Revolutionizing Lasmiditan Production: Advanced Catalytic Technology for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active pharmaceutical ingredients, and the recent technological advancements detailed in patent CN115477635A represent a significant leap forward for Lasmiditan production. This novel methodology addresses critical bottlenecks associated with traditional manufacturing processes by introducing a copper-catalyzed oxidative coupling strategy that fundamentally alters the reaction landscape. By shifting away from hazardous acid chloride intermediates, this approach not only enhances operational safety but also drastically improves the impurity profile of the final drug substance. For global supply chain stakeholders, this innovation translates into a more reliable source of high-purity migraine treatment medications, ensuring consistent availability for patients worldwide. The strategic implementation of this chemistry positions manufacturers to meet the growing demand for CNS therapeutics with greater efficiency and regulatory compliance. 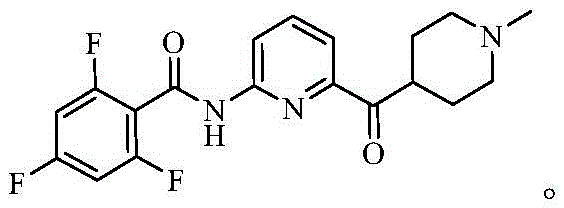
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lasmiditan has relied heavily on routes involving 2,4,6-trifluorobenzoyl chloride, which presents severe challenges for industrial scale-up and process safety. The use of acid chlorides necessitates rigorous anhydrous conditions and generates corrosive by-products like hydrochloric acid, which can damage reactor linings and complicate waste management protocols. Furthermore, these conventional pathways are prone to forming diacylated impurities that are structurally similar to the target molecule, requiring extensive and costly purification steps to ensure pharmaceutical grade quality. Older methods also frequently employ expensive noble metal catalysts such as palladium complexes alongside sensitive organometallic reagents like n-butyllithium, which demand ultra-low temperature controls and inert atmospheres. These factors collectively drive up production costs and introduce significant supply chain vulnerabilities due to the reliance on specialized raw materials and equipment. 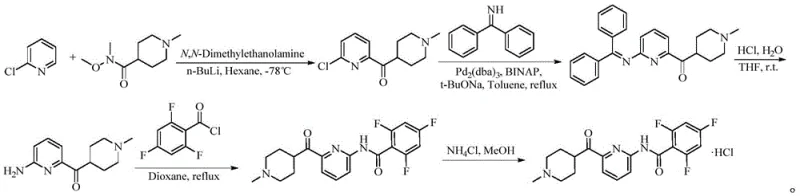
The Novel Approach
In stark contrast, the innovative method disclosed in the patent utilizes a direct oxidative coupling between an amine substrate and an aldehyde, catalyzed by abundant and cost-effective copper salts. This transformation proceeds under mild thermal conditions using molecular oxygen or air as the terminal oxidant, eliminating the need for hazardous activating agents and reducing the overall environmental footprint of the manufacturing process. The reaction demonstrates exceptional chemoselectivity, effectively suppressing the formation of over-acylated side products that plague traditional acid chloride routes. By simplifying the synthetic sequence and removing the requirement for cryogenic temperatures or high-pressure ammonia treatments, this new approach offers a streamlined pathway that is inherently safer and more economically viable for large-scale operations. The robustness of this copper-catalyzed system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining regulatory approval and market supply continuity. 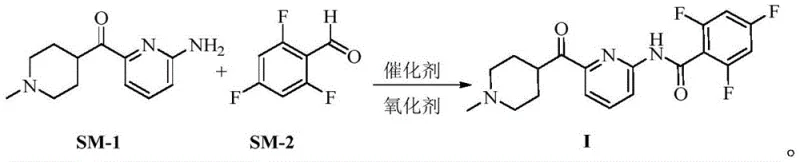
Mechanistic Insights into Copper-Catalyzed Oxidative Amidation
The core of this technological breakthrough lies in the mechanistic efficiency of the copper-catalyzed oxidative amidation, which facilitates the direct formation of the amide bond without pre-activation of the carboxylic acid component. The catalytic cycle likely involves the coordination of the copper species with the amine and aldehyde substrates, followed by oxidation to generate a reactive intermediate that undergoes nucleophilic attack. This mechanism bypasses the formation of highly reactive acyl chlorides, thereby avoiding the electrophilic aromatic substitution side reactions that lead to diacylation on the electron-rich pyridine ring. The use of molecular oxygen as the oxidant ensures that the only by-product is water, aligning with green chemistry principles and simplifying downstream processing requirements. Understanding this mechanistic pathway allows process chemists to fine-tune reaction parameters such as catalyst loading and oxygen flow rates to maximize conversion and minimize trace impurities.
