Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalysis
Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for highly efficient and structurally diverse chiral catalysts. A significant breakthrough in this domain is detailed in Chinese Patent CN110452150B, which discloses a novel class of axial chiral indole-naphthalene compounds and a robust preparation method thereof. These compounds, characterized by the general structure shown in Formula 9, represent a critical advancement in organocatalysis, offering a streamlined pathway to access complex chiral scaffolds that were previously difficult to synthesize with high fidelity. For R&D directors and procurement specialists in the fine chemical sector, this technology presents a compelling opportunity to enhance the supply chain of high-value chiral intermediates. The methodology leverages a sophisticated yet operationally simple organocatalytic system, moving away from harsh transition metal processes toward more sustainable and selective small-molecule catalysis.
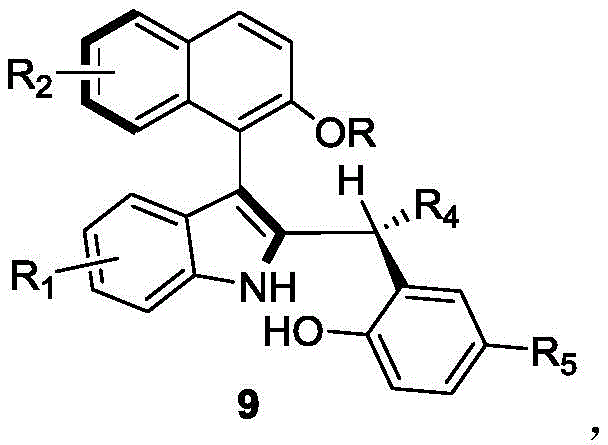
The core innovation lies in the ability to construct the axially chiral biaryl skeleton in a single step from readily available racemic starting materials. This approach not only reduces the number of synthetic steps but also inherently addresses the challenge of controlling axial chirality, a feature essential for the performance of next-generation catalysts in pharmaceutical and agrochemical manufacturing. By establishing a reliable protocol for generating these structures, the patent opens new avenues for the development of specialized ligands and catalysts that can drive asymmetric transformations with unprecedented precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral indole-naphthalene frameworks has relied heavily on cross-coupling reactions between pre-functionalized indole and naphthalene derivatives. These traditional pathways often suffer from significant drawbacks, including the requirement for expensive transition metal catalysts, stringent reaction conditions, and the necessity for protecting group manipulations that increase waste and cost. Furthermore, achieving high enantioselectivity in these coupling reactions frequently demands chiral ligands that are themselves costly and difficult to prepare. The existing literature, such as reports in Angewandte Chemie and Nature Chemistry, highlights that while some progress has been made using chiral phosphoric acids for coupling 2-indolylmethanol with naphthols, the scope remains limited. There is a distinct lack of methods capable of efficiently resolving racemic mixtures to form these specific axial chiral skeletons directly, creating a bottleneck in the production of diverse catalyst libraries.
The Novel Approach
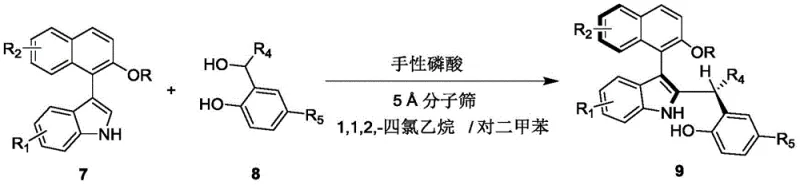
The method described in CN110452150B fundamentally shifts the paradigm by employing a dynamic kinetic resolution strategy via asymmetric addition. Instead of coupling two separate aromatic systems, the process reacts a specific indole-naphthol derivative (Formula 7) with a benzylic alcohol derivative (Formula 8) under the influence of a chiral phosphoric acid catalyst. This reaction proceeds under remarkably mild conditions, typically at temperatures between 20°C and 30°C, utilizing a mixed solvent system of 1,1,2,2-tetrachloroethane and p-xylene. The inclusion of 5A molecular sieves plays a crucial role in driving the equilibrium forward by removing water generated during the addition. This one-step construction from racemic原料 (raw materials) eliminates the need for prior resolution of starting materials, thereby maximizing atom economy and significantly reducing the overall cost of goods sold (COGS) for the final chiral product.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
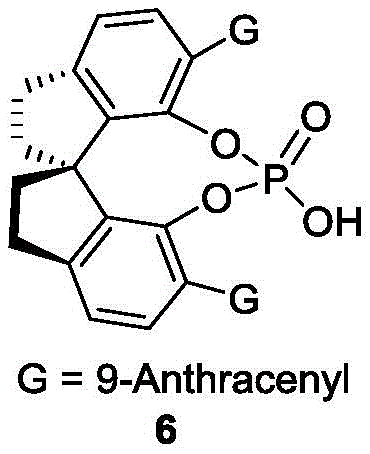
The success of this transformation hinges on the precise interaction between the substrate and the chiral phosphoric acid catalyst. The catalyst, particularly the spiro skeleton derivative designated as Formula 6, acts as a bifunctional activator. Through a network of hydrogen bonds, the phosphoryl oxygen and the acidic proton of the catalyst simultaneously activate the electrophilic imine or carbocation intermediate derived from the alcohol and the nucleophilic indole ring. This dual activation lowers the energy barrier for the C-C bond formation while imposing a rigid chiral environment that dictates the trajectory of the attack. The bulky substituents on the catalyst backbone, such as the 9-anthracenyl groups, create a steric pocket that effectively discriminates between the pro-chiral faces of the reacting species, leading to the observed high enantioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages. The high specificity of the organocatalyst minimizes the formation of regioisomers and side products that are common in metal-catalyzed couplings. The reaction profile suggests that the rate of the desired asymmetric addition far exceeds that of non-selective background reactions, ensuring that the crude product mixture is clean and easy to purify. This inherent selectivity reduces the burden on downstream processing, allowing for simpler purification protocols such as standard silica gel column chromatography. For manufacturing teams, this translates to higher throughput and reduced solvent consumption, aligning with green chemistry principles while maintaining the rigorous purity standards required for catalytic applications.