Advanced Salt Formation Technology for Scalable Trabectedin Intermediate Manufacturing
Introduction to Advanced Trabectedin Synthesis
The pharmaceutical landscape for oncology treatments continues to evolve, with Trabectedin (ET-743) standing out as a potent marine-derived antineoplastic agent approved for treating advanced soft tissue sarcomas. However, the commercial viability of such complex molecules often hinges on the efficiency of their synthetic routes. Patent CN108101934B introduces a transformative approach to synthesizing Trabectedin by addressing a critical bottleneck: the purification of its key intermediate, Corey 3. Traditionally, this intermediate exists as an oily substance, necessitating cumbersome column chromatography that hinders industrial scalability. This patent discloses a novel compound salt of formula I-1, which converts this problematic oil into a stable, crystalline solid, thereby streamlining the manufacturing process for reliable pharmaceutical intermediate supplier operations.
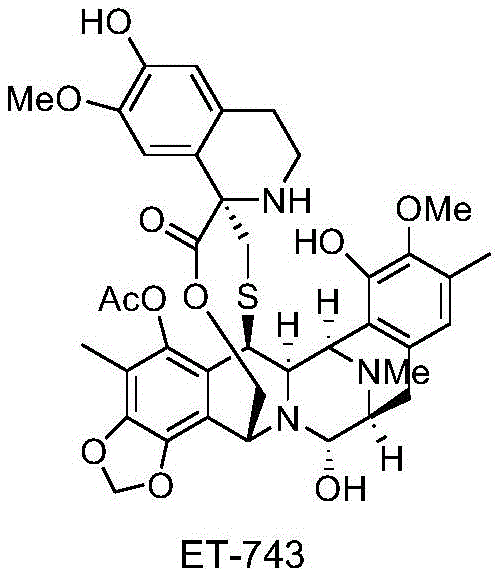
The significance of this innovation lies in its ability to enhance the physicochemical properties of the intermediate, making it amenable to standard industrial purification techniques like crystallization and filtration. By shifting from an oil to a salt, the process not only improves purity profiles but also facilitates safer packaging and transfer, crucial factors for maintaining supply chain continuity in the production of high-value anticancer drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
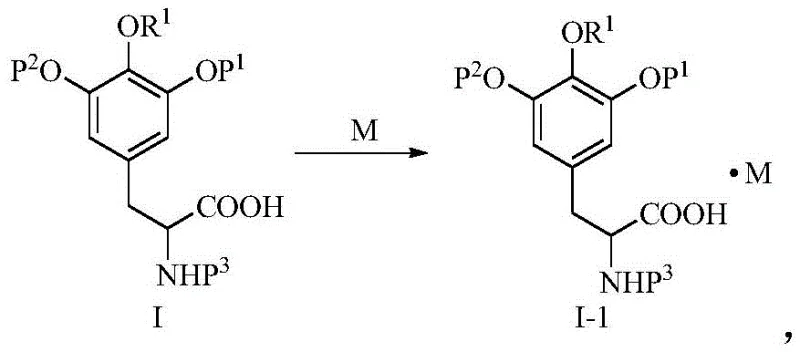
In the conventional synthesis of Trabectedin, the intermediate known as Corey 3, or (S)-3-[4-methoxy-3,5-bis(tert-butyldimethylsilyloxy)]phenyl-2-(allyloxyacylamino)propionic acid, presents a significant processing challenge. As described in prior art such as WO2001058905, this compound is inherently an oily substance at room temperature. In a manufacturing context, oils are notoriously difficult to handle; they do not flow like powders, they trap solvents, and most critically, they resist simple purification. To achieve the requisite purity for downstream coupling, manufacturers are forced to rely on column chromatography. This technique is not only solvent-intensive and expensive but also difficult to scale beyond laboratory quantities, creating a severe bottleneck for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by converting the free acid form of Corey 3 into a salt form, designated as formula I-1. By reacting the acid with specific organic bases, the physical state of the molecule changes from an amorphous oil to a defined crystalline solid. This phase change is pivotal because it allows the use of recrystallization—a far more scalable and cost-effective purification method than chromatography. The result is an intermediate that can be isolated via simple filtration, washed to remove impurities, and dried to high purity standards, effectively solving the scalability issue inherent in the traditional route.
Mechanistic Insights into Salt Formation and Purification
The core of this technology relies on the precise selection of a base molecule (M) that interacts with the carboxylic acid group of the Corey 3 intermediate to form a stable lattice structure. The patent specifies that the base can be inorganic or organic, but organic bases such as L-phenylglycinamide and diisopropylamine are particularly effective. The mechanism involves the deprotonation of the carboxylic acid by the amine base, resulting in an ammonium carboxylate salt. The steric and electronic properties of the chosen base are critical; for instance, screening data reveals that while triethylamine and dicyclohexylamine fail to precipitate solids, L-phenylglycinamide successfully induces crystallization. This suggests that the specific hydrogen bonding network and molecular packing facilitated by the phenylglycinamide moiety are essential for stabilizing the crystal lattice.
