Advanced Salt Formation Strategy for High-Purity Trabectedin Intermediates and Commercial Scalability
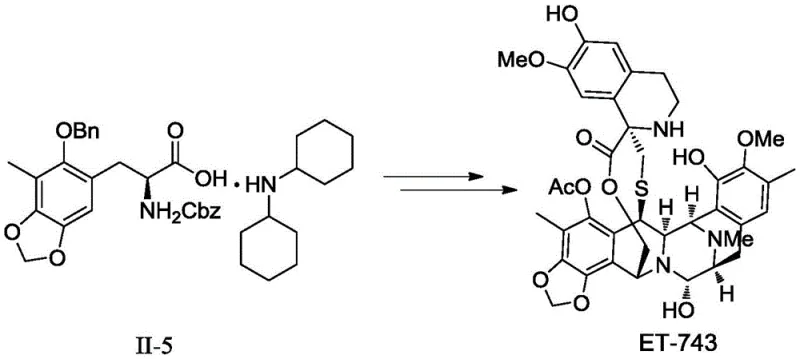
The development of efficient synthetic routes for complex marine-derived antineoplastic agents remains a critical challenge in modern pharmaceutical manufacturing. Patent CN108101881B addresses a significant bottleneck in the production of Trabectedin (ET-743), a potent anticancer drug approved for treating advanced soft tissue sarcomas. The core innovation lies in the preparation of a specific salt form of the key intermediate, (S)-3-(3-methyl-2-benzyloxy-4,5-methylenedioxy)phenyl-2-(benzyloxyacylamino)propionic acid. Historically, the free acid form of this intermediate (designated as II-5a) presents severe processing difficulties due to its physical state as an oil, which necessitates cumbersome purification methods like column chromatography. This patent introduces a transformative approach by converting this oily intermediate into a stable, crystalline salt (specifically the dicyclohexylamine salt, II-5), thereby enabling purification through standard recrystallization techniques. This shift from chromatographic purification to crystallization is not merely a procedural tweak but a fundamental upgrade that aligns the synthesis with Good Manufacturing Practice (GMP) standards required for commercial API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in CN104557850A, rely on the isolation of the intermediate compound II-5a in its free acid form. The fundamental flaw in this conventional approach is the physicochemical nature of II-5a, which exists as a viscous oil at room temperature. In an industrial setting, handling oily intermediates is notoriously difficult; they do not flow easily, are prone to trapping solvent impurities, and cannot be purified by the gold-standard method of recrystallization. Consequently, manufacturers are forced to rely on column chromatography to achieve the necessary purity levels. Column chromatography is inherently batch-limited, consumes vast quantities of silica gel and organic solvents, and is extremely difficult to scale up beyond kilogram quantities without exponential increases in cost and operational complexity. Furthermore, the lack of a defined crystal lattice in the oily product leads to issues with long-term stability and reproducibility, posing significant risks for supply chain consistency in the production of high-value oncology drugs.
The Novel Approach
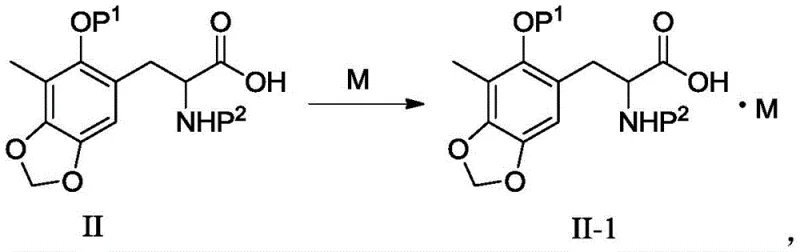
The methodology presented in Patent CN108101881B circumvents these limitations by introducing a salt formation step that fundamentally alters the physical properties of the intermediate. By reacting the carboxylic acid group of compound II with a suitable organic base, specifically dicyclohexylamine, the process generates the ammonium carboxylate salt designated as Formula II-5. Unlike the parent acid, this salt exhibits excellent crystallinity. This physical transformation allows manufacturers to replace the inefficient column chromatography step with a simple, scalable recrystallization process using common solvents like acetonitrile or ethanol. The result is a dramatic improvement in operational simplicity and product quality. The patent data indicates that this method can achieve HPLC purities exceeding 99% with high recovery yields (up to 92% in optimized examples). This approach not only simplifies the workflow but also ensures that the intermediate is in a stable, solid form that is ideal for packaging, long-term storage, and global transportation to downstream synthesis sites.
