Advanced Catalytic Synthesis of Allyl Phosphorus Compounds for Commercial Pharmaceutical Intermediates
Advanced Catalytic Synthesis of Allyl Phosphorus Compounds for Commercial Pharmaceutical Intermediates
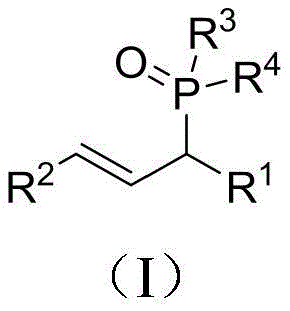
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic methodologies in the fine chemical industry. Patent CN111943980B introduces a groundbreaking approach to synthesizing allyl phosphorus compounds, utilizing a Lewis acid catalytic system that bypasses the traditional reliance on expensive transition metals. This technology represents a pivotal shift for manufacturers of pharmaceutical intermediates and agrochemical precursors, offering a pathway to high-purity products with exceptional atom economy. By leveraging tris(pentafluorophenyl)borane as a robust catalyst, the process achieves efficient dehydrative coupling between secondary allylic alcohols and phosphorus-hydrogen compounds under remarkably mild conditions. For R&D directors and procurement strategists alike, this innovation addresses critical pain points regarding catalyst residue, operational complexity, and environmental compliance, positioning it as a superior alternative for the commercial scale-up of complex organophosphorus structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-phosphorus bonds in allylic systems has been dominated by transition metal-catalyzed cross-coupling reactions, which, while effective, impose severe economic and logistical burdens on large-scale manufacturing. Traditional methods often necessitate the use of precious metal catalysts such as palladium or nickel, accompanied by sophisticated and costly ligand systems to ensure regioselectivity and yield. These processes typically require harsh reaction conditions, including strong bases or quantitative oxidants, which not only increase energy consumption but also generate substantial amounts of hazardous waste streams that require expensive disposal protocols. Furthermore, the presence of transition metals in the final product poses a significant regulatory hurdle for pharmaceutical applications, necessitating rigorous and often yield-reducing purification steps to meet stringent heavy metal limits. The reliance on pre-functionalized substrates, such as allylic halides, further exacerbates the cost profile by adding synthetic steps and reducing overall atom economy, making these conventional routes increasingly untenable for cost-sensitive bulk production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct dehydrative coupling strategy that fundamentally simplifies the synthetic architecture while enhancing sustainability metrics. By employing tris(pentafluorophenyl)borane as a potent Lewis acid catalyst, the process activates the hydroxyl group of readily available secondary allylic alcohols, facilitating nucleophilic attack by phosphorus-hydrogen compounds without the need for pre-activation or halogenation. This metal-free approach operates efficiently at temperatures between 80°C and 100°C, significantly lowering the thermal energy input compared to many high-temperature coupling reactions. The reaction is remarkably clean, producing water as the sole byproduct, which aligns perfectly with green chemistry principles and drastically reduces the environmental footprint of the manufacturing process. Additionally, the broad substrate scope allows for the use of diverse aryl, heterocyclic, and alkyl-substituted allylic alcohols, providing medicinal chemists with a versatile toolbox for generating structural diversity in lead optimization campaigns without compromising on process efficiency.
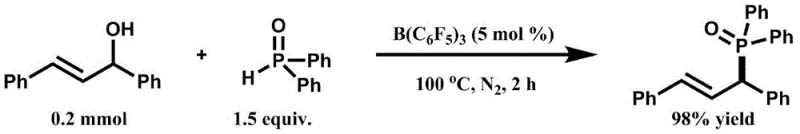
Mechanistic Insights into B(C6F5)3-Catalyzed Dehydrative Coupling
The efficacy of this novel synthetic route lies in the unique ability of the boron catalyst to coordinate with the oxygen atom of the allylic alcohol, thereby increasing the electrophilicity of the allylic carbon center through the formation of a transient cationic species. This activation mechanism lowers the energy barrier for the nucleophilic substitution by the phosphorus-hydrogen bond, allowing the reaction to proceed smoothly under neutral to mildly acidic conditions without the need for external bases. The catalytic cycle is highly efficient, requiring only 5 mol% of the boron species to drive the conversion to completion, which speaks to the high turnover number and stability of the catalyst under the reaction conditions. From a mechanistic standpoint, the preservation of the double bond geometry and the high regioselectivity observed suggest a tightly controlled transition state that minimizes side reactions such as polymerization or isomerization, which are common pitfalls in allylic substitutions. This precise control over the reaction trajectory is crucial for maintaining the stereochemical integrity of chiral centers adjacent to the phosphorus moiety, a feature that is often critical for the biological activity of the resulting pharmaceutical intermediates.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the absence of metal-mediated side reactions and the specificity of the Lewis acid activation. In traditional transition metal catalysis, issues such as beta-hydride elimination or homocoupling of the phosphorus reagent can lead to complex mixtures that are difficult to separate, often requiring multiple chromatographic passes that erode yield. In this boron-catalyzed system, the primary impurities are typically unreacted starting materials or simple dehydration products of the alcohol, both of which are easily removed during standard workup procedures. The lack of metal residues also means that the downstream purification burden is significantly reduced, allowing for simpler crystallization or extraction protocols that are more amenable to continuous flow processing. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the risk of unexpected impurity formation is minimized, thereby ensuring a consistent and reliable supply of high-purity material for subsequent synthetic steps.