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and reproducibility, making it highly attractive for scale-up. The process involves mixing the indole-naphthol substrate and the benzylic alcohol in the specified solvent blend, followed by the addition of the catalyst and molecular sieves. The reaction is allowed to proceed at ambient temperature, monitored by thin-layer chromatography (TLC) until the starting material is consumed. Workup involves simple filtration to remove the desiccant, concentration of the filtrate, and purification. This straightforward workflow minimizes the need for specialized equipment or hazardous reagents, facilitating a smooth transfer from laboratory discovery to pilot plant production.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene.
- Add 5A molecular sieves and a chiral phosphoric acid catalyst (specifically formula 6 derivatives) to the mixture.
- Stir the reaction at 20-30°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The shift from multi-step metal-catalyzed sequences to a one-step organocatalytic process drastically simplifies the supply chain complexity. By eliminating the need for precious metal catalysts and their associated removal scavengers, the process reduces both material costs and the environmental footprint related to heavy metal waste disposal. The use of commercially available and economically priced raw materials further enhances the cost-effectiveness of the route, ensuring stable pricing even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and complex ligand systems leads to significant cost savings. Additionally, the mild reaction conditions (20-30°C) reduce energy consumption compared to high-temperature coupling reactions. The simplified workup procedure, which avoids complex aqueous extractions often needed to remove metal residues, further lowers operational expenditures and waste treatment costs.
- Enhanced Supply Chain Reliability: The starting materials, Formula 7 and Formula 8 derivatives, are synthesized via established routes or are commercially accessible, reducing the risk of raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or moisture, ensuring consistent batch-to-bquality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable at ambient temperatures and the absence of hazardous reagents. The use of organic small molecule catalysts aligns with increasingly stringent regulatory requirements regarding metal impurities in pharmaceutical intermediates. This compliance facilitates easier registration and approval of the final products in regulated markets, speeding up time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axial chiral compounds. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: Unlike traditional methods that often require complex coupling of pre-functionalized rings, this patent describes a dynamic kinetic resolution approach. It constructs the axial chiral skeleton in a single step directly from racemic raw materials, significantly simplifying the synthetic route and improving atom economy.
Q: What level of enantioselectivity can be achieved with this process?
A: The process utilizes specific chiral phosphoric acid catalysts, such as the spiro skeleton derivative (Formula 6), to achieve excellent stereocontrol. Experimental data in the patent indicates enantiomeric ratios (er) as high as 98:2, ensuring high optical purity suitable for demanding catalytic applications.
Q: Can these compounds be further derivatized into functional catalysts?
A: Yes, the resulting axial chiral indole-naphthalene compounds serve as versatile precursors. The patent demonstrates their conversion into chiral phosphine catalysts (e.g., Compound 12), which have been proven effective in promoting asymmetric [4+1] cyclization reactions with high yield and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN110452150B for the asymmetric catalysis sector. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these sophisticated molecules to the global market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and purification requirements of this process, ensuring that we can meet your demand for high-purity axial chiral indole-naphthalene compounds with stringent purity specifications and rigorous QC labs.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific catalytic needs. Whether you require custom synthesis of specific derivatives or large-scale supply of the core scaffold, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project requirements. Let us help you accelerate your R&D timeline with reliable, high-quality chiral intermediates.
