Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic methodologies to access privileged scaffolds that exhibit potent biological activities, and quinazolinones stand out as a cornerstone class of heterocycles found in numerous therapeutic agents ranging from antifungals to anticancer drugs. Patent CN112480015B introduces a transformative approach to constructing these valuable cores by specifically targeting the incorporation of the trifluoromethyl group, a modification known to drastically enhance metabolic stability and lipophilicity in drug candidates. This innovation addresses the longstanding challenges associated with introducing fluorine motifs into complex heterocyclic systems, offering a streamlined pathway that bypasses the need for hazardous high-pressure carbon monoxide gas or expensive pre-activated substrates. By leveraging a palladium-catalyzed multicomponent cascade, this technology enables the direct assembly of 2-trifluoromethyl substituted quinazolinones from simple nitro compounds and trifluoroethylimidoyl chlorides. For research and development teams, this represents a significant leap forward in process chemistry, providing a versatile platform for generating diverse libraries of bioactive molecules with improved physicochemical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has relied heavily on methodologies that impose severe constraints on operational safety and economic feasibility, often requiring the use of toxic carbon monoxide gas under high-pressure conditions which necessitates specialized autoclaves and rigorous safety protocols. Traditional routes frequently involve the use of ruthenium or platinum catalysts that are not only prohibitively expensive but also suffer from limited substrate scope, failing to tolerate sensitive functional groups that are common in advanced drug intermediates. Furthermore, many existing protocols demand the pre-activation of starting materials, such as converting amines into amides or utilizing unstable acid anhydrides, which adds multiple synthetic steps and generates substantial chemical waste prior to the actual ring-forming event. These multi-step sequences inevitably lead to lower overall yields and increased production costs, creating bottlenecks for procurement managers who are tasked with sourcing key intermediates at competitive prices. The reliance on harsh reaction conditions and narrow substrate compatibility further complicates the scale-up process, making it difficult for supply chain heads to guarantee consistent quality and continuity of supply for commercial manufacturing campaigns.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112480015B utilizes a cleverly designed one-pot strategy that merges nitro reduction, amidine formation, and carbonylative cyclization into a single seamless operation driven by a palladium catalyst system. This novel approach employs molybdenum hexacarbonyl as a safe and convenient solid source of carbon monoxide, thereby eliminating the logistical nightmares and safety risks associated with handling high-pressure CO gas cylinders in a production environment. The reaction proceeds efficiently at a moderate temperature of 120°C in common organic solvents like dioxane, utilizing inexpensive and widely available nitro compounds as the nitrogen source which significantly lowers the raw material cost basis. 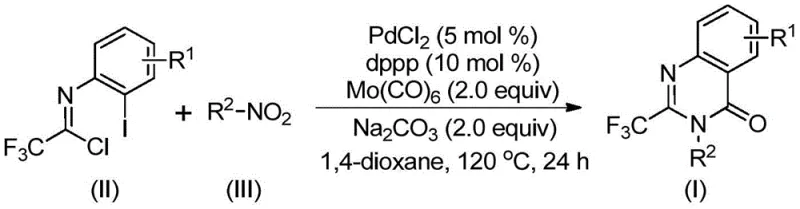 This methodology demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkyls, and electron-withdrawing groups on the aromatic rings without compromising reaction efficiency. By consolidating multiple transformation steps into a single vessel, this process drastically reduces solvent consumption, labor hours, and purification burdens, presenting a compelling value proposition for manufacturers aiming to optimize their production workflows.
This methodology demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkyls, and electron-withdrawing groups on the aromatic rings without compromising reaction efficiency. By consolidating multiple transformation steps into a single vessel, this process drastically reduces solvent consumption, labor hours, and purification burdens, presenting a compelling value proposition for manufacturers aiming to optimize their production workflows.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The elegance of this synthesis lies in its intricate catalytic cycle which orchestrates a series of distinct chemical transformations with high precision, beginning with the in situ reduction of the nitro group to an amine by the molybdenum hexacarbonyl species. Once the amine is generated, it undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate, setting the stage for the subsequent metal-catalyzed events. The palladium catalyst, coordinated with the dppp ligand, then inserts into the carbon-iodine bond of the imidoyl chloride moiety to generate a reactive organopalladium species that is primed for carbonyl insertion. As the molybdenum complex thermally decomposes, it releases carbon monoxide which inserts into the carbon-palladium bond to form an acyl-palladium intermediate, effectively building the carbonyl functionality of the quinazolinone ring. 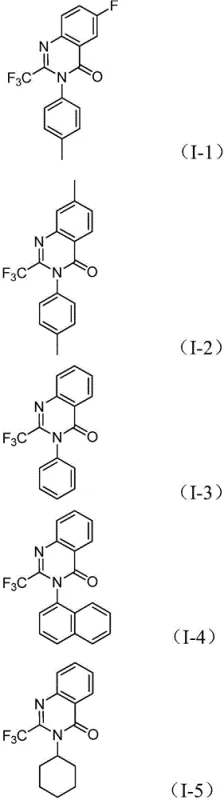 Following this insertion, the nitrogen atom of the amidine moiety coordinates with the palladium center to facilitate the formation of a seven-membered palladacycle, which subsequently undergoes reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product and regenerate the active catalyst. This mechanistic pathway ensures that the trifluoromethyl group is retained intact throughout the process, preserving the desired electronic and steric properties essential for the biological activity of the target molecule.