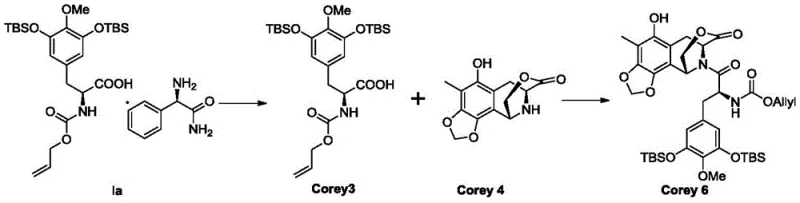
Furthermore, the preservation of stereochemistry is paramount in the synthesis of chiral drugs like Trabectedin. The salt formation process described does not involve harsh conditions that might racemize the chiral center at the alpha-position of the amino acid backbone. Instead, it operates under mild thermal conditions (e.g., stirring at 50°C), ensuring that the optical purity of the intermediate is maintained or even enhanced through the selective crystallization of the desired enantiomeric salt. This mechanistic robustness ensures that the downstream coupling with the complex tetrahydroisoquinoline fragment (Corey 4) proceeds with high fidelity, minimizing the formation of diastereomeric impurities that are difficult to separate in later stages.
How to Synthesize Trabectedin Intermediate Salt Efficiently
The synthesis of the high-purity salt form of the Trabectedin intermediate represents a significant optimization in process chemistry. By utilizing specific organic bases and solvent systems, manufacturers can transition from laborious chromatographic purification to efficient crystallization protocols. This section outlines the general operational framework derived from the patent examples, demonstrating how to achieve yields exceeding 90% with purity levels approaching 99%. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation provided below.
- Dissolve the crude Corey 3 intermediate (S)-3-[4-methoxy-3,5-bis(tert-butyldimethylsilyloxy)]phenyl-2-(allyloxyacylamino)propionic acid in a suitable organic solvent such as methyl tert-butyl ether (MTBE) or acetonitrile.
- Add a stoichiometric amount of an organic base, specifically L-phenylglycinamide or diisopropylamine, to the solution under stirring conditions at elevated temperatures (e.g., 50°C).
- Allow the mixture to cool or concentrate under reduced pressure to induce crystallization, then filter and dry the resulting solid salt to achieve high purity (>98%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from an oily intermediate to a crystalline salt offers profound logistical and economic benefits. The primary advantage is the elimination of column chromatography, which is a major cost driver in fine chemical manufacturing due to high solvent consumption and silica gel usage. By replacing this with crystallization, the process significantly reduces raw material costs and waste generation, aligning with green chemistry principles. Additionally, solid intermediates are far easier to package, store, and transport than oils, reducing the risk of leakage and degradation during logistics, thus enhancing supply chain reliability for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to a salt-based purification strategy eliminates the need for expensive chromatographic columns and the vast quantities of solvents required for elution. This structural change in the process flow leads to substantial cost savings by reducing both material expenses and the energy costs associated with solvent recovery. Furthermore, the higher throughput achievable with filtration compared to chromatography allows for better utilization of reactor capacity, driving down the unit cost of production without compromising quality.
- Enhanced Supply Chain Reliability: Solid intermediates possess superior stability profiles compared to their oily counterparts, which can be prone to oxidation or hydrolysis over time. The crystalline nature of the I-1 salt ensures a longer shelf life and consistent quality upon arrival at the customer's facility. This reliability is critical for just-in-time manufacturing schedules, as it minimizes the risk of batch failures due to degraded starting materials, thereby securing the continuity of the entire Trabectedin production line.
- Scalability and Environmental Compliance: Crystallization is a unit operation that scales linearly from grams to tons, whereas chromatography often faces non-linear challenges at large scales. This inherent scalability means that the process can be easily adapted for commercial scale-up of complex pharmaceutical intermediates to meet market demand. Moreover, the reduction in solvent waste simplifies environmental compliance and waste treatment protocols, making the manufacturing process more sustainable and less burdensome on facility infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this salt formation technology. These answers are derived directly from the experimental data and embodiments within the patent, providing clarity on solvent selection, base compatibility, and downstream processing. Understanding these nuances is essential for R&D teams looking to integrate this methodology into their existing workflows.
Q: Why is the salt form of Corey 3 preferred over the free acid for industrial production?
A: The free acid form of Corey 3 is an oily substance that requires complex and costly column chromatography for purification. Converting it into a salt form (such as with L-phenylglycinamide or diisopropylamine) transforms it into a crystalline solid, allowing for simple filtration and recrystallization, which drastically improves purity and operational efficiency.
Q: What are the optimal bases for forming the Trabectedin intermediate salt?
A: According to the patent data, organic bases such as L-phenylglycinamide and diisopropylamine are highly effective. Screening data indicates that while bases like dicyclohexylamine and triethylamine fail to form stable solids, L-phenylglycinamide yields salts with up to 99% purity and excellent recovery rates.
Q: How does this process impact the downstream synthesis of Trabectedin (ET-743)?
A: By providing a high-purity, solid intermediate, this process ensures consistent quality for the subsequent coupling reactions with Corey 4. This reduces the risk of impurities carrying through to the final API, simplifying the overall workflow and enhancing the reliability of the supply chain for this potent antineoplastic agent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trabectedin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate synthesis in the production of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Trabectedin intermediate meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this advanced salt formation technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can drive efficiency and reliability in your Trabectedin manufacturing operations.
