Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
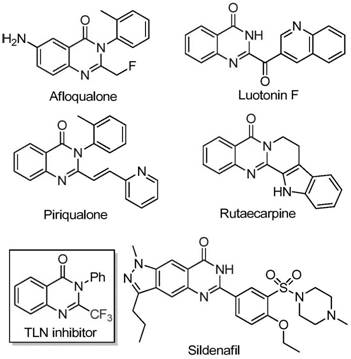
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. As detailed in the recent patent CN111675662B, a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds has been disclosed, addressing critical bottlenecks in current synthetic routes. This technology leverages an iron-catalyzed cyclization strategy that transforms readily available isatin derivatives and trifluoroethylimidoyl chlorides into high-value intermediates. The introduction of the trifluoromethyl group is strategically vital, as it significantly enhances the electronegativity, metabolic stability, and lipophilicity of the target molecules, properties that are highly sought after in the development of anti-cancer, anticonvulsant, and anti-inflammatory agents. For R&D directors and procurement specialists, understanding this shift from precious metal catalysis to base-metal catalysis represents a pivotal opportunity for optimizing supply chains and reducing the cost of goods sold (COGS) for complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on the cyclization of synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate with substrates like anthranilamide or isatoic anhydride. While these methods are chemically valid, they suffer from severe practical limitations that hinder their utility in large-scale manufacturing. The reaction conditions are often harsh, requiring strong acids or bases and elevated temperatures that can degrade sensitive functional groups on the substrate. Furthermore, the atom economy of these traditional routes is frequently poor, generating stoichiometric amounts of waste. From a commercial perspective, the reliance on specific, sometimes expensive, trifluoromethylating reagents increases the raw material burden. Additionally, the narrow substrate scope of these older methods often necessitates lengthy optimization campaigns for each new analog, delaying project timelines and increasing R&D expenditure significantly.
The Novel Approach
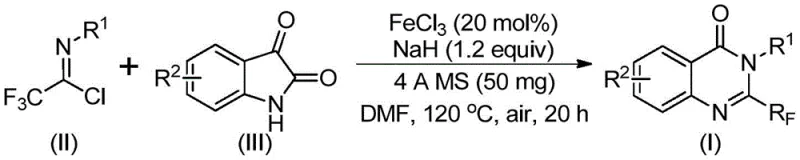
The methodology described in patent CN111675662B introduces a paradigm shift by utilizing trifluoroethylimidoyl chloride and isatin as the primary building blocks. This approach bypasses the need for pre-functionalized anthranilic acid derivatives, streamlining the synthetic sequence. The core innovation lies in the use of ferric chloride (FeCl3) as a catalyst in conjunction with sodium hydride (NaH) and 4A molecular sieves. This combination facilitates a tandem process involving initial carbon-nitrogen bond formation followed by an iron-catalyzed decarbonylation and cyclization. The result is a direct, one-pot transformation that constructs the quinazolinone core with high efficiency. This novel route not only simplifies the operational procedure but also dramatically expands the range of accessible chemical space, allowing for the incorporation of diverse substituents at multiple positions on the fused ring system without compromising yield or purity.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal catalysis. Initially, the sodium hydride acts as a base to deprotonate the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a transient trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst coordinates with the carbonyl oxygen of the isatin moiety, activating the system for the crucial decarbonylation step. The iron center facilitates the extrusion of carbon monoxide, driving the equilibrium towards the formation of the thermodynamically stable quinazolinone ring. The presence of 4A molecular sieves is critical in this mechanism; they act as a scavenger for trace water and potentially trap small molecule byproducts, shifting the reaction equilibrium forward and preventing the hydrolysis of the sensitive imidoyl chloride starting material. This careful orchestration of reagents ensures high conversion rates and minimizes the formation of hydrolytic impurities, which is a common challenge in trifluoromethyl chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for process chemistry teams. The use of an iron catalyst, as opposed to palladium or copper, eliminates the risk of heavy metal contamination that requires stringent and costly purification steps to meet ICH Q3D guidelines for elemental impurities in drug substances. The reaction proceeds through a well-defined cyclic transition state that favors the formation of the desired regioisomer. Furthermore, the tolerance of the catalytic system towards various electronic environments on the aromatic rings (electron-donating methyl groups or electron-withdrawing halogens) suggests a robust catalytic cycle that is not easily poisoned by substrate variations. This robustness translates directly to a cleaner crude reaction profile, reducing the load on downstream purification units and improving the overall mass balance of the manufacturing process.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and safety. The protocol involves charging a reactor with ferric chloride (20 mol%) and sodium hydride (1.2 equivalents) under an inert atmosphere, followed by the addition of 4A molecular sieves to maintain anhydrous conditions. The substrates, trifluoroethylimidoyl chloride and the chosen isatin derivative, are dissolved in an aprotic polar solvent such as DMF. The reaction is initiated at a moderate temperature of 40°C for 8-10 hours to allow for the initial coupling, followed by heating to 120°C for 18-20 hours to drive the cyclization to completion. This two-stage temperature profile is essential for balancing reaction rate with selectivity. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical documentation below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for 8-10 hours to initiate the coupling, then heat to 120°C for 18-20 hours to complete the cyclization.
