Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as core structures for a wide array of bioactive molecules exhibiting anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial activities. The strategic introduction of a trifluoromethyl group into these frameworks is particularly valuable, as it significantly enhances the electronegativity, bioavailability, metabolic stability, and lipophilicity of the target drug candidates. This patent presents a robust methodology that addresses long-standing challenges in synthesizing these critical building blocks, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
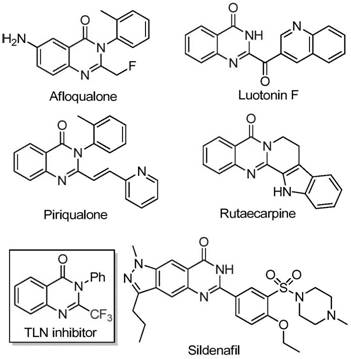
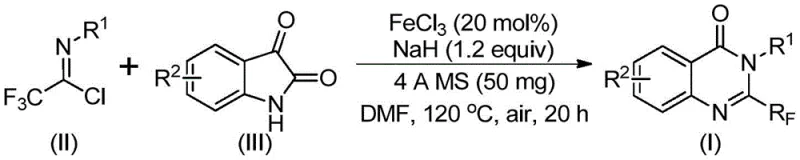
Quinazolinone derivatives represent a privileged class of nitrogen-containing fused ring six-membered heterocycles found in numerous natural products and approved drugs. The structural diversity and biological potency of these compounds make them a focal point for R&D teams globally. However, the traditional synthetic approaches to installing the trifluoromethyl motif often involve complex multi-step sequences or harsh reaction conditions that limit their practical utility. The innovation described in CN111675662B leverages a direct cyclization strategy using readily available starting materials, specifically trifluoroethylimidoyl chloride and isatin derivatives. By utilizing a cheap iron catalyst system, this method circumvents the need for expensive transition metals and severe conditions, thereby aligning perfectly with the modern principles of green chemistry and process intensification required by today's supply chain leaders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing a trifluoromethyl functional group has relied heavily on the cyclization of specific synthons with substrates such as anthranilamide, anthranilic acid, or isatoic anhydride. While these methods are chemically feasible, they are fraught with significant operational and economic disadvantages that hinder their adoption in commercial manufacturing. Common trifluoromethylating agents like trifluoroacetic anhydride or ethyl trifluoroacetate often require stringent reaction conditions, including extreme temperatures or pressures, which pose safety risks and increase energy consumption. Furthermore, these conventional routes frequently suffer from narrow substrate scopes, meaning they fail to accommodate diverse functional groups without extensive protection-deprotection strategies. The resulting low yields and poor atom economy translate directly into higher production costs and increased waste generation, creating bottlenecks for procurement managers seeking reliable and cost-efficient sources of high-purity intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a synergistic combination of trifluoroethylimidoyl chloride and isatin as the primary building blocks. This strategy is catalyzed by ferric chloride (FeCl3), an inexpensive and earth-abundant metal salt, in the presence of sodium hydride as a base. The reaction proceeds through a streamlined sequence that tolerates a wide range of functional groups, including halogens, alkyls, and electron-withdrawing nitro groups, without compromising yield or purity. The operational simplicity is remarkable; the reaction can be conducted in common organic solvents like DMF under air atmosphere, eliminating the need for inert gas protection which simplifies reactor setup and reduces operational complexity. This shift from precious metal catalysis to base-metal catalysis represents a substantial leap forward in process economics, offering a scalable solution that meets the rigorous demands of modern API manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of iron catalysis in organic synthesis. The reaction initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This initial coupling generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent cyclization event. The presence of the iron catalyst is crucial here, facilitating a decarbonylation process that drives the thermodynamic equilibrium towards the formation of the stable quinazolinone ring system. This decarbonylation step is particularly efficient under the specified conditions (120 °C), ensuring high conversion rates even for sterically hindered substrates. The use of 4A molecular sieves in the reaction mixture plays a pivotal role in scavenging moisture, which is essential for maintaining the activity of the sodium hydride base and preventing hydrolysis of the sensitive imidoyl chloride starting material.
