Advanced Metal-Free Synthesis of Fluoroalkyl-Substituted Furans for Commercial Scale-Up
Advanced Metal-Free Synthesis of Fluoroalkyl-Substituted Furans for Commercial Scale-Up
The rapid evolution of medicinal chemistry has placed a premium on fluorinated heterocycles, particularly furan derivatives, due to their unique metabolic stability and bioavailability profiles. A groundbreaking development in this sector is detailed in Chinese Patent CN110357842B, which discloses a novel, highly efficient preparation method for fluoroalkyl-substituted furan compounds. This technology represents a significant leap forward by enabling the selective cleavage of four carbon-fluorine bonds to construct complex five-membered ring systems in a single operational step. For R&D directors and procurement specialists seeking reliable sources of high-purity pharmaceutical intermediates, this patent offers a robust pathway that bypasses the limitations of classical synthesis. The core innovation lies in the use of readily available polyfluorinated peroxy compounds and organic sulfinates, coupled under mild basic conditions without the need for expensive transition metal catalysts.
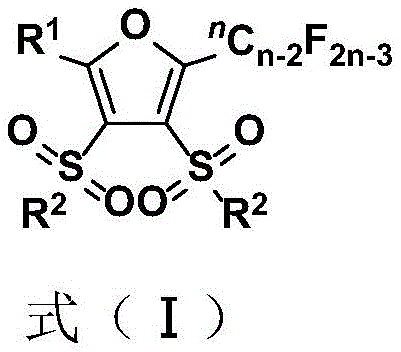
This structural versatility allows for the precise tuning of physicochemical properties, making these compounds ideal candidates for next-generation drug discovery programs. By leveraging this proprietary methodology, manufacturers can access a diverse library of fluorinated scaffolds that were previously difficult or prohibitively expensive to synthesize. The ability to introduce both perfluoroalkyl and sulfone fragments simultaneously enhances the molecular complexity and potential biological activity of the final products, addressing a critical gap in the current supply chain for specialized fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan derivatives has relied heavily on classical methodologies such as the Feist-Benary and Paal-Knorr reactions, which, while foundational, suffer from significant drawbacks in modern industrial applications. These traditional routes often necessitate the use of harsh reaction conditions, including strong inorganic acids or bases, which can lead to poor functional group compatibility and the degradation of sensitive substrates. Furthermore, many contemporary alternatives depend on transition metal catalysis, introducing the risk of toxic metal residues that require rigorous and costly removal processes to meet stringent pharmaceutical purity standards. The generation of substantial by-products and the difficulty in controlling regioselectivity further complicate the purification workflow, resulting in lower overall yields and increased waste disposal burdens. Additionally, existing methods rarely allow for the simultaneous introduction of fluoroalkyl groups during the ring construction phase, often requiring multi-step sequences that inflate production costs and extend lead times.
The Novel Approach
In stark contrast, the methodology disclosed in Patent CN110357842B introduces a transformative three-component coupling strategy that operates under remarkably mild and environmentally benign conditions. This novel approach utilizes a base-promoted reaction between polyfluorinated peroxy compounds and organic sulfinates, proceeding efficiently in an air atmosphere at moderate temperatures ranging from 50°C to 90°C. The elimination of transition metal catalysts not only simplifies the reaction setup but also ensures that the final product is free from heavy metal contamination, a critical advantage for API intermediate manufacturing. The process demonstrates exceptional substrate universality, tolerating a wide array of functional groups including halogens, nitro, cyano, and ester moieties without compromising yield or selectivity.
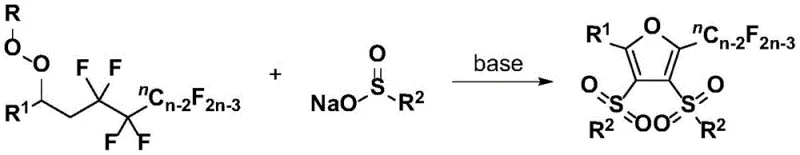
As illustrated in the reaction scheme, this one-pot synthesis achieves the selective cleavage of four carbon-fluorine bonds while constructing the furan core, a feat that significantly shortens the synthetic route. The use of simple inorganic bases like cesium carbonate and common solvents such as tert-butanol further underscores the practicality and scalability of this method for commercial production. By consolidating multiple bond-forming events into a single step, this technology drastically reduces the number of unit operations required, thereby enhancing overall process efficiency and reducing the environmental footprint associated with solvent usage and waste generation.
Mechanistic Insights into Base-Promoted C-F Bond Activation
The mechanistic elegance of this synthesis lies in its ability to activate inert carbon-fluorine bonds under relatively mild basic conditions, a challenge that has long plagued organofluorine chemistry. The reaction likely proceeds through a base-mediated activation of the polyfluorinated peroxy compound, generating a reactive intermediate that undergoes nucleophilic attack by the organic sulfinate. This initial interaction triggers a cascade of transformations involving the sequential cleavage of C-F bonds, driven by the thermodynamic stability of the resulting fluoride ions and the formation of strong sulfur-oxygen bonds in the sulfone moiety. The intramolecular cyclization step is facilitated by the proximity of the reacting centers within the transient intermediate, leading to the rapid formation of the five-membered furan ring. This mechanism avoids the high-energy barriers typically associated with C-F bond activation, allowing the reaction to proceed at temperatures as low as 50°C.