- Filter the reaction mixture, purify via silica gel column chromatography to isolate the high-purity 2-trifluoromethyl substituted quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost reduction and supply security. The shift away from precious metal catalysts to abundant iron salts drastically reduces the direct material costs associated with catalysis. Moreover, the starting materials—isatin derivatives and trifluoroethylimidoyl chlorides—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The operational simplicity of the reaction, which does not require specialized high-pressure equipment or cryogenic conditions, further lowers the barrier to entry for contract manufacturing organizations (CMOs), fostering a more competitive bidding environment for production services.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts such as palladium or rhodium results in substantial cost savings per kilogram of produced intermediate. Additionally, the high atom economy of the decarbonylative cyclization reduces the volume of waste solvent and reagents required for workup, lowering waste disposal costs. The use of DMF, a common industrial solvent, allows for efficient recovery and recycling, further enhancing the economic viability of the process compared to methods requiring exotic or difficult-to-recycle solvents.
- Enhanced Supply Chain Reliability: By utilizing widely available feedstock chemicals like isatin and simple aromatic amines (precursors to the imidoyl chloride), the supply chain becomes more resilient to market fluctuations. Unlike specialized fluorinating reagents that may have long lead times or limited production capacity, the precursors for this method are produced on a multi-ton scale globally. This abundance ensures consistent availability and stabilizes pricing, allowing for more accurate long-term budgeting and inventory planning for pharmaceutical projects.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up in standard stainless steel reactors, avoiding the need for glass-lined vessels required for highly corrosive alternatives. The absence of heavy metals simplifies the environmental compliance landscape, reducing the regulatory burden associated with effluent treatment and final product testing. This 'green chemistry' aspect aligns with modern sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN111675662B, providing a factual basis for evaluating the technology's fit within your existing pipeline. Understanding these nuances is critical for making informed decisions about process adoption and vendor selection.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost advantages over precious metal catalysts like palladium or rhodium. It is inexpensive, readily available, and demonstrates high catalytic efficiency for this specific decarbonylative cyclization, reducing the overall raw material cost for API intermediates.
Q: How does this method improve functional group tolerance compared to prior art?
A: This protocol utilizes mild initial conditions (40°C) followed by thermal cyclization, which allows for excellent tolerance of sensitive functional groups such as halogens (F, Cl, Br), nitro groups, and methoxy substituents on the aromatic rings, enabling the synthesis of diverse drug-like scaffolds without extensive protection-deprotection steps.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states the method is suitable for gram-level expansion and industrial scale application. The use of common solvents like DMF and robust inorganic reagents simplifies the engineering controls required for large-scale manufacturing compared to air-sensitive organometallic processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in accelerating drug discovery and development. Our technical team has extensively analyzed the FeCl3-catalyzed route described in CN111675662B and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. Our facilities are designed to handle the specific thermal profiles and solvent systems required for this chemistry, backed by rigorous QC labs that guarantee stringent purity specifications for every batch delivered.
We invite you to leverage our expertise to optimize your supply chain for quinazolinone-based APIs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for related intermediates and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable, cost-effective, and high-quality supply of these critical pharmaceutical building blocks.