How to Synthesize Allyl Phosphorus Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol that combines the allylic alcohol and the phosphorus-hydrogen compound in the presence of the boron catalyst under an inert atmosphere. The reaction can be conducted either neat or in common organic solvents such as xylene or acetonitrile, offering flexibility depending on the solubility of the specific substrates involved. Following the reaction period of 2 to 12 hours, the crude mixture is subjected to standard purification techniques, typically yielding the desired product in high purity and excellent yields ranging from 86% to 98% as demonstrated in the patent examples. For detailed operational parameters, safety considerations, and specific stoichiometric ratios tailored to your specific substrate, please refer to the standardized synthesis guide provided below.
- Mix allyl alcohol, phosphorus hydrogen compound, and tris(pentafluorophenyl)borane catalyst in a molar ratio of roughly 1: 1.5:0.05 in a reaction vessel under inert gas.
- Heat the mixture to 80-100°C and stir for 2 to 12 hours to facilitate the dehydrative coupling reaction.
- Purify the resulting reaction product using thin layer chromatography or standard column chromatography to isolate the high-purity allyl phosphorus compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this boron-catalyzed technology offers compelling economic and logistical benefits that directly impact the bottom line of chemical manufacturing operations. The elimination of precious metal catalysts removes a major cost driver from the bill of materials, while simultaneously mitigating the supply chain risks associated with the volatility of palladium and rhodium markets. Moreover, the simplified purification process reduces the consumption of silica gel and solvents, leading to significant savings in waste disposal costs and improving the overall throughput of the production facility. The ability to run the reaction solvent-free or in inexpensive solvents further enhances the cost-efficiency, making this route highly competitive for the production of bulk pharmaceutical intermediates where margin pressure is intense.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts and specialized ligands, which traditionally account for a substantial portion of the raw material costs in organophosphorus synthesis. By replacing these with a catalytic amount of a commercially available boron Lewis acid, the direct material cost is drastically lowered, and the need for expensive metal scavenging resins during purification is entirely eliminated. This streamlining of the process not only reduces the cost of goods sold but also shortens the production cycle time by removing complex metal removal steps, thereby increasing the overall capacity utilization of the manufacturing plant without requiring additional capital investment.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade starting materials such as secondary allylic alcohols and simple phosphine oxides ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike specialized organometallic reagents that may have long lead times or single-source suppliers, the inputs for this process are widely sourced from multiple global vendors, providing procurement teams with greater negotiating power and security of supply. Additionally, the mild reaction conditions reduce the wear and tear on reactor equipment and lower the energy requirements for heating and cooling, contributing to more predictable and stable production schedules that can reliably meet customer demand fluctuations.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, specifically the generation of water as the only byproduct, significantly simplify environmental compliance and permitting for new production lines. The absence of heavy metal waste streams alleviates the regulatory burden associated with hazardous waste disposal, allowing for faster approval of manufacturing changes and expansions. This environmental friendliness, combined with the proven scalability of the reaction from milligram to multi-gram scales in the patent data, positions this technology as an ideal candidate for rapid commercial scale-up, enabling companies to bring new phosphorus-containing drugs to market faster while adhering to increasingly strict global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this boron-catalyzed route for your specific production needs. We encourage technical teams to review these details to fully appreciate the transformative potential of this methodology in modern organic synthesis.
Q: What are the advantages of using B(C6F5)3 over transition metal catalysts for this synthesis?
A: The use of tris(pentafluorophenyl)borane eliminates the need for expensive transition metals like palladium and complex ligands, significantly reducing raw material costs and simplifying the removal of heavy metal residues from the final API intermediate.
Q: Does this process generate hazardous waste?
A: No, the process is environmentally friendly as the only byproduct generated during the dehydrative coupling is water, avoiding the production of toxic halogenated salts or heavy metal waste streams common in traditional cross-coupling methods.
Q: Can this method be scaled for industrial production?
A: Yes, the reaction conditions are mild (80-100°C) and can even be performed solvent-free, which greatly enhances the feasibility of scaling up to multi-kilogram or tonnage production while maintaining high atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Phosphorus Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this boron-catalyzed synthesis in delivering high-value organophosphorus intermediates to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of allyl phosphorus compound delivered meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging this innovative technology to provide our clients with a competitive edge through superior quality and consistent supply.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume and timeline. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process, ensuring that your partnership with us is built on a foundation of transparency, technical excellence, and shared success in bringing life-saving medicines to patients worldwide.