Following this insertion, the nitrogen atom of the amidine moiety coordinates with the palladium center to facilitate the formation of a seven-membered palladacycle, which subsequently undergoes reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product and regenerate the active catalyst. This mechanistic pathway ensures that the trifluoromethyl group is retained intact throughout the process, preserving the desired electronic and steric properties essential for the biological activity of the target molecule.
From an impurity control perspective, the use of a well-defined palladium-ligand system combined with sodium carbonate as a mild base helps to suppress side reactions such as homocoupling or hydrolysis of the sensitive imidoyl chloride starting material. The one-pot nature of the reaction minimizes the exposure of reactive intermediates to external contaminants, thereby simplifying the impurity profile and facilitating downstream purification efforts. The compatibility of the catalyst system with a wide range of substituents means that process chemists can fine-tune the electronic properties of the substrate to further enhance selectivity and yield without needing to redesign the entire synthetic route. This level of mechanistic understanding provides R&D directors with the confidence to adapt this chemistry for the synthesis of novel analogs, knowing that the core transformation is robust and predictable across different structural variations.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize the conversion of starting materials into the desired heterocyclic product. The protocol dictates the combination of palladium chloride, the bidentate phosphine ligand dppp, and sodium carbonate alongside the key reactants in an anhydrous organic solvent to ensure optimal catalyst performance. Heating the mixture to 120°C for a duration of 16 to 30 hours allows sufficient time for the slow release of carbon monoxide from the molybdenum source and the completion of the cyclization cascade. Detailed standardized synthetic steps see the guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent such as dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere chemical efficiency, primarily driven by the utilization of commodity-grade starting materials that are abundant in the global chemical market. The shift away from high-pressure gas infrastructure and precious metal catalysts like ruthenium towards a palladium system with a solid CO source translates directly into reduced capital expenditure and lower operational overheads for manufacturing facilities. Cost Reduction in Manufacturing: The elimination of specialized high-pressure reactors and the use of inexpensive nitro compounds as feedstocks significantly lowers the barrier to entry for producing these high-value intermediates, allowing for more competitive pricing structures in the final API market. By avoiding complex multi-step sequences and protecting group manipulations, the process reduces solvent usage and waste generation, which in turn lowers the costs associated with environmental compliance and waste disposal. This streamlined approach ensures that the cost of goods sold remains optimized even when scaling up to multi-kilogram or ton-level production runs required for commercial drug launches.
- Enhanced Supply Chain Reliability: The reliance on widely available nitro compounds and stable imidoyl chlorides mitigates the risk of supply disruptions that often plague specialized or custom-synthesized precursors, ensuring a steady flow of materials for continuous manufacturing operations. The robustness of the reaction conditions means that production schedules are less likely to be impacted by equipment failures or safety incidents related to hazardous gas handling, thereby improving overall plant uptime and delivery reliability. Furthermore, the broad substrate scope allows for the rapid sourcing of alternative starting materials if a specific supplier faces issues, providing procurement teams with greater flexibility and negotiating power in the marketplace.
- Scalability and Environmental Compliance: The one-pot nature of this reaction inherently reduces the number of unit operations required, simplifying the scale-up process from benchtop to commercial production while minimizing the footprint of the manufacturing facility. The use of a solid carbon monoxide surrogate eliminates the need for complex gas scrubbing systems and reduces the potential for atmospheric emissions, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry aspect not only facilitates regulatory approval but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity on its practical application and benefits for industrial partners. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines for quinazolinone-based therapeutics.
Q: What are the safety advantages of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using molybdenum hexacarbonyl (Mo(CO)6) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized pressure reactors, significantly reducing operational hazards and infrastructure costs associated with handling toxic CO gas.
Q: How does this method improve substrate compatibility compared to traditional routes?
A: The protocol demonstrates excellent tolerance for various functional groups including halogens, alkyls, and trifluoromethyl groups on both the nitro compound and the imidoyl chloride, allowing for the synthesis of diverse derivatives without extensive protecting group strategies.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes cheap and readily available starting materials like nitro compounds and operates under relatively mild thermal conditions without high-pressure equipment, making it highly amenable to commercial scale-up for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate innovative academic research into reliable commercial reality, and our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, ensuring that stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We are committed to delivering high-purity 2-trifluoromethyl quinazolinone intermediates that meet the exacting standards of the global pharmaceutical industry, supporting your drug development programs from early-stage discovery through to full-scale manufacturing.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs, where we can demonstrate how this efficient synthesis route can optimize your budget. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions about your supply chain strategy. Partner with us to leverage this cutting-edge technology and secure a competitive advantage in the development of next-generation quinazolinone-based medicines.
