Advanced Synthesis of Allyl Phosphorus Compounds Using Metal-Free Borane Catalysis for Commercial Scale-Up
Advanced Synthesis of Allyl Phosphorus Compounds Using Metal-Free Borane Catalysis for Commercial Scale-Up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic methodologies in the fine chemical industry. Patent CN111943980B introduces a groundbreaking approach to synthesizing allyl phosphorus compounds, a critical class of molecules serving as versatile intermediates in the production of bioactive pharmaceuticals and agrochemicals. This technology leverages the potent Lewis acidity of tris(pentafluorophenyl)borane, B(C6F5)3, to catalyze the direct dehydrative coupling of secondary allyl alcohols with phosphorus-hydrogen compounds. Unlike traditional methods that rely on harsh conditions or precious metal catalysts, this innovation operates under mild thermal conditions ranging from 80°C to 100°C, achieving exceptional conversion rates while generating water as the only byproduct. For R&D directors and process chemists, this represents a paradigm shift towards atom-economical synthesis that aligns perfectly with modern sustainability goals and regulatory pressures regarding heavy metal residues in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon-phosphorus bond in allylic systems has been fraught with significant technical and economic challenges that hinder large-scale manufacturing efficiency. Conventional synthetic routes typically depend on transition metal-catalyzed cross-coupling reactions, such as those utilizing palladium or nickel complexes, which necessitate the use of expensive ligands and strictly anhydrous conditions to prevent catalyst deactivation. Furthermore, alternative approaches involving the addition of phosphorus-hydrogen compounds to unsaturated bonds often require strong bases or stoichiometric oxidants, leading to poor atom economy and the generation of substantial quantities of hazardous chemical waste. These legacy processes not only inflate the cost of goods sold due to the high price of catalysts and reagents but also complicate downstream purification, as removing trace amounts of toxic transition metals from the final product requires additional, costly processing steps that can erode overall yield and profitability.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the novel methodology disclosed in the patent utilizes a metal-free Lewis acid catalytic system that streamlines the synthesis into a single, efficient operational step. By employing readily available secondary allyl alcohols and phosphine compounds in the presence of a catalytic amount of B(C6F5)3, the reaction proceeds smoothly to form the desired allyl phosphorus backbone with high selectivity. This approach effectively bypasses the need for pre-functionalized substrates or hazardous activating agents, thereby simplifying the supply chain logistics and reducing the inventory of dangerous chemicals required on-site. The robustness of this catalytic system allows for a broad substrate scope, accommodating various aryl, heterocyclic, and alkyl substituents without compromising reaction efficiency, making it an ideal candidate for the diversified production needs of a reliable allyl phosphorus compound supplier serving the global pharmaceutical market.
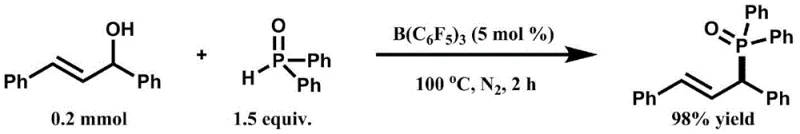
Mechanistic Insights into B(C6F5)3-Catalyzed Dehydrative Coupling
The mechanistic elegance of this transformation lies in the ability of the tris(pentafluorophenyl)borane catalyst to activate the hydroxyl group of the secondary allyl alcohol towards nucleophilic substitution. As a strong Lewis acid, the boron center coordinates with the oxygen atom of the alcohol, significantly increasing the electrophilicity of the adjacent carbon atom and facilitating the departure of the hydroxyl group as water. This activation generates a reactive allylic cation intermediate or a tight ion pair, which is then rapidly intercepted by the nucleophilic phosphorus species, such as diphenylphosphine oxide or dialkyl phosphites. This pathway avoids the formation of radical intermediates often seen in other phosphonylation reactions, thereby ensuring high stereochemical control and minimizing the formation of unwanted regioisomers that could complicate the impurity profile of the final drug substance.
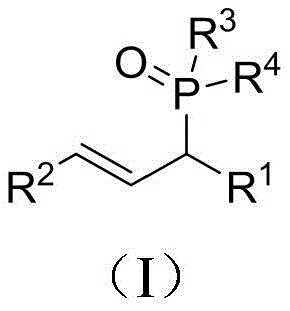
From an impurity control perspective, this mechanism offers distinct advantages for manufacturing high-purity intermediates required for clinical applications. Since the reaction does not involve transition metals, the risk of metal-catalyzed side reactions, such as homocoupling of the phosphine or isomerization of the double bond, is drastically reduced. The general structural formula of the resulting compounds, as depicted in the patent, showcases the versatility of the R groups, which can be tuned to introduce specific electronic or steric properties required for downstream biological activity. The clean reaction profile, characterized by the exclusive formation of water as a byproduct, means that the workup procedure is straightforward, typically requiring only simple extraction and chromatography to achieve purity levels that meet stringent quality specifications for pharmaceutical intermediates.
