Advanced Organocatalytic Synthesis of Chiral Indoxazinones for High-Purity Pharmaceutical Intermediates
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for High-Purity Pharmaceutical Intermediates
The landscape of modern pharmaceutical development is increasingly defined by the demand for complex chiral scaffolds that offer both structural diversity and potent biological activity. Patent CN112209947A introduces a groundbreaking methodology for the synthesis of chiral indoxazinone compounds, a class of molecules characterized by their fused indole-oxazinone ring systems. These structures are not merely academic curiosities; they represent a critical frontier in medicinal chemistry, with documented potential as S1P1 receptor antagonists, antimalarial agents, and androgen receptor modulators. The disclosed technology leverages a sophisticated dual-catalyst system involving chiral phosphoric acid and hexafluoroisopropanol to achieve exceptional stereocontrol. This innovation addresses the urgent industry need for efficient access to single-enantiomer indole-fused rings, which are often the active components in racemate mixtures but difficult to isolate through traditional resolution methods.
Furthermore, the biological validation provided within the patent underscores the commercial viability of these intermediates. The synthesized compounds exhibit measurable cytotoxic activity against PC-3 human prostate cancer cells, positioning them as valuable leads for novel anti-tumor drug research and development. For a reliable pharmaceutical intermediate supplier, mastering this synthetic route opens doors to supplying high-value building blocks for oncology pipelines. The ability to generate structural diversity through variable substituents on the indole and nitrone moieties allows for the rapid exploration of structure-activity relationships (SAR), accelerating the drug discovery timeline for downstream partners.
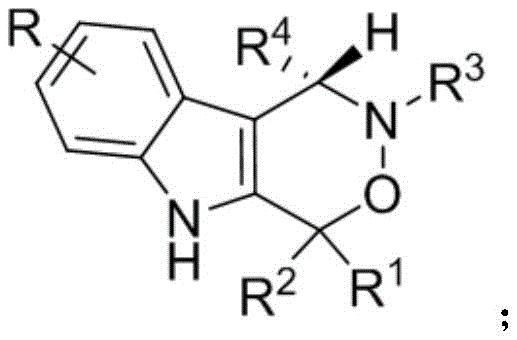
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole-fused ring systems has been plagued by significant technical hurdles that impede efficient manufacturing. Prior art methodologies often rely on harsh reaction conditions, including extreme temperatures and aggressive reagents, which not only pose safety risks in an industrial setting but also lead to the degradation of sensitive functional groups. These violent conditions frequently result in poor atom economy and the formation of complex impurity profiles that are costly and time-consuming to remove. Moreover, conventional approaches often suffer from low enantioselectivity, necessitating additional resolution steps that drastically reduce overall yield and increase the cost of goods sold (COGS). The inability to consistently produce high-purity single enantiomers limits the therapeutic utility of the final drug candidates, as the inactive or toxic enantiomer can compromise safety profiles.
The Novel Approach
In stark contrast, the methodology described in CN112209947A offers a paradigm shift towards green and efficient synthesis. The core innovation lies in the [1,3]-dipolar cycloaddition reaction between 2-indolylmethanol and nitrones, catalyzed by a chiral phosphoric acid system. This reaction proceeds under remarkably mild conditions, specifically within a temperature range of 20-30°C, which eliminates the need for energy-intensive heating or cryogenic cooling. The use of toluene as a solvent, combined with anhydrous sodium sulfate as an additive, creates an optimized reaction environment that drives the equilibrium towards the desired product while minimizing side reactions. This approach not only achieves high yields—often exceeding 90% in optimized examples—but also delivers exceptional enantiomeric excess (ee), frequently surpassing 90%. Such performance metrics are critical for cost reduction in API manufacturing, as they minimize waste and maximize the throughput of valuable chiral material.