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity of the iron-catalyzed cycle minimizes the formation of side products that are commonly associated with radical pathways or non-selective nucleophilic attacks. The tolerance for various substituents on the aryl rings (R1 and R2) indicates that the electronic properties of the substrate do not drastically alter the reaction trajectory, allowing for a predictable impurity profile across different analogues. For R&D directors, this predictability is invaluable, as it simplifies the purification process and ensures consistent quality of the final product. The ability to synthesize diverse derivatives, such as those with methyl, fluoro, bromo, or chloro substitutions, using the same core protocol demonstrates the robustness of the catalytic system and its suitability for generating libraries of compounds for structure-activity relationship (SAR) studies.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis of these valuable intermediates is designed to be straightforward and adaptable to various scales of production. The protocol involves mixing the key reagents—isatin, trifluoroethylimidoyl chloride, ferric chloride, and sodium hydride—in an appropriate solvent system. The reaction profile typically involves an initial heating phase at 40 °C followed by a higher temperature phase at 120 °C to drive the cyclization to completion. This two-stage temperature profile optimizes the reaction kinetics, ensuring that the initial coupling occurs smoothly before the energetically demanding decarbonylation step takes place. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety in your laboratory or pilot plant.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in DMF solvent.
- Stir the mixture at 40°C for 8-10 hours to initiate the reaction, then increase temperature to 120°C.
- Maintain reaction at 120°C for 18-20 hours under air, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere chemical curiosity. The primary driver for adoption is the significant reduction in manufacturing costs achieved through the substitution of expensive catalysts and reagents. By replacing precious metals with commodity-grade iron salts and utilizing widely available isatin derivatives, the overall bill of materials is drastically reduced. This cost efficiency is further amplified by the simplified downstream processing; the reaction mixture can be purified using standard silica gel column chromatography, a technique that is easily scalable and does not require specialized equipment or exotic scavengers. The elimination of complex purification steps translates directly into shorter production cycles and lower labor costs, enhancing the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of switching to this iron-catalyzed protocol is profound. Traditional methods often rely on palladium or rhodium catalysts, which are subject to volatile market prices and supply constraints. In contrast, ferric chloride is a bulk chemical with a stable and low price point, insulating the manufacturing process from raw material price fluctuations. Additionally, the high atom economy of the reaction means that less waste is generated per kilogram of product, reducing the costs associated with waste disposal and environmental compliance. The use of excess aromatic amine precursors, which are relatively inexpensive, further drives down the cost per mole of the final active pharmaceutical ingredient (API) intermediate, making this route highly attractive for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical industry, and this method strengthens it by relying on commoditized starting materials. Isatin and its derivatives are produced on a multi-ton scale globally, ensuring a steady and reliable supply that is not prone to the bottlenecks often seen with specialized fine chemicals. The robustness of the reaction conditions, which tolerate air and moisture to a reasonable extent compared to sensitive organometallic reactions, reduces the risk of batch failures due to minor operational deviations. This reliability allows for more accurate forecasting and inventory planning, enabling supply chain managers to maintain optimal stock levels without the need for excessive safety buffers. The ability to source key reagents from multiple vendors further mitigates the risk of single-source dependency.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant is often fraught with challenges, but this protocol is inherently designed for scalability. The reaction conditions are mild enough to be managed in standard stainless steel reactors, and the exothermic profile is manageable, reducing the need for complex cooling systems. From an environmental standpoint, the use of an earth-abundant metal catalyst aligns with green chemistry principles, reducing the heavy metal burden in the final product and the associated environmental footprint. The simplified workup procedure minimizes solvent usage and waste generation, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability angle is not just a regulatory requirement but also a competitive advantage when partnering with major pharmaceutical companies that prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of using FeCl3 over precious metal catalysts?
A: Ferric chloride is significantly cheaper and more abundant than precious metals like palladium or rhodium, drastically reducing raw material costs while maintaining high catalytic efficiency for this cyclization.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to simple operation and readily available starting materials.
Q: What is the substrate scope for this reaction?
A: The method exhibits excellent functional group tolerance, accommodating various substituents on the aryl ring including alkyl, halogen (F, Cl, Br, I), methoxy, and nitro groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensively evaluated the FeCl3-catalyzed synthesis route described in CN111675662B and has optimized it for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or preparing for market launch. Our state-of-the-art facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone we deliver meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule and volume needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your timeline to market with a supply chain partner that combines technical excellence with commercial reliability.