Mechanistic Insights into Acid-Base Salt Formation and Crystallization
The core chemical mechanism driving this innovation is a classic acid-base neutralization reaction, yet its application here is highly specific to the steric and electronic environment of the trabectedin scaffold. The intermediate possesses a carboxylic acid functionality adjacent to a chiral center protected by a benzyloxycarbonyl (Cbz) group. When treated with a secondary amine base like dicyclohexylamine, the proton from the carboxylic acid is transferred to the nitrogen of the amine, forming an ionic bond between the carboxylate anion and the ammonium cation. The choice of dicyclohexylamine is critical; its bulky cyclohexyl rings provide the necessary steric bulk and symmetry to facilitate tight packing in the crystal lattice, which is essential for precipitation from solution. Smaller or less symmetric bases, such as triethylamine, failed to induce crystallization in the screening experiments described in the patent, highlighting the importance of molecular geometry in solid-state engineering. This specific ion-pair interaction lowers the solubility of the species in the chosen solvent system (e.g., acetonitrile), driving the equilibrium towards the solid phase upon concentration.
From an impurity control perspective, this crystallization mechanism acts as a powerful purification driver. As the salt crystals nucleate and grow, the regular lattice structure selectively incorporates the target molecule while excluding structurally related impurities and residual solvents into the mother liquor. This phenomenon, known as lattice rejection, is far more effective than the adsorption-based separation of chromatography. The patent demonstrates that even when starting with crude materials, a single crystallization step can elevate purity to >99%. Furthermore, the salt form protects the sensitive functionalities of the molecule during storage. The ionic nature of the salt reduces the likelihood of hydrolysis or racemization that might occur in the free acid form, ensuring that the stereochemical integrity of the chiral center—critical for the biological activity of the final trabectedin drug—is maintained throughout the supply chain.
How to Synthesize Trabectedin Intermediate Salt Efficiently
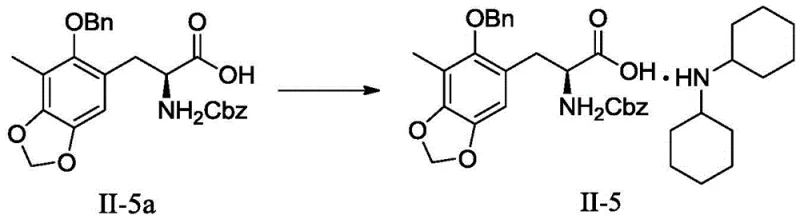
The synthesis of the high-purity intermediate salt II-5 is a straightforward yet precise operation that leverages the solubility differences between the reactants and the product. The process begins by dissolving the precursor oil (II-5a) in a polar aprotic solvent such as acetonitrile, which effectively solvates both the organic acid and the amine base. Upon the addition of dicyclohexylamine, typically in a slight molar excess (1.0 to 2.0 equivalents), the reaction mixture is heated to facilitate complete salt formation. The subsequent removal of solvent under reduced pressure is the critical trigger for crystallization; by concentrating the solution, the solubility limit of the salt is exceeded, prompting nucleation. The detailed standardized synthesis steps, including specific temperature profiles, stirring rates, and filtration protocols required to maximize yield and crystal habit, are outlined in the guide below.
- Dissolve the crude oily intermediate (S)-3-(3-methyl-2-benzyloxy-4,5-methylenedioxy)phenyl-2-(benzyloxyacylamino)propionic acid (II-5a) in a suitable organic solvent such as acetonitrile.
- Add the organic base, specifically dicyclohexylamine, to the solution under stirring conditions at elevated temperatures (e.g., 50°C) to initiate the salt formation reaction.
