Revolutionizing Prostaglandin Synthesis: A Novel Intermediate for Efficient Commercial Manufacturing
Revolutionizing Prostaglandin Synthesis: A Novel Intermediate for Efficient Commercial Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways to produce complex active pharmaceutical ingredients (APIs), particularly for cardiovascular treatments where demand is high. A pivotal advancement in this domain is detailed in patent CN107325122B, which introduces a novel intermediate structure specifically designed for the synthesis of prostaglandins, such as Beraprost sodium. This compound, known chemically as a racemic mixture containing four stereoisomers, is critical for treating chronic arterial occlusive diseases due to its potent antiplatelet and vasodilatory properties. The traditional synthesis of such molecules has long been plagued by excessive step counts and hazardous reagents, creating bottlenecks for reliable pharmaceutical intermediate suppliers. The new technology described in this patent offers a transformative solution by introducing a brand-new intermediate, designated as Formula I, which streamlines the entire production workflow. By adopting this innovative precursor, manufacturers can achieve a drastic reduction in synthesis steps while simultaneously improving overall yield and operational safety. This report provides a deep technical analysis of this breakthrough, evaluating its mechanistic advantages and commercial viability for global supply chains.
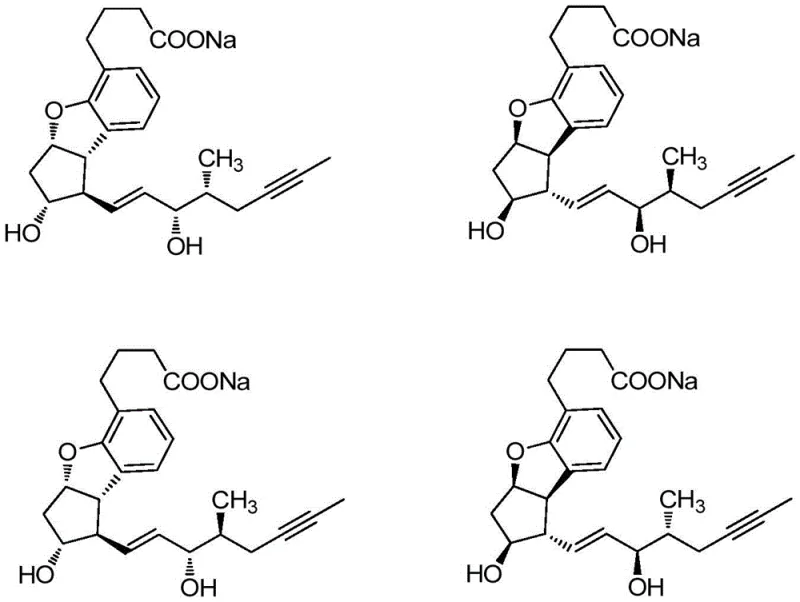
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of prostaglandin derivatives like Beraprost sodium has relied on arduous multi-step routes that are inefficient for modern commercial scale-up. One prominent legacy method, described in patents such as US5202447, initiates from cyclopentadiene. As illustrated in the reaction schemes of the prior art, this pathway involves a plethora of synthetic transformations that result in very low overall yields. The complexity of managing stereochemistry across so many steps introduces significant opportunities for impurity generation, complicating downstream purification and increasing waste. Another common approach, outlined in documents like WO2004/005274, utilizes phenol as a starting material. While this route offers slightly better yields than the cyclopentadiene method, it suffers from a critical flaw: the reliance on osmium tetroxide for double bond cleavage oxidation. Osmium tetroxide is not only extremely expensive but also highly toxic, posing severe safety risks to personnel and requiring specialized containment infrastructure. Furthermore, the 1,4-addition reactions in these older routes often suffer from poor regioselectivity and low conversion rates, necessitating extensive recycling of materials and driving up the cost of goods sold (COGS).
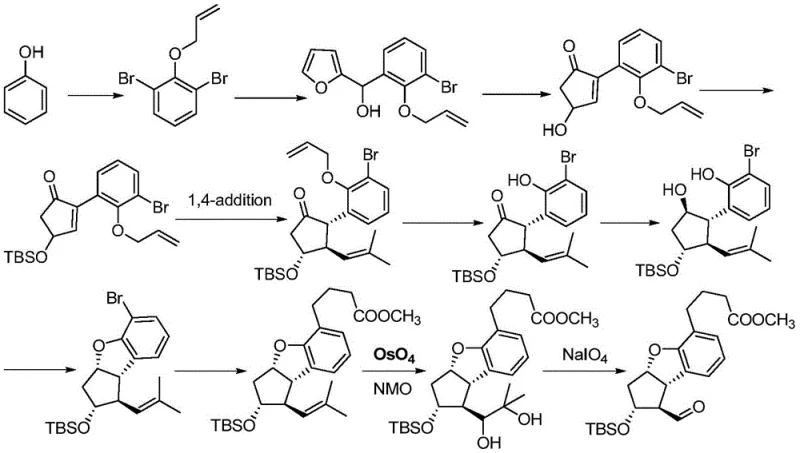
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN107325122B presents a streamlined strategy centered around the novel intermediate Formula I. This approach fundamentally reimagines the construction of the core bicyclic framework. Instead of building complexity slowly from simple dienes or relying on dangerous oxidants, the new method employs a convergent strategy. The core innovation lies in the early establishment of the critical carbon-carbon bonds and stereocenters through a highly controlled organocopper-mediated addition. This allows for the rapid assembly of the cyclopentane-fused benzofuran skeleton with high fidelity. The subsequent steps involve robust and scalable transformations, such as palladium-catalyzed deprotection and intramolecular cyclization, which proceed under mild conditions. By bypassing the need for osmium tetroxide and reducing the total number of unit operations, this novel approach offers a clear path toward cost reduction in API manufacturing. The structural integrity of Formula I serves as a versatile platform, enabling the efficient derivation of the final prostaglandin sodium salt with significantly improved process economics.
