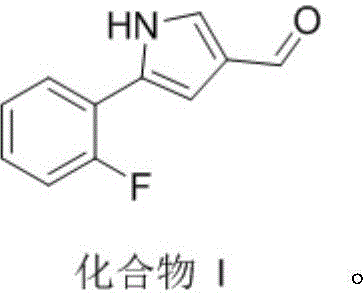
Scalable One-Pot Catalytic Synthesis of 5-(2-Fluorophenyl)-1H-Pyrrole-3-Formaldehyde for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates, particularly for high-demand drugs like Vonoprazan. Patent CN113845459B introduces a groundbreaking preparation method for 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde, a pivotal building block in the synthesis of potassium-competitive acid blockers. This innovation addresses long-standing challenges in process chemistry by utilizing a streamlined one-pot catalytic approach that transforms 2-(2-fluorobenzoyl)malononitrile directly into the target aldehyde. By integrating sequential hydrogenation and cyclization steps within a single reactor setup, this technology eliminates the need for isolating unstable intermediates, thereby enhancing operational safety and throughput. For global procurement teams and R&D directors, this represents a significant opportunity to optimize supply chains for gastroenterology therapeutics. The following analysis details the technical superiority and commercial viability of this novel route compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrrole aldehydes has relied on cumbersome multi-step sequences that impose heavy burdens on manufacturing efficiency and environmental compliance. Prior art, such as the process described in patent EP2327692, typically initiates with o-fluoro acetophenone and proceeds through a series of discrete transformations including bromination, substitution condensation, and palladium-carbon dechlorination. Each of these stages necessitates separate reaction vessels, extensive workup procedures, and the isolation of intermediates, which cumulatively drives up production costs and extends lead times. Furthermore, the reliance on stoichiometric oxidants and chlorinating agents in these traditional routes generates substantial volumes of hazardous organic waste liquids. The accumulation of these waste streams not only complicates disposal logistics but also increases the carbon footprint of the manufacturing process, making it increasingly difficult to meet modern green chemistry standards required by top-tier pharmaceutical clients.
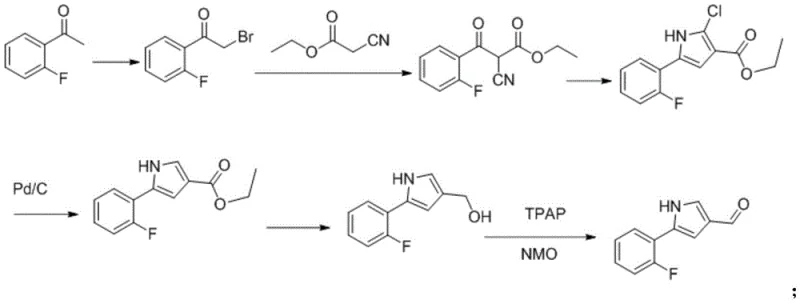
The Novel Approach
In stark contrast to the fragmented nature of legacy syntheses, the method disclosed in CN113845459B employs a convergent one-pot strategy that dramatically simplifies the molecular construction of the target compound. By starting directly from 2-(2-fluorobenzoyl)malononitrile, the process bypasses the initial halogenation and coupling steps entirely, collapsing what was once a linear sequence into a cohesive dual-reduction protocol. This approach utilizes a synergistic combination of a metal catalyst in the presence of glacial acetic acid followed by a secondary reduction with Raney nickel, facilitating both the reduction of the nitrile groups and the subsequent cyclization to form the pyrrole ring in situ. The elimination of intermediate isolation steps not only reduces the physical handling of materials but also minimizes product loss associated with filtration and drying operations. Consequently, this methodology offers a pathway to significantly higher overall yields while simultaneously reducing the consumption of solvents and reagents, aligning perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into One-Pot Hydrogenation and Cyclization
The core innovation of this synthesis lies in the precise control of catalytic hydrogenation conditions to achieve selective transformation without over-reduction or side reactions. In the first stage, the dissolution of the malononitrile precursor in a polar aprotic solvent such as tetrahydrofuran creates an optimal environment for the metal catalyst, which may include palladium carbon or platinum carbon, to activate molecular hydrogen. The addition of glacial acetic acid plays a critical dual role: it acts as a proton source to facilitate the reduction of the carbonyl and nitrile functionalities while also promoting the intramolecular cyclization required to close the pyrrole ring. Maintaining the reaction temperature between 45°C and 50°C ensures sufficient kinetic energy for the transformation while preventing thermal degradation of the sensitive aldehyde moiety. This carefully balanced chemical environment allows for the conversion of the starting material with residual levels dropping below 0.5%, setting the stage for the subsequent refinement step.
