Advanced Pd-Catalyzed Carbonylation for Efficient 2-Trifluoromethyl Quinazolinone Production
Advanced Pd-Catalyzed Carbonylation for Efficient 2-Trifluoromethyl Quinazolinone Production
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. A significant breakthrough in this domain is documented in patent CN113045503B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology addresses critical bottlenecks in accessing these bioactive cores by utilizing a transition metal palladium-catalyzed carbonylation cascade. The introduction of the trifluoromethyl group is strategically vital, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, properties that are essential for developing next-generation therapeutics ranging from antifungal agents to anticancer drugs. By leveraging cheap and readily available starting materials, this innovation provides a scalable pathway for producing high-purity intermediates.
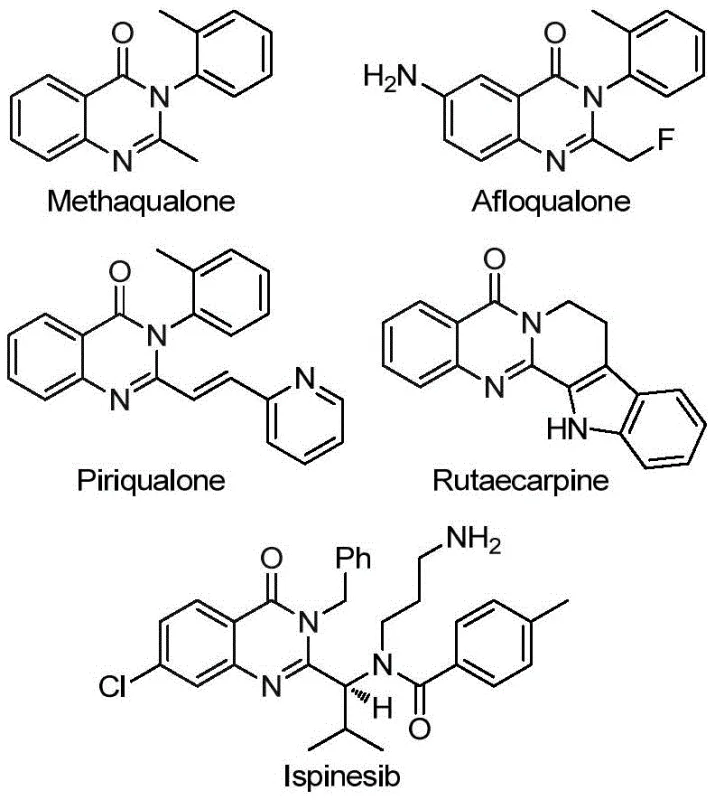
Quinazolinone derivatives are ubiquitous in nature and pharmacology, exemplified by market-approved drugs such as Methaqualone and Afloqualone, as well as natural products like Rutaecarpine shown in the structural overview. The ability to functionalize these cores with trifluoromethyl groups opens new avenues for drug discovery, yet traditional synthetic approaches have often been plagued by limitations. The methodology described in CN113045503B represents a paradigm shift, moving away from stoichiometric activation towards catalytic efficiency. For R&D directors and process chemists, understanding the nuances of this transformation is key to integrating it into existing pipelines for API intermediate production, ensuring both chemical elegance and economic viability in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinones has relied on methodologies that impose significant operational and economic burdens on chemical manufacturing. Common literature procedures often involve the cyclization of anthranilamides with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. These routes frequently suffer from harsh reaction requirements, necessitating extreme temperatures or pressures that complicate reactor design and safety protocols. Furthermore, alternative strategies utilizing isatoic anhydride or T3P-promoted cascade reactions often depend on expensive, pre-activated substrates that drive up the cost of goods sold (COGS). Another critical drawback is the narrow substrate scope; many conventional methods fail to tolerate diverse functional groups, limiting their utility in the late-stage functionalization of complex drug candidates. Additionally, the use of unstable trifluoroacetamides or gaseous carbon monoxide directly poses significant safety hazards and handling difficulties in a commercial plant setting, making these older routes less attractive for modern, safety-conscious supply chains.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a palladium-catalyzed carbonylation cascade that fundamentally simplifies the synthetic logic. This method employs trifluoroethylimidoyl chloride and various amines as starting materials, which are not only commercially available but also cost-effective compared to specialized anhydrides. The reaction proceeds smoothly in an organic solvent like 1,4-dioxane at a moderate temperature of 110°C, eliminating the need for cryogenic conditions or high-pressure autoclaves typically associated with carbonylation chemistry. Crucially, the system uses TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, which releases CO in situ upon heating. This innovation drastically improves operational safety by avoiding the direct handling of toxic CO gas. The process demonstrates exceptional functional group tolerance, accommodating halogens, alkyl groups, and electron-withdrawing substituents on the aromatic ring, thereby enabling the synthesis of a diverse library of analogs for SAR studies without extensive protecting group manipulations.