From an impurity control perspective, the absence of transition metals eliminates a major source of variability and contamination often seen in catalytic cycles. The reaction's high chemoselectivity ensures that sensitive functional groups on the aromatic rings of the sulfinate or the peroxy substrate remain intact, minimizing the formation of side products that could complicate downstream purification. The use of TLC for monitoring allows for precise determination of reaction endpoints, preventing over-reaction or decomposition of the product. Furthermore, the workup procedure involving simple aqueous washing and extraction effectively removes inorganic salts and unreacted starting materials, yielding a crude product of high purity that requires minimal chromatographic refinement. This streamlined purification profile is essential for maintaining cost-effectiveness in large-scale manufacturing environments.
How to Synthesize Fluoroalkyl-Substituted Furan Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and industrial production. The process begins with the careful selection of high-quality polyfluorinated peroxy compounds and organic sulfinates, which are mixed in a suitable solvent system. The addition of a base promoter initiates the reaction, which is then maintained under controlled thermal conditions to ensure consistent kinetics. Detailed standard operating procedures regarding stoichiometry, temperature ramps, and quenching protocols are critical for maximizing yield and reproducibility. For a comprehensive breakdown of the specific experimental parameters and safety considerations, please refer to the standardized synthesis guide below.
- Mix polyfluorinated peroxy compounds with organic sulfinates and add an alkali promoter like cesium carbonate in a solvent such as tert-butanol.
- Stir the reaction mixture under an air atmosphere at temperatures between 50°C and 90°C for 5 to 24 hours until TLC indicates completion.
- Work up the reaction by washing, extracting with ethyl acetate, drying over anhydrous sodium sulfate, and purifying via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from precious metal catalysts to inexpensive inorganic bases represents a fundamental change in the cost structure of furan production, eliminating the volatility associated with rhodium, palladium, or platinum pricing. Moreover, the simplified reaction conditions reduce the need for specialized equipment capable of handling high pressures or inert atmospheres, lowering capital expenditure requirements for new production lines. The robustness of the method against variations in substrate structure ensures a stable supply of diverse intermediates, mitigating the risk of production delays caused by raw material shortages or batch failures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commercially available, low-cost reagents such as cesium carbonate and tert-butanol significantly drive down the raw material costs per kilogram of product. By avoiding complex metal scavenging steps and extensive purification protocols required to remove trace metals, the overall processing time and utility consumption are drastically reduced. This streamlined workflow translates into substantial cost savings in both labor and energy, making the final fluoroalkyl-substituted furans highly competitive in the global market. The high atom economy of the three-component coupling further minimizes waste disposal costs, contributing to a more sustainable and economically viable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on widely sourced organic sulfinates and peroxy compounds ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The reaction's tolerance for air and moisture simplifies logistics and storage requirements, as strict anhydrous or anaerobic conditions are not necessary for successful execution. This operational flexibility allows for faster turnaround times from order to delivery, enabling suppliers to respond more agilely to fluctuating market demands. The ability to produce a broad spectrum of derivatives from a common platform technology also means that inventory risks are minimized, as the same production assets can be rapidly repurposed for different customer specifications.
- Scalability and Environmental Compliance: The mild thermal profile and ambient pressure operation of this synthesis make it inherently safer and easier to scale from pilot plant to multi-ton commercial production without significant engineering hurdles. The absence of toxic heavy metals aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals, reducing the regulatory burden associated with effluent treatment and hazardous waste management. The green chemistry attributes of this method, including reduced solvent usage and higher yields, enhance the company's environmental profile, which is becoming a key differentiator in supplier selection processes for major pharmaceutical and agrochemical corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its potential integration into their supply chains. The following questions address common inquiries regarding the operational feasibility, safety, and applicability of this technology in real-world scenarios. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this furan synthesis method over traditional Paal-Knorr reactions?
A: Unlike traditional methods requiring strong acids or toxic transition metals, this patent-disclosed method operates under mild, metal-free conditions using simple inorganic bases, significantly reducing toxic waste and purification costs.
Q: Does this process require inert gas protection?
A: No, the reaction proceeds efficiently under an air atmosphere, which simplifies the engineering requirements for large-scale production and reduces operational complexity compared to oxygen-sensitive protocols.
Q: What is the substrate scope for the R1 and R2 groups in the final furan product?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of substituents including alkyl, aryl, halogen, nitro, and cyano groups, making it highly versatile for diverse medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl-Substituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in Patent CN110357842B for the production of high-value fluorinated intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from concept to market. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug discovery or agrochemical development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can accelerate your project while optimizing your overall supply chain costs.