Controlling the impurity profile is paramount for pharmaceutical intermediates, and this new route excels by inherently preventing the generation of the problematic diacylated species often seen in prior art. In traditional methods, the presence of excess acid chloride and the basicity of the reaction medium can lead to double acylation, creating impurities that are difficult to separate due to their similar polarity and solubility characteristics. The oxidative coupling method operates under neutral to mildly basic conditions without strong acylating agents, significantly reducing the thermodynamic driving force for secondary acylation events. This results in a crude product profile that is much cleaner, reducing the burden on crystallization and chromatography units during purification. Consequently, the overall yield is preserved at high levels, and the final API meets stringent specifications for related substances without requiring aggressive recrystallization cycles that can erode mass balance. 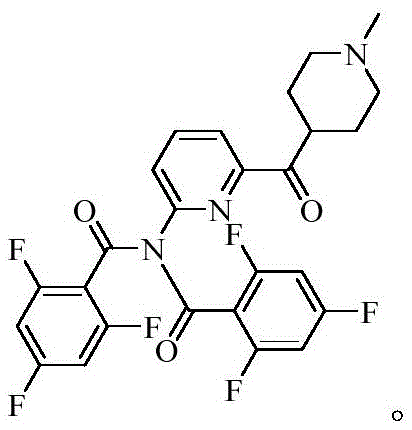
How to Synthesize Lasmiditan Efficiently
Implementing this synthesis requires careful attention to the selection of the copper catalyst and the control of the oxidation environment to ensure optimal reaction kinetics. The process begins with the dissolution of the amine and aldehyde starting materials in a polar aprotic solvent, followed by the addition of the copper catalyst and the introduction of oxygen gas. Detailed standardized operating procedures regarding temperature ramping, gas flow rates, and workup protocols are essential to replicate the high yields and purity reported in the patent literature. Manufacturers should focus on maintaining precise stoichiometric ratios and monitoring reaction progress via HPLC to determine the exact endpoint for quenching.
- Prepare the reaction mixture by combining 2-amino-6-(1-methylpiperidin-4-yl acyl)-pyridine and 2,4,6-trifluorobenzaldehyde in a polar aprotic solvent.
- Add a copper-based catalyst such as CuI and introduce an oxidant like oxygen or air into the system under controlled temperature conditions.
- Upon completion, filter the reaction mixture, extract with organic solvents, and purify to obtain high-purity Lasmiditan without diacylated impurities.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed oxidative coupling technology offers substantial advantages in terms of cost structure and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for expensive palladium catalysts and hazardous acid chlorides, the raw material costs are significantly reduced, allowing for more competitive pricing in the global API market. The simplified process flow also reduces the consumption of solvents and energy, contributing to lower operational expenditures and a smaller carbon footprint for the production facility. These efficiencies translate into a more stable supply of Lasmiditan, mitigating the risks of production delays caused by the scarcity of specialized reagents or equipment maintenance issues associated with corrosive chemicals.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with inexpensive copper salts drastically lowers the catalyst cost per kilogram of product, while the avoidance of cryogenic conditions reduces energy consumption for cooling systems. Furthermore, the elimination of distillation steps required to purify acid chlorides removes a significant unit operation, streamlining the production timeline and reducing labor costs associated with complex handling procedures. The higher overall yield of the process means less starting material is wasted, directly improving the cost of goods sold and enhancing profit margins for manufacturers.
- Enhanced Supply Chain Reliability: Utilizing air or oxygen as the oxidant removes the dependency on specialized chemical oxidants that may face supply constraints or regulatory shipping restrictions. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities without the need for specialized Hastelloy reactors resistant to strong acids, thereby expanding the potential supplier base. This flexibility ensures that procurement teams can secure multiple sources of supply, reducing the risk of single-source bottlenecks and ensuring continuous availability for downstream formulation partners.
- Scalability and Environmental Compliance: The benign nature of the reagents and by-products simplifies waste treatment processes, making it easier to comply with increasingly stringent environmental regulations regarding hazardous waste disposal. The process is inherently safer, reducing the risk of accidents related to exothermic reactions or toxic gas releases, which lowers insurance premiums and liability risks for the manufacturing site. Scalability is enhanced by the straightforward workup procedure, which involves simple filtration and extraction, allowing for seamless transition from pilot plant to multi-ton commercial production without significant process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Lasmiditan synthesis method. These insights are derived directly from the patent data and practical considerations for scaling oxidative coupling reactions in a GMP environment. Understanding these aspects helps stakeholders evaluate the feasibility and benefits of adopting this technology for their supply chains.
Q: How does the new copper-catalyzed method improve upon traditional acid chloride routes?
A: The novel oxidative coupling method eliminates the use of corrosive 2,4,6-trifluorobenzoyl chloride, thereby preventing the formation of difficult-to-remove diacylated impurities and reducing equipment corrosion risks significantly.
Q: What are the purity specifications achievable with this synthetic route?
A: This process consistently delivers Lasmiditan with purity levels exceeding 99.8%, meeting stringent pharmaceutical standards without the need for complex chromatographic purification steps often required in older methods.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, inexpensive copper catalysts, and avoids hazardous reagents like n-butyllithium, making it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lasmiditan Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge synthetic methodologies like the copper-catalyzed oxidative coupling route to deliver superior quality Lasmiditan to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the highest international pharmacopoeia standards for CNS active pharmaceutical ingredients. Our commitment to technological innovation allows us to offer a product that is not only cost-effective but also environmentally sustainable and safe to handle.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains and reduce their overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term business goals and ensure a steady supply of this critical migraine medication.