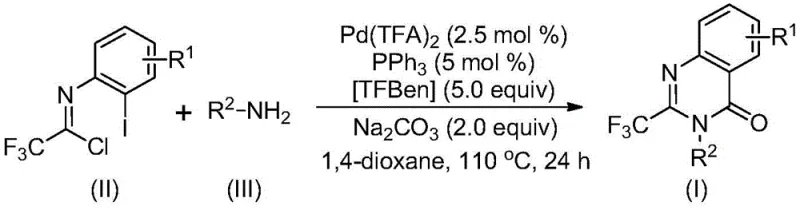
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis that ensures high selectivity and yield. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the trifluoroethylimidoyl chloride and the amine, generating a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst, generated from Pd(TFA)2 and PPh3, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium intermediate. At this stage, the TFBen additive plays a pivotal role; under the thermal conditions of 110°C, it decomposes to release carbon monoxide. This CO molecule then inserts into the carbon-palladium bond, creating an acyl-palladium species. This insertion step is the defining feature that constructs the carbonyl functionality of the quinazolinone core. Following CO insertion, the base facilitates the formation of a palladium-nitrogen bond, closing the ring to form a seven-membered palladacycle intermediate. The catalytic cycle concludes with a reductive elimination step, which releases the final 2-trifluoromethyl-substituted quinazolinone product and regenerates the active Pd(0) catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or harsh acidic cyclizations. The concerted nature of the palladium cycle minimizes the formation of side products typically associated with uncontrolled polymerization or decomposition of sensitive trifluoromethyl groups. The use of a solid CO surrogate like TFBen ensures a steady, low-concentration supply of carbon monoxide, preventing the formation of bis-carbonylated byproducts that can occur when CO pressure is too high. Furthermore, the compatibility with sodium carbonate as a mild base prevents the hydrolysis of the imidoyl chloride starting material before it can react with the amine, thereby maximizing atom economy. For quality control teams, this translates to a cleaner crude reaction profile, reducing the burden on downstream purification processes such as column chromatography or recrystallization, and ultimately leading to higher purity specifications for the final API intermediate.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation in a standard laboratory or pilot plant setting. The protocol requires precise stoichiometry to maintain catalytic efficiency, specifically utilizing a molar ratio of trifluoroethylimidoyl chloride to amine to palladium catalyst of approximately 1:2.5:0.025. The reaction is conducted in 1,4-dioxane, which has been identified as the optimal solvent for solubilizing all reagents while promoting the catalytic cycle. Operators should note that while other aprotic solvents like THF or acetonitrile can support the reaction, dioxane provides superior conversion rates. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined below to ensure consistent batch-to-batch quality.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in 1,4-dioxane solvent.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the raw material supply chain. By utilizing trifluoroethylimidoyl chlorides and simple amines, manufacturers can bypass the volatility and price fluctuations associated with specialized fluorinating reagents or anhydrides. The reliance on a palladium catalyst, while requiring recovery protocols, is offset by the low loading (2.5 mol%) and the high turnover number demonstrated in the examples, where yields frequently exceed 90%. This efficiency directly correlates to reduced waste generation and lower disposal costs, aligning with modern green chemistry mandates. Furthermore, the elimination of high-pressure gas equipment reduces capital expenditure (CAPEX) for facility upgrades, allowing existing reactors to be repurposed for this chemistry with minimal modification.