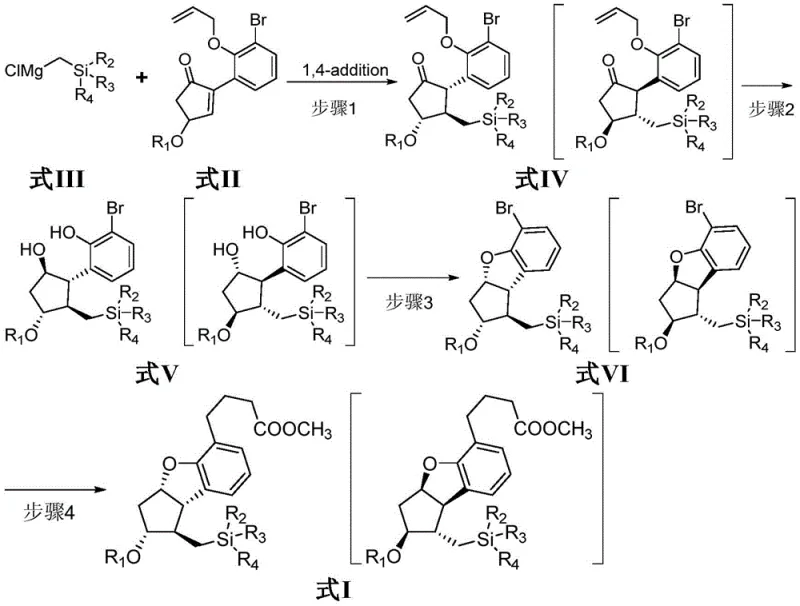
Mechanistic Insights into Organocopper 1,4-Addition and Cyclization
The heart of this synthetic breakthrough is the initial 1,4-addition reaction, which sets the stage for the entire molecular architecture. In Step 1 of the process, a silyl-substituted Grignard reagent (Formula III) is transmetallated with a halogenated cuprous salt, preferably cuprous iodide, in tetrahydrofuran (THF) at cryogenic temperatures ranging from -60°C to -20°C. This generates a reactive organocopper species in situ. When this nucleophile encounters the enone substrate (Formula II), it undergoes a conjugate addition with exceptional regioselectivity. The presence of the bulky silyl groups plays a crucial role in directing the stereochemical outcome, ensuring that the substituents are positioned correctly for the subsequent ring closure. The patent data indicates that maintaining the temperature between -40°C and -30°C is optimal, yielding up to 76-85% of the desired ketone intermediate (Formula IV). This precision in temperature control minimizes side reactions such as 1,2-addition or polymerization, which are common pitfalls in enone chemistry. The resulting intermediate possesses the necessary functionality to undergo further elaboration without requiring tedious protection-deprotection sequences that would otherwise inflate the step count.
Following the initial bond formation, the synthesis proceeds through a cleverly designed cascade of functional group manipulations to lock in the stereochemistry. Step 3 involves an intramolecular cyclization to form the benzofuran ring, a defining feature of the prostaglandin core. This is achieved by activating the phenolic hydroxyl group using triphenylphosphine and an azo condensing agent, specifically diisopropyl azodicarboxylate (DIAD), at temperatures between -50°C and -20°C. This Mitsunobu-type cyclization is highly efficient, converting the linear precursor into the rigid bicyclic structure of Formula VI with yields reaching 85%. The mechanism relies on the formation of an alkoxyphosphonium ion, which is then displaced by the proximal phenol oxygen. This step is critical for impurity control; by ensuring complete conversion here, the formation of open-chain byproducts is suppressed. The final installation of the carboxylic acid side chain via a palladium-catalyzed coupling with methyl 3-butenoate completes the scaffold. The use of 9-boron bicyclo(3,3,1)-nonane (9-BBN) ensures selective hydroboration, preventing isomerization of the sensitive alkene moieties. This mechanistic elegance translates directly into a cleaner crude product profile, reducing the burden on purification teams.
