reaction that operates under relatively mild conditions. Instead of hazardous CO gas, the process employs molybdenum hexacarbonyl [Mo(CO)6] as a safe and convenient solid carbon monoxide surrogate. This substitution drastically simplifies the reactor setup, allowing the reaction to proceed in standard glassware or stainless steel reactors without the need for high-pressure infrastructure. The use of cheap and readily available nitro compounds as starting materials is another game-changer, as it bypasses the need for pre-reduction to amines or the use of expensive acylating agents. This direct utilization of nitro precursors significantly lowers the entry barrier for synthesis and reduces the overall step count.
Moreover, this novel approach exhibits exceptional functional group compatibility, enabling the synthesis of a wide array of 2-trifluoromethyl quinazolinone derivatives with diverse substitution patterns. The reaction system tolerates halogens, alkyl groups, and electron-withdrawing groups, providing medicinal chemists with the flexibility to explore structure-activity relationships (SAR) extensively. From a commercial perspective, the high yields reported (often exceeding 90% for optimized substrates) and the straightforward one-pot operation translate into substantial cost reduction in API manufacturing. The elimination of intermediate isolation steps and the use of common organic solvents like 1,4-dioxane further enhance the process's attractiveness for industrial scale-up, ensuring a stable and continuous supply of high-value intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific targets. The reaction initiates with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which serves a dual role as both a CO source and a reducing agent. This in situ generated amine then undergoes a base-promoted intermolecular coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, typically PdCl2 coordinated with a bidentate phosphine ligand like dppp, inserts into the carbon-iodine bond of the imidoyl chloride moiety. This oxidative addition step generates a reactive organopalladium species that is poised for carbonyl insertion.
As the temperature rises to 120°C, Mo(CO)6 decomposes to release carbon monoxide, which inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This step is critical for introducing the carbonyl functionality required for ring closure. Under the influence of the base, an intramolecular nucleophilic attack occurs, forming a seven-membered palladacycle intermediate. Finally, reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This elegant cascade ensures high atom efficiency and minimizes the formation of side products, resulting in a clean impurity profile that simplifies downstream purification efforts.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol is designed to be robust, accommodating slight variations in conditions while maintaining high performance. The use of a polar aprotic solvent such as 1,4-dioxane is preferred to ensure the solubility of all components and facilitate the catalytic cycle. The following guide outlines the standardized procedure derived from the patent data, serving as a foundational reference for process chemists looking to replicate or adapt this chemistry for their specific needs.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere to facilitate the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing nitro compounds and trifluoroethylimidoyl chlorides, which are commodity chemicals available from multiple global vendors, manufacturers can mitigate the risk of single-source dependency. This diversification of the supplier base enhances supply chain resilience, ensuring that production schedules are not disrupted by shortages of exotic or proprietary reagents. Furthermore, the elimination of high-pressure gas requirements reduces capital expenditure on specialized equipment, allowing for faster deployment of production capacity.
- Cost Reduction in Manufacturing: The economic implications of this method are profound. By replacing expensive noble metal catalysts like ruthenium with more abundant palladium systems and utilizing Mo(CO)6 as a safe CO source, the direct material costs are significantly lowered. The one-pot nature of the reaction eliminates the need for isolating unstable intermediates, which reduces solvent consumption, labor hours, and waste disposal costs. Although specific percentage savings depend on the scale, the qualitative reduction in unit operations translates to a leaner manufacturing process with a lower overall cost of goods, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents rather than gaseous CO or sensitive organometallics improves the logistics of raw material storage and transport. Nitro compounds and the requisite imidoyl chlorides are widely produced and have established supply chains, reducing lead times for procurement. This stability allows for better inventory management and forecasting, ensuring that production lines can run continuously without interruption. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in raw material quality, further securing the continuity of supply.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonnage levels is facilitated by the absence of high-pressure hazards. The use of standard heating and stirring equipment makes technology transfer to contract manufacturing organizations (CMOs) straightforward. Additionally, the high atom economy and reduced solvent usage align with green chemistry principles, helping companies meet increasingly strict environmental regulations. The simplified workup procedure, involving filtration and chromatography, minimizes the generation of hazardous waste streams, reducing the environmental footprint and associated compliance costs of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this route for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of using nitro compounds in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than their amine counterparts. This method utilizes them directly, avoiding pre-activation steps and reducing overall raw material costs while maintaining high reaction efficiency.
Q: Does this process require high-pressure carbon monoxide equipment?
A: No, the process utilizes Mo(CO)6 as a solid carbon monoxide substitute. This eliminates the need for dangerous high-pressure CO gas cylinders and specialized autoclaves, greatly simplifying safety protocols and equipment requirements.
Q: What is the substrate scope for this quinazolinone synthesis?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on the aromatic ring including halogens, alkyl groups, and trifluoromethyl groups, allowing for the synthesis of diverse libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable commercial supplies. We are committed to delivering high-purity pharmaceutical intermediates that meet the rigorous quality standards required by global regulatory agencies. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling and structural confirmation, guaranteeing the consistency and reliability of every batch we produce.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Whether you require custom synthesis of novel quinazolinone derivatives or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and discuss route feasibility assessments that can optimize your supply chain and reduce your time to market. Let us be your partner in turning complex chemical challenges into commercial successes.