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the use of commodity chemicals rather than bespoke reagents. The substitution of expensive activating agents like T3P or trifluoroacetic anhydride with inexpensive sodium carbonate and TFBen results in substantial cost savings per kilogram of product. Additionally, the high reaction efficiency minimizes the loss of valuable fluorinated starting materials, which are often the most costly component of the bill of materials. The simplified workup procedure, involving basic filtration and silica treatment, reduces solvent consumption and labor hours compared to multi-step extractions or complex distillations required by older methods. These factors combine to lower the overall manufacturing cost, providing a competitive edge in pricing for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is critical for API production, and this method enhances reliability by relying on widely available starting materials. Trifluoroethylimidoyl chlorides and various substituted anilines are produced by multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions—tolerating moisture and air better than many organometallic processes—reduces the likelihood of batch failures due to minor environmental deviations. This resilience ensures consistent delivery schedules to downstream customers. Moreover, the scalability of the process from gram to kilogram scales without significant re-optimization means that supply can be ramped up quickly to meet surging demand for clinical trial materials or commercial launch stocks without lengthy process validation delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly cleaner. The use of a solid CO surrogate eliminates the regulatory hurdles and safety risks associated with storing and transporting carbon monoxide cylinders. The reaction generates fewer hazardous byproducts, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing site. The high atom economy ensures that a larger proportion of the input mass ends up in the final product, minimizing solid waste. For facilities operating under strict environmental regulations, this technology offers a compliant pathway to produce complex heterocycles. The ability to run the reaction at atmospheric pressure in standard glass-lined steel reactors further facilitates seamless scale-up from pilot to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these specifics helps in assessing the feasibility of integrating this chemistry into existing production workflows and ensures that all stakeholders are aligned on the capabilities and limitations of the process.
Q: What are the key advantages of this Pd-catalyzed method over traditional cyclization?
A: Unlike traditional methods requiring harsh conditions or unstable reagents like trifluoroacetic anhydride, this protocol uses stable imidoyl chlorides and a solid CO surrogate (TFBen), offering milder conditions (110°C) and broader substrate tolerance.
Q: Is this method suitable for synthesizing complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield synthesis of Rutaecarpine, achieving a 77% overall yield through a three-step sequence involving cyclization and hydrolysis.
Q: What is the role of TFBen in this reaction system?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and effective carbon monoxide surrogate. Under heating conditions, it releases CO in situ, which inserts into the palladium-carbon bond to form the necessary acyl intermediate without requiring high-pressure CO gas.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The technological potential of 2-trifluoromethyl quinazolinones is immense, serving as a cornerstone for developing novel therapeutics with improved pharmacokinetic profiles. At NINGBO INNO PHARMCHEM, we recognize the complexity involved in translating such advanced academic and patent methodologies into commercial reality. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our infrastructure is equipped to handle the specific requirements of palladium-catalyzed reactions, including efficient metal scavenging systems to meet stringent purity specifications for pharmaceutical ingredients. Our rigorous QC labs ensure that every batch of high-purity 2-trifluoromethyl quinazolinone intermediates meets the exacting standards required by global regulatory bodies, guaranteeing consistency and safety for your drug development programs.
We invite you to leverage our expertise to accelerate your project timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and target markets. We encourage you to contact our technical procurement team to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us collaborate to bring your next-generation quinazolinone-based drugs from the bench to the market with speed, efficiency, and uncompromised quality.