How to Synthesize Novel Prostaglandin Intermediate Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters, particularly temperature and reagent stoichiometry, to maximize the benefits of this novel route. The process is divided into four distinct stages, beginning with the preparation of the organometallic reagent and concluding with the side-chain attachment. Each step has been optimized in the patent examples to demonstrate reproducibility and scalability. For instance, the selection of sodium borohydride over lithium or potassium borohydride in the reduction step (Step 2) was found to provide superior yields (74% vs 44-52%), highlighting the importance of reagent screening. Similarly, the choice of DIAD as the condensing agent in the cyclization step proved far more effective than diethyl or dimethyl analogues. Operators must ensure strict anhydrous conditions, especially during the Grignard formation and organocopper generation phases, to prevent quenching of the reactive intermediates. The following guide outlines the standardized procedure derived from the patent disclosure, serving as a blueprint for process chemists aiming to implement this technology.
- Perform a 1,4-addition reaction between a silyl-substituted Grignard reagent (Formula III) and a cyclopentenone derivative (Formula II) using cuprous iodide at -40°C to -30°C to form Formula IV.
- Execute deprotection of the allyl group using palladium tetrakis followed by reduction with sodium borohydride at -5°C to 0°C to yield the diol intermediate Formula V.
- Conduct an intramolecular cyclization using triphenylphosphine and diisopropyl azodicarboxylate (DIAD) at -35°C to -30°C to construct the benzofuran ring system (Formula VI).
- Complete the synthesis by coupling the side chain via a Suzuki-Miyaura type reaction using 9-BBN and methyl 3-butenoate with a palladium catalyst at 50°C to 60°C to obtain Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new intermediate represents a strategic opportunity to optimize the sourcing of high-purity pharmaceutical intermediates. The primary value driver is the substantial simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the use of osmium tetroxide, companies can avoid the high costs associated with purchasing, handling, and disposing of this hazardous heavy metal. Furthermore, the reduction in synthetic steps means less solvent consumption, lower energy usage for heating and cooling cycles, and decreased labor hours per kilogram of output. These factors combine to create a significantly leaner cost structure, allowing for more competitive pricing in the global market without sacrificing margin. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more consistent supply of material for downstream API production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and toxic reagents like osmium tetroxide, which require specialized waste treatment protocols. Additionally, the higher yields observed in key steps, such as the 85% yield in the cyclization reaction, mean that less raw material is wasted. The convergence of the synthesis allows for larger batch sizes with fewer unit operations, effectively lowering the fixed cost allocation per unit. This efficiency gain is critical for maintaining profitability in the face of fluctuating raw material prices.
- Enhanced Supply Chain Reliability: The starting materials for this route, including substituted phenols and silyl chlorides, are commodity chemicals with stable global supply chains. Unlike specialized chiral pool starting materials that may have limited availability, these precursors can be sourced from multiple vendors, mitigating the risk of supply disruption. The simplified process flow also reduces the lead time required to manufacture the intermediate, enabling faster response to market demand spikes. This reliability is essential for maintaining continuous API production schedules and meeting regulatory delivery commitments.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior to legacy methods. The absence of osmium removes a major regulatory hurdle and reduces the facility's environmental footprint. The reaction temperatures, while requiring cooling capability, are within the range of standard industrial chillers (-40°C), avoiding the need for cryogenic infrastructure. The use of standard solvents like THF and methanol facilitates solvent recovery and recycling, aligning with green chemistry principles. This makes the process highly scalable from pilot plant to multi-ton commercial production with minimal environmental impact.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent literature. Understanding these nuances is vital for assessing the feasibility of integrating this intermediate into your existing manufacturing portfolio. The following section addresses key concerns regarding purity, safety, and scalability.
Q: How does the new intermediate Formula I improve upon traditional prostaglandin synthesis routes?
A: The novel intermediate significantly shortens the synthetic route compared to traditional methods starting from cyclopentadiene or phenol. It eliminates the need for highly toxic osmium tetroxide and reduces the total number of steps, leading to higher overall efficiency and lower production costs.
Q: What are the critical safety advantages of this new process for industrial scale-up?
A: The process avoids the use of osmium tetroxide, a substance with high toxicity and stringent safety requirements. Additionally, the reaction conditions are relatively mild, with key steps occurring between -40°C and 60°C, making thermal management and operational safety much easier to control in a commercial plant.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for industrial applicability. It utilizes commercially available starting materials and standard reagents like cuprous iodide and sodium borohydride. The robust yields reported in the patent examples (e.g., up to 85% in cyclization steps) indicate strong potential for scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prostaglandin Intermediate Supplier
The development of Formula I marks a significant milestone in the evolution of prostaglandin synthesis, offering a pathway that is both economically and environmentally sustainable. As the industry moves towards more efficient manufacturing paradigms, the ability to execute such complex organic transformations with high precision becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including low-temperature reactors and advanced purification systems. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that the downstream synthesis of Beraprost sodium proceeds without interruption or yield loss.
We invite potential partners to explore how this innovative intermediate can transform their supply chain dynamics. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of switching to this superior synthetic route. Contact us today to discuss how we can support your long-term goals for cost-effective and reliable API manufacturing.