Following the initial reduction, the introduction of Raney nickel serves as a powerful refining agent to complete the reduction of any remaining intermediate states and ensure the formation of the aromatic pyrrole system. This second reduction phase is conducted at a lower temperature range of 15°C to 25°C, which is crucial for preserving the integrity of the newly formed aldehyde group against further reduction to an alcohol. The use of water in conjunction with Raney nickel aids in the hydrolysis of any imine intermediates that may form during the cyclization process, driving the equilibrium towards the desired aldehyde product. Rigorous monitoring of the reaction progress ensures that intermediate transition states are reduced to less than 0.2%, guaranteeing a high-purity profile before the workup even begins. This mechanistic precision is what enables the process to consistently deliver material with purity exceeding 99%, a critical metric for downstream API synthesis.

How to Synthesize 5-(2-Fluorophenyl)-1H-Pyrrole-3-Formaldehyde Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition of reagents and control of hydrogen pressure to maximize safety and yield. The process begins with the charging of the reactor with the malononitrile precursor and solvent, followed by the careful addition of the primary metal catalyst and acetic acid under an inert atmosphere. Once the first reduction is complete and the catalyst filtered, the reaction mixture is transferred or treated in situ with Raney nickel and water for the second hydrogenation phase. Detailed standardized synthetic steps see the guide below for exact parameters regarding pressure, temperature, and stirring rates optimized for commercial scale-up.
- Dissolve 2-(2-fluorobenzoyl)malononitrile in a solvent like tetrahydrofuran, add a metal catalyst and glacial acetic acid, then perform the first reduction reaction under hydrogen pressure at 45-50°C.
- After cooling and filtering the first catalyst, introduce Raney nickel and water to the reaction solution for a second reduction reaction under hydrogen pressure at 15-25°C.
- Filter off the Raney nickel, concentrate the filtrate under reduced pressure, and purify the crude solid by pulping in a tetrahydrofuran-water mixture to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere technical elegance. The consolidation of multiple reaction steps into a single pot significantly reduces the total processing time per batch, allowing manufacturing facilities to increase throughput without expanding physical infrastructure. This efficiency gain is compounded by the drastic reduction in solvent usage and waste generation, which lowers the operational expenditures associated with waste treatment and environmental compliance. By minimizing the number of unit operations, the process also reduces the risk of cross-contamination and human error, leading to more consistent batch-to-batch quality and reliable supply continuity for downstream customers.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive drying and purification processes that typically consume significant energy and time. Furthermore, by avoiding the use of expensive stoichiometric oxidants and chlorinating agents found in older routes, the raw material costs are substantially lowered. The ability to recycle solvents more effectively due to the cleaner reaction profile further contributes to a leaner cost structure, enabling competitive pricing for high-purity pharmaceutical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of this one-pot method makes it less susceptible to disruptions caused by the availability of niche reagents or specialized equipment. Since the process relies on standard hydrogenation technology and commonly available solvents like tetrahydrofuran and acetonitrile, sourcing risks are minimized. The simplified workflow also means that production schedules are more predictable, reducing lead time for high-purity pharmaceutical intermediates and ensuring that Just-In-Time delivery commitments to global API manufacturers can be met with greater confidence.
- Scalability and Environmental Compliance: The patent data demonstrates successful scaling from gram quantities to 100kg batches, proving the viability of this route for commercial scale-up of complex pharmaceutical intermediates. The significant reduction in three wastes (waste water, waste gas, and solid waste) aligns with increasingly stringent global environmental regulations, future-proofing the supply chain against regulatory tightening. This environmental stewardship not only mitigates compliance risks but also enhances the brand reputation of the supply chain partners involved in the production of sustainable medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing production pipelines.
Q: How does this one-pot method compare to conventional multi-step synthesis routes?
A: Unlike conventional routes such as those in EP2327692 which require bromination, substitution, dechlorination, and separate oxidation steps, this patented one-pot method combines reduction and cyclization. This significantly reduces solvent waste, eliminates intermediate isolation steps, and improves overall yield efficiency for industrial manufacturing.
Q: What purity levels can be achieved with this catalytic process?
A: According to the patent data, the process consistently achieves product purity exceeding 99% as verified by HPLC analysis. The specific workup procedure involving tetrahydrofuran-water pulping effectively removes impurities and residual catalysts, ensuring the material meets stringent specifications for API intermediate usage.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly demonstrates scalability with examples ranging from gram scale up to 100kg batches in 1000L reactors. The use of standard hydrogenation equipment and common solvents like tetrahydrofuran and acetonitrile ensures that the process is readily transferable to commercial manufacturing facilities without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-Fluorophenyl)-1H-Pyrrole-3-Formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and unwavering commitment to quality. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-(2-fluorophenyl)-1H-pyrrole-3-formaldehyde meets the exacting standards required for modern drug development. We are dedicated to supporting your R&D and commercial goals through superior process execution and supply chain stability.
We invite you to collaborate with us to optimize your sourcing strategy for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance the efficiency and profitability of your pharmaceutical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
