Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Strategy for Global Pharma
Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Strategy for Global Pharma
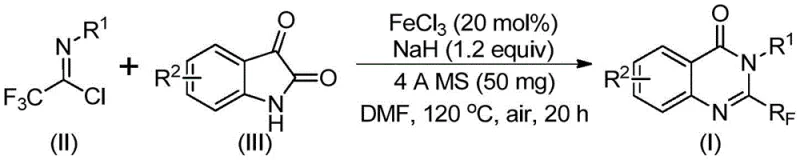
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and sustainable synthetic routes for bioactive scaffolds. A pivotal advancement in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are ubiquitous in medicinal chemistry, serving as the core structure for numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. The strategic introduction of a trifluoromethyl group further enhances the pharmacokinetic profile of these molecules by improving metabolic stability and lipophilicity. This patent presents a breakthrough methodology that utilizes inexpensive iron catalysis to construct these valuable heterocycles from readily available isatin and trifluoroethylimidoyl chloride precursors, marking a significant departure from traditional, resource-intensive synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with various trifluoromethylating agents. While effective in academic settings, these conventional methods suffer from severe limitations when viewed through the lens of industrial manufacturing. Commonly used trifluoromethyl synthons like trifluoroacetic anhydride or ethyl trifluoroacetate often necessitate harsh reaction conditions, including extreme temperatures or highly acidic/basic environments that can degrade sensitive functional groups. Moreover, these traditional routes frequently exhibit narrow substrate scope, meaning that slight modifications to the molecular scaffold can lead to drastic drops in yield or complete reaction failure. The reliance on expensive reagents and the generation of significant chemical waste further exacerbate the cost and environmental burden, making these methods less attractive for the large-scale production required by the global pharmaceutical supply chain.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined and economically viable alternative that leverages the power of earth-abundant iron catalysis. By utilizing trifluoroethylimidoyl chloride and isatin as the primary building blocks, this new route bypasses the need for costly precious metal catalysts and aggressive reagents. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by an iron-catalyzed decarbonylation and cyclization. This approach not only simplifies the operational procedure but also dramatically expands the functional group tolerance, allowing chemists to access a wider array of substituted quinazolinone derivatives with high efficiency. The ability to conduct the reaction under relatively mild conditions in common solvents like DMF represents a paradigm shift towards greener and more cost-effective manufacturing processes for these high-value intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by ferric chloride (FeCl3). The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin derivative in the presence of sodium hydride, which acts as a base to promote the initial nucleophilic attack. This step generates a transient trifluoroacetamidine intermediate, setting the stage for the subsequent cyclization event. The iron catalyst plays a dual role here: it likely coordinates with the carbonyl oxygen of the isatin moiety, activating it towards nucleophilic attack, and subsequently facilitates the crucial decarbonylation step. This decarbonylation is essential for aromatization and the formation of the stable quinazolinone ring system. The presence of 4A molecular sieves in the reaction mixture is also critical, as they serve to scavenge trace moisture that could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the catalyst, thereby ensuring high conversion rates and reproducibility.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods which often suffer from poor selectivity. The concerted nature of the cyclization and decarbonylation steps minimizes the formation of side products such as regioisomers or over-fluorinated species. The specific choice of FeCl3 as a Lewis acid ensures that the reaction trajectory is tightly controlled, favoring the formation of the desired 2-trifluoromethyl quinazolinone scaffold. Furthermore, the tolerance of the catalytic system towards various substituents on both the aryl ring of the imidoyl chloride and the benzene ring of the isatin suggests a robust mechanism that is not easily perturbed by electronic effects. This reliability is paramount for R&D teams aiming to develop libraries of analogs for structure-activity relationship (SAR) studies without encountering unpredictable purification challenges.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters defined in the patent to ensure optimal yields and purity. The process is designed to be operationally simple, avoiding the need for inert atmosphere techniques which adds to its practical appeal. The standard protocol involves mixing the catalyst, base, and molecular sieves in the solvent before introducing the substrates, followed by a staged heating process that begins at a lower temperature to facilitate mixing and initial bond formation before ramping up to drive the cyclization. For detailed procedural specifics regarding stoichiometry, exact temperature ramps, and work-up procedures, please refer to the standardized synthesis guide below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), and 4A molecular sieves in anhydrous DMF under air.
- Add trifluoroethylimidoyl chloride and isatin derivatives to the reaction mixture.
- Heat the reaction to 120°C for 18-20 hours, then purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology translates directly into tangible strategic benefits that enhance the overall resilience and profitability of the manufacturing operation. The shift away from precious metals and exotic reagents towards commodity chemicals fundamentally alters the cost structure of producing these complex heterocycles. By leveraging a catalyst that is abundant and inexpensive, manufacturers can insulate themselves from the volatile price fluctuations often associated with rare earth or noble metal markets. Additionally, the use of isatin and imidoyl chlorides as starting materials ensures a stable supply chain, as these are well-established bulk chemicals with multiple global suppliers, reducing the risk of single-source bottlenecks that can plague specialized synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts results in a drastic reduction in raw material costs per kilogram of product. Furthermore, the simplified post-treatment process, which relies on standard filtration and column chromatography rather than complex metal scavenging steps, lowers the operational expenditure associated with purification. This streamlined workflow reduces solvent consumption and labor hours, contributing to a leaner and more competitive cost base for the final active pharmaceutical ingredient (API) or intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics means that sourcing flexibility is significantly improved. Procurement teams are not locked into finding ultra-high purity grades of sensitive reagents, as the reaction tolerates a degree of variability. This flexibility allows for broader supplier qualification and faster turnaround times for raw material acquisition. Consequently, the lead time for producing high-purity quinazolinone derivatives is reduced, enabling faster response to market demands and clinical trial timelines.
- Scalability and Environmental Compliance: The protocol's compatibility with standard organic solvents like DMF and its operation under air (rather than strict inert gas conditions) greatly simplifies the engineering requirements for scale-up. This ease of translation from bench to plant reduces the capital investment needed for specialized reactor infrastructure. Moreover, the use of iron, a non-toxic metal, aligns with increasingly stringent environmental regulations regarding heavy metal residues in pharmaceutical products, simplifying the regulatory filing process and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what potential partners and licensees can expect from this methodology. Understanding these nuances is critical for making informed decisions about process adoption and integration into existing manufacturing portfolios.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers a substantial economic advantage as it is significantly cheaper than precious metal catalysts like palladium or rhodium. Furthermore, the protocol demonstrates excellent functional group tolerance, allowing for the synthesis of diverse derivatives without complex protection-deprotection steps.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides possibilities for industrial scale application. The use of readily available starting materials like isatin and simple post-treatment procedures supports robust scalability.
Q: What represents the primary impurity profile concern in this cyclization reaction?
A: The primary concern typically involves unreacted starting materials or incomplete decarbonylation intermediates. However, the optimized conditions using 4A molecular sieves and specific temperature ramps (40°C to 120°C) effectively drive the reaction to completion, minimizing these impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the next generation of therapeutic agents. As a leading CDMO partner, we possess the technical expertise to rapidly adapt and optimize this patented route to meet the specific needs of your drug development pipeline. Our facilities are equipped to handle complex heterocyclic chemistry with precision, ensuring that every batch meets the rigorous quality standards demanded by the global pharmaceutical industry. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and reliability regardless of the project phase.
We invite you to leverage our deep technical knowledge to accelerate your project timelines. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a seamless path from discovery to commercial success.