How to Synthesize Allyl Phosphorus Compounds Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for process development teams aiming to transfer laboratory protocols to pilot and commercial plants. The standard procedure involves charging a reaction vessel with the secondary allyl alcohol and the phosphorus-hydrogen compound under an inert atmosphere, followed by the addition of the borane catalyst. The mixture is then heated to the optimal temperature range, typically around 100°C, and maintained for a duration of 2 to 12 hours depending on the specific reactivity of the substrates involved. Detailed standardized synthetic steps, including precise molar ratios and purification parameters, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Mix secondary allyl alcohol, phosphine compound, and tris(pentafluorophenyl)borane catalyst in a molar ratio of approximately 1: 1.5:0.05 under an inert atmosphere.
- Heat the reaction mixture to a temperature between 80°C and 100°C and stir for 2 to 12 hours to facilitate dehydrative coupling.
- Purify the resulting crude reaction product using thin layer chromatography or column chromatography to isolate the high-purity allyl phosphorus compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this borane-catalyzed technology translates into tangible strategic benefits that directly impact the bottom line and operational resilience. The elimination of expensive transition metal catalysts and specialized ligands results in a significant reduction in raw material costs, allowing for more competitive pricing structures in the supply of complex organic intermediates. Moreover, the use of commercially available and stable starting materials, such as simple allyl alcohols and phosphine oxides, mitigates supply chain risks associated with sourcing exotic or proprietary reagents that may be subject to geopolitical constraints or volatile market pricing. This stability ensures a continuous and reliable flow of materials, which is critical for maintaining uninterrupted production schedules in the fast-paced pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the drastic simplification of the reaction workflow, which removes the need for costly metal scavenging steps and extensive waste treatment protocols associated with heavy metal usage. By operating under solvent-free conditions or using minimal amounts of benign solvents like xylene, the process further reduces expenditure on solvent purchase, recovery, and disposal, contributing to substantial overall cost savings in pharmaceutical intermediates manufacturing. Additionally, the high atom economy of the dehydrative coupling ensures that the majority of the mass of the starting materials is incorporated into the final product, maximizing yield and minimizing the cost per kilogram of the active intermediate produced.
- Enhanced Supply Chain Reliability: The reliance on robust, off-the-shelf chemicals rather than sensitive organometallic reagents significantly shortens the lead time for high-purity allyl phosphorus compounds, as suppliers can maintain larger inventories of stable precursors without fear of degradation. This flexibility allows manufacturers to respond rapidly to fluctuating demand signals from downstream clients, ensuring that critical project timelines are met without delay. Furthermore, the simplified logistics of handling non-hazardous catalysts compared to pyrophoric metal complexes reduces transportation costs and regulatory burdens, streamlining the entire procurement cycle from vendor selection to final delivery at the manufacturing site.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the generation of water as the sole byproduct positions this technology as a leader in green chemistry, facilitating easier compliance with increasingly strict environmental regulations regarding industrial effluent. The absence of halogenated waste streams and heavy metal contamination simplifies the permitting process for new production lines and reduces the long-term liability associated with waste management. This eco-friendly profile not only enhances the corporate social responsibility standing of the manufacturing entity but also future-proofs the supply chain against potential regulatory bans on certain chemical processes, ensuring long-term commercial viability and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method, providing clarity for partners evaluating its potential for their specific projects. These insights are derived directly from the experimental data and embodiments described in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for assessing the feasibility of integrating this technology into existing production workflows or new product development pipelines.
Q: What are the advantages of using B(C6F5)3 over transition metal catalysts for this synthesis?
A: The use of tris(pentafluorophenyl)borane eliminates the need for expensive transition metals like palladium and complex ligands, significantly reducing raw material costs and simplifying the removal of toxic metal residues from the final pharmaceutical intermediate.
Q: What is the primary byproduct of this dehydrative coupling reaction?
A: The reaction produces water as the sole byproduct, which offers significant environmental benefits by avoiding the generation of stoichiometric salt waste or halogenated organic waste common in traditional cross-coupling methods.
Q: Can this method accommodate diverse substrate structures?
A: Yes, the protocol demonstrates broad substrate scope, successfully converting various secondary allyl alcohols containing aryl, heterocyclic, and alkyl groups, as well as different phosphine oxides and phosphites, into the corresponding allyl phosphorus derivatives with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Phosphorus Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free catalytic technology in advancing the synthesis of complex organophosphorus architectures. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of allyl phosphorus compound delivered meets the highest standards of quality required for pharmaceutical and agrochemical applications. We are committed to leveraging our technical expertise to optimize this green synthesis route, delivering cost-effective solutions that empower our clients to bring life-saving therapies to market faster.
We invite you to engage with our technical procurement team to discuss how this innovative methodology can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project volume and timeline. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted allyl phosphorus compound supplier dedicated to driving value through chemical innovation and operational excellence.