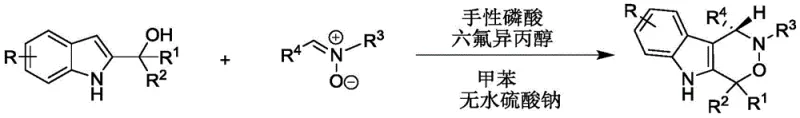
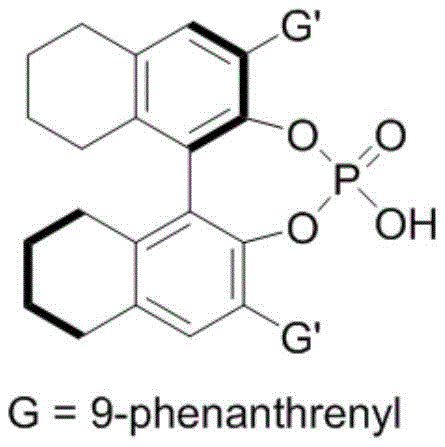
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cycloaddition
The success of this synthetic route is fundamentally rooted in the precise mechanistic interplay between the chiral phosphoric acid catalyst and the substrates. The chiral phosphoric acid, typically derived from a BINOL (1,1'-bi-2-naphthol) skeleton, acts as a bifunctional organocatalyst. It simultaneously activates the electrophilic nitrone and the nucleophilic 2-indolylmethanol through a network of hydrogen bonding interactions. The phosphoryl oxygen serves as a hydrogen bond acceptor, while the acidic proton acts as a donor, organizing the transition state within a well-defined chiral pocket. This rigid organization ensures that the cycloaddition occurs with high facial selectivity, dictating the absolute configuration of the newly formed stereocenters. The addition of hexafluoroisopropanol (HFIP) further enhances this effect by strengthening the hydrogen bonding network and stabilizing charged intermediates, thereby boosting both reaction rate and stereoselectivity.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. By avoiding transition metals such as palladium or rhodium, the process eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human use. The absence of metal residues simplifies the purification workflow, removing the need for expensive scavenging resins or complex extraction protocols. Furthermore, the mild acidic nature of the catalyst prevents the decomposition of acid-sensitive groups that might be present on the diverse array of substrates tolerated by this method. This robustness allows for the synthesis of high-purity chiral indoxazinones with complex substitution patterns, facilitating the creation of focused libraries for biological screening without the bottleneck of difficult purification.
How to Synthesize Chiral Indoxazinone Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves a straightforward one-pot procedure where the reactants are mixed in toluene with the catalytic system and a drying agent. The reaction progress is easily monitored via thin-layer chromatography (TLC), allowing for precise endpoint determination to prevent over-reaction or degradation. Upon completion, the workup involves a simple filtration to remove the solid additive, followed by concentration and standard silica gel column chromatography. This streamlined process minimizes unit operations, reducing the overall processing time and labor costs associated with manufacturing. For detailed procedural specifics, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining 2-indolylmethanol and nitrone in toluene solvent with a molar ratio between 1: 1 and 2:1.
- Add anhydrous sodium sulfate, chiral phosphoric acid catalyst, and hexafluoroisopropanol to the mixture.
- Stir the reaction at 20-30°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic technology translates into tangible strategic benefits that extend beyond mere chemical elegance. The shift from violent, metal-dependent processes to this mild organocatalytic method fundamentally alters the cost structure and risk profile of the supply chain. By utilizing readily available starting materials like substituted 2-indolylmethanols and nitrones, the dependency on exotic or supply-constrained reagents is minimized. This availability ensures a stable supply base, reducing the vulnerability of the production schedule to raw material shortages. Additionally, the simplified workup procedure—requiring only filtration and chromatography rather than complex aqueous extractions or metal scavenging—significantly reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a direct saving in raw material costs. More importantly, it removes the downstream costs associated with metal removal and validation, which can be substantial in GMP environments. The high yields and enantioselectivity achieved mean that less starting material is wasted, and the need for recrystallization or chiral separation is largely obviated. This efficiency leads to a drastic simplification of the production process, lowering the overall cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater batch-to-batch consistency. Operating at ambient temperatures (20-30°C) reduces the energy load on manufacturing facilities and minimizes the risk of thermal runaways, enhancing plant safety. The use of common solvents like toluene and standard purification techniques ensures that the process can be transferred easily between different manufacturing sites without requiring specialized equipment, thereby securing supply continuity.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its mild exothermic profile and simple phase handling. The reduced generation of hazardous waste, particularly heavy metal sludge, simplifies waste disposal and lowers environmental compliance costs. This green chemistry profile is increasingly valued by downstream pharmaceutical customers who are under pressure to reduce the carbon footprint of their supply chains, making this intermediate a more attractive option for sustainable drug development.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral indoxazinone compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing development pipelines.
Q: What are the key advantages of this synthesis method over conventional routes?
A: This method utilizes mild reaction conditions (20-30°C) and avoids violent processes common in prior art, resulting in significantly higher yields and enantioselectivity while reducing safety risks.
Q: Does the synthesized compound exhibit biological activity?
A: Yes, the chiral indoxazinone compounds demonstrate specific cytotoxic activity against PC-3 human prostate cancer cells, with certain derivatives showing potent inhibitory effects.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process uses readily available raw materials, simple post-treatment procedures like filtration and concentration, and operates under mild conditions, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112209947A for the development of next-generation anti-tumor therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the quality and stereochemical integrity of every batch we produce.
We invite you to collaborate with us to leverage this advanced chemistry for your drug discovery programs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing for these high-value chiral intermediates. Together, we can accelerate the delivery of life-saving medicines to patients worldwide.
