Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Manufacturing
Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Manufacturing
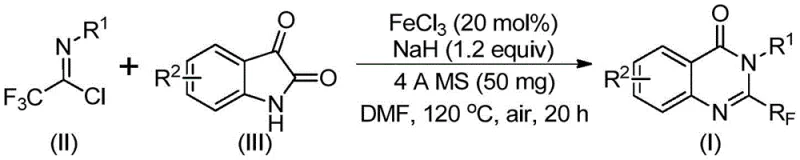
The pharmaceutical industry continuously seeks robust and economically viable pathways to access bioactive heterocyclic scaffolds, particularly those containing fluorine motifs which are renowned for enhancing metabolic stability and lipophilicity. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current synthetic methodologies. This technology leverages a cost-effective iron-catalyzed cyclization strategy that transforms readily available isatin derivatives and trifluoroethylimidoyl chlorides into high-value intermediates. For R&D directors and procurement specialists, this represents a paradigm shift away from expensive precious metal catalysis towards sustainable base-metal chemistry. The process not only demonstrates excellent functional group tolerance but also operates under relatively mild conditions compared to traditional harsh cyclization protocols, making it an ideal candidate for the reliable pharmaceutical intermediate supplier looking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core bearing a trifluoromethyl group has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoromethylating agents. While effective in academic settings, these legacy routes suffer from severe practical limitations when translated to commercial manufacturing. Common trifluoromethyl sources like trifluoroacetic anhydride or ethyl trifluoroacetate often require stoichiometric amounts of strong bases or acids, leading to corrosive waste streams and difficult purification challenges. Furthermore, these reactions frequently exhibit narrow substrate scope, failing to accommodate sensitive functional groups without extensive protection-deprotection sequences. The reliance on expensive starting materials and the generation of significant chemical waste result in inflated production costs and extended lead times, creating substantial friction for supply chain heads aiming for lean manufacturing operations.
The Novel Approach
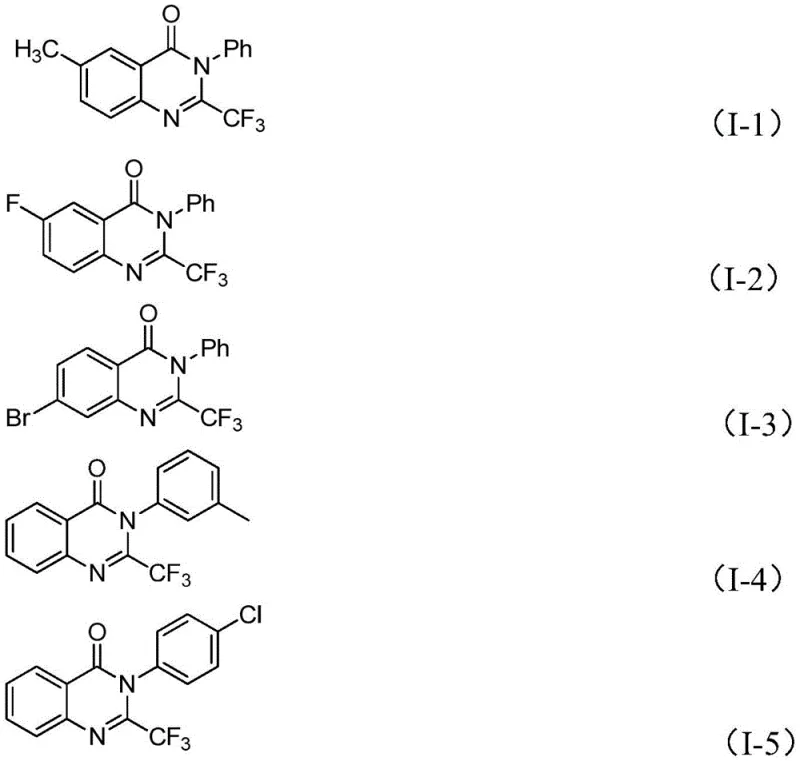
In stark contrast, the methodology outlined in CN111675662B introduces a streamlined convergent synthesis utilizing isatin and trifluoroethylimidoyl chloride as key building blocks. This novel approach capitalizes on the inherent reactivity of the isatin carbonyl system, which undergoes a facile condensation followed by an iron-mediated decarbonylative cyclization. By employing ferric chloride as the catalyst, the process bypasses the need for costly noble metals while maintaining high catalytic efficiency. The reaction proceeds through a distinct mechanistic pathway that tolerates a wide array of substituents, including halogens, alkyl, and alkoxy groups, as evidenced by the successful synthesis of diverse derivatives. This strategic redesign of the synthetic route significantly reduces the complexity of the operation, offering a direct path to high-purity 2-trifluoromethyl quinazolinones with minimized environmental impact.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique role of the ferric chloride and sodium hydride system in promoting carbon-nitrogen bond formation and subsequent ring closure. Mechanistically, the reaction initiates with the base-promoted nucleophilic attack of the isatin nitrogen on the imidoyl chloride, forming a transient trifluoroacetamidine intermediate. The presence of 4A molecular sieves is crucial at this stage to sequester moisture and drive the equilibrium forward. Subsequently, the iron catalyst facilitates a decarbonylation event, effectively removing the carbonyl oxygen from the isatin moiety and enabling the aromatization of the pyrimidine ring. This cascade transformation is highly efficient, converting simple precursors into complex fused heterocycles in a single pot. Understanding this mechanism allows process chemists to fine-tune reaction parameters, ensuring that the delicate balance between condensation and cyclization is maintained to maximize yield and minimize the formation of hydrolysis byproducts.