- Remove a portion of the solvent under reduced pressure to induce supersaturation, followed by filtration to isolate the high-purity crystalline salt product (II-5).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from an oily intermediate to a crystalline salt represents a substantial strategic advantage in terms of cost structure and risk mitigation. The elimination of column chromatography removes one of the most expensive and variable unit operations in fine chemical manufacturing. Chromatography requires significant capital investment in columns, massive volumes of high-purity solvents, and extensive labor for fraction collection and analysis. By replacing this with a crystallization-filtration-drying sequence, the manufacturing process becomes significantly more lean and cost-effective. This reduction in processing complexity directly translates to a lower cost of goods sold (COGS), allowing for more competitive pricing of the final API without compromising on quality margins. Additionally, the robustness of the crystallization process reduces batch-to-batch variability, minimizing the risk of costly reworks or batch rejections.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the complete avoidance of preparative chromatography. In traditional processes for trabectedin intermediates, silica gel consumption and solvent disposal costs can be prohibitive at scale. The new salt formation route utilizes common, recoverable solvents like acetonitrile and ethanol, which are cheaper and easier to recycle than the mixed solvent systems often required for chromatography. Furthermore, the high yield reported in the patent examples (up to 92%) ensures that raw material costs are optimized, as less starting material is lost to purification inefficiencies. This streamlined workflow reduces the overall processing time and utility consumption, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the stability and transportability of intermediates. Oily substances like the free acid II-5a are prone to degradation, leakage, and difficulty in handling during international shipping. In contrast, the crystalline salt II-5 is a stable solid that can be packaged in standard drums or bags, significantly reducing logistics risks and costs. The improved stability also extends the shelf-life of the intermediate, allowing manufacturers to produce in larger batches and hold inventory strategically to buffer against demand fluctuations. This reliability ensures a continuous supply of high-quality material to the final API synthesis sites, preventing production stoppages that could delay patient access to this critical cancer therapy.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the new process offers a greener alternative to the status quo. Chromatography generates large volumes of hazardous waste containing silica and mixed organic solvents, which require specialized treatment and disposal. The crystallization process generates significantly less waste, primarily consisting of mother liquor that can often be distilled and reused. This reduction in waste volume simplifies compliance with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste disposal. Moreover, the scalability of crystallization is well-understood in the chemical industry; moving from a 1-liter lab flask to a 1000-liter reactor involves predictable engineering parameters, whereas scaling chromatography is non-linear and fraught with technical challenges. This makes the technology ready for immediate commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this salt formation technology. These answers are derived directly from the experimental data and claims within Patent CN108101881B, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for technical teams assessing the integration of this intermediate into their existing trabectedin supply chains.
Q: Why is the salt form of the trabectedin intermediate preferred over the free acid form?
A: The free acid form (II-5a) exists as an oily substance that is difficult to purify without complex column chromatography, making it unsuitable for large-scale industrial production. Converting it into a salt form (such as the dicyclohexylamine salt II-5) transforms the physical state into a crystalline solid, allowing for purification via simple recrystallization and significantly improving stability for storage and transport.
Q: What represents the most effective base for this salt formation process?
A: According to the experimental data in Patent CN108101881B, dicyclohexylamine demonstrates superior salt-forming efficiency compared to other bases like triethylamine or aniline derivatives. It yields a product with high HPLC purity (>99%) and excellent recovery rates, whereas other bases either failed to form solids or resulted in lower purity profiles.
Q: How does this process impact the overall synthesis of Trabectedin (ET-743)?
A: By establishing a robust method to produce high-purity intermediate II-5, this process ensures a consistent and reliable feedstock for the subsequent coupling reactions required to build the complex trabectedin molecule. It eliminates the bottleneck of purifying oily intermediates, thereby streamlining the supply chain for this potent antineoplastic agent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trabectedin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs like Trabectedin depends on the reliability and purity of every building block in the synthesis chain. Our technical team has thoroughly analyzed the salt formation technology described in CN108101881B and is fully equipped to implement this advanced crystallization process at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are designed to handle sensitive intermediates with stringent purity specifications, utilizing rigorous QC labs to verify that every batch of the dicyclohexylamine salt meets the >99% purity benchmark required for downstream coupling reactions.
We invite pharmaceutical partners to leverage our expertise to optimize their trabectedin supply chains. By adopting this crystalline intermediate strategy, you can significantly de-risk your manufacturing process and achieve better cost efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our implementation of this patent technology can accelerate your project timelines and enhance your competitive position in the global market.