Impurity control is another critical aspect where this iron-catalyzed system excels. Traditional methods often struggle with the over-fluorination or incomplete cyclization of substrates, leading to difficult-to-remove impurities that compromise the quality of the final API intermediate. The specific temperature profile employed in this patent—starting at 40 °C and ramping to 120 °C—provides a kinetic window that favors the desired pathway. The initial lower temperature allows for the controlled formation of the N-C bond without triggering rapid decomposition of the sensitive imidoyl chloride. The subsequent heating phase provides the activation energy required for the decarbonylation step. This staged thermal approach ensures that side reactions are kinetically suppressed, resulting in a cleaner crude reaction mixture that simplifies downstream purification and enhances the overall purity profile of the product.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory scale-up and industrial production. The procedure involves charging a reactor with ferric chloride, sodium hydride, and activated 4A molecular sieves in an anhydrous polar aprotic solvent such as DMF. To this mixture, the trifluoroethylimidoyl chloride and the substituted isatin are added under an air atmosphere, eliminating the need for inert gas handling which further reduces operational costs. The reaction mixture is stirred at 40 °C for approximately 10 hours to allow the initial coupling, followed by heating to 120 °C for an additional 20 hours to complete the cyclization. Upon completion, the workup is straightforward, involving filtration to remove solids and standard column chromatography or crystallization to isolate the pure product. For detailed standard operating procedures and safety guidelines regarding the handling of sodium hydride and specific purification protocols, please refer to the technical documentation below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in DMF solvent.
- Stir the mixture at 40 °C for 10 hours to initiate the reaction, then increase temperature to 120 °C.
- Maintain reaction at 120 °C for 20 hours under air, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from precious metal catalysts to abundant iron salts represents a fundamental reduction in raw material volatility and cost exposure. Unlike palladium or rhodium, whose prices fluctuate wildly based on geopolitical factors, iron is a commodity chemical with a stable and secure global supply chain. Furthermore, the elimination of expensive ligands and the use of air as the oxidant atmosphere drastically simplify the bill of materials. This simplification not only lowers the direct cost of goods sold but also reduces the logistical burden associated with sourcing specialized reagents, thereby enhancing the resilience of the supply network against disruptions.
- Cost Reduction in Manufacturing: The economic impact of replacing noble metal catalysts with ferric chloride cannot be overstated in the context of large-scale manufacturing. By removing the necessity for expensive metal scavengers and complex purification steps required to meet strict residual metal limits in pharmaceuticals, the overall processing cost is significantly optimized. Additionally, the high atom economy of the decarbonylative cyclization means less waste is generated per kilogram of product, leading to reduced waste disposal fees and lower solvent consumption. These cumulative efficiencies result in a substantially more competitive cost structure for the final active pharmaceutical ingredient, allowing for better margin management in a price-sensitive market.
- Enhanced Supply Chain Reliability: The robustness of the starting materials—isatin derivatives and trifluoroethylimidoyl chlorides—ensures a steady and reliable flow of inputs for production. These chemicals are widely produced by multiple vendors globally, mitigating the risk of single-source dependency. The tolerance of the reaction to various functional groups also means that supply chain managers have greater flexibility in sourcing different substituted isatins without needing to revalidate the entire process for each new analog. This versatility shortens the lead time for introducing new derivatives to the market and ensures continuity of supply even if specific precursor availability fluctuates, providing a buffer against market volatility.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with modern green chemistry principles. The use of iron, a non-toxic metal, simplifies the environmental compliance landscape compared to processes generating heavy metal waste. The reaction conditions, while requiring elevated temperatures, do not necessitate extreme pressures or cryogenic cooling, making the technology easily transferable to existing standard stainless steel reactors. This ease of scale-up from gram to multi-ton quantities ensures that production can be ramped up quickly to meet surging demand without requiring capital-intensive infrastructure upgrades, facilitating a smoother transition from pilot plant to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational advantages compared to precious metal catalysts or harsh acidic conditions. It is inexpensive, readily available, and eliminates the need for complex heavy metal removal steps often required with palladium or rhodium systems, thereby simplifying downstream processing and reducing overall production costs.
Q: How does the two-stage temperature profile improve reaction yield?
A: The patented process utilizes a specific temperature gradient, starting at 40 °C for 10 hours followed by heating to 120 °C for 20 hours. This controlled thermal profile facilitates the initial formation of intermediate bonds without premature decomposition, while the subsequent higher temperature drives the decarbonylation and cyclization to completion, ensuring high conversion rates and minimizing side products.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is designed for scalability. The use of common solvents like DMF, air as the oxidant atmosphere, and robust inorganic reagents makes the process highly adaptable for large-scale manufacturing. The tolerance for various functional groups on the substrate further enhances its utility for producing diverse libraries of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis described in CN111675662B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal profiles and reagent handling requirements of this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to delivering consistent quality and reliability, acting as a true extension of your R&D and supply chain teams.
We invite you to leverage our technical expertise to optimize your supply chain for 2-trifluoromethyl quinazolinones and related heterocyclic scaffolds. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Let us help you navigate the complexities of modern chemical manufacturing with confidence and precision.
