Scalable Copper-Catalyzed Synthesis of Lasmiditan for Commercial API Production
Introduction to Advanced Lasmiditan Manufacturing
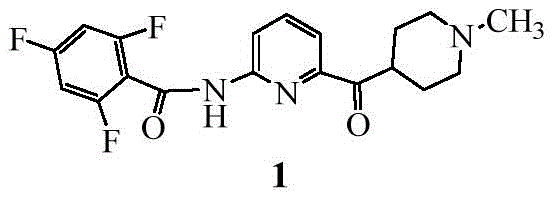
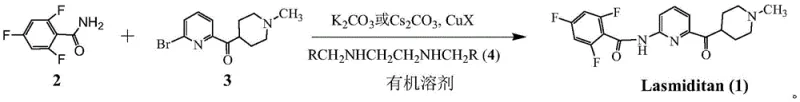
The pharmaceutical landscape for migraine treatment has shifted significantly with the advent of Lasmiditan, a selective 5-HT1F receptor agonist. As demand for this active pharmaceutical ingredient (API) grows, the industry requires robust, scalable, and cost-effective synthetic routes. Patent CN111943930A introduces a transformative synthesis process that addresses the critical bottlenecks of earlier methodologies. This technology leverages a modified Goldberg amidation reaction, utilizing a copper-catalyzed system to couple 2,4,6-trifluorobenzamide with a bromopyridine derivative. Unlike traditional approaches that rely on harsh conditions or precious metals, this innovation operates under mild temperatures ranging from 0°C to 60°C, specifically optimizing around room temperature. The strategic implementation of specific diamine ligands and weak inorganic bases allows for high conversion rates while maintaining exceptional product purity. For global supply chains, this represents a pivotal shift towards sustainable and economically viable manufacturing of complex neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for Lasmiditan have been plagued by significant technical and economic inefficiencies that hinder large-scale commercialization. Route A, for instance, relies on a palladium-catalyzed amination followed by hydrolysis and subsequent acylation with trifluorobenzoyl chloride. This pathway is fraught with dangers; the preparation and distillation of trifluorobenzoyl chloride generate volatile, corrosive acidic substances that severely damage equipment and pose safety risks. Furthermore, the acylation step often leads to di-acylated byproducts, necessitating complex hydrolysis steps to recover the desired product, which drastically lowers overall yield and increases unit consumption of reagents. Similarly, Route C employs a palladium-catalyzed Goldberg reaction using expensive ligands like BINAP and bases like sodium tert-butoxide, yet it suffers from disappointingly low yields of approximately 37%. These conventional methods not only inflate production costs due to the reliance on noble metals but also create substantial waste streams that complicate environmental compliance and purification processes.

The Novel Approach
The methodology disclosed in CN111943930A fundamentally reengineers the coupling step to overcome these historical barriers. By replacing palladium with abundant cuprous salts (CuI, CuBr, or CuCl) and employing specific N,N'-dialkyl or N,N'-diarylmethyl ethylenediamine ligands, the process achieves efficient C-N bond formation without the need for precious metals. A key breakthrough lies in the solvent system; the inventors discovered that low molecular weight solvents such as tetrahydrofuran, ethyl acetate, or acetonitrile effectively solubilize the trifluorobenzamide substrate at room temperature, a condition where traditional solvents like toluene fail. This allows the reaction to proceed smoothly at 20-30°C, avoiding the thermal decomposition observed at temperatures above 60°C. The result is a streamlined process that eliminates the need for hazardous acyl chloride intermediates and expensive catalysts, delivering Lasmiditan with superior yield and quality suitable for industrial application.
Mechanistic Insights into Copper-Catalyzed Goldberg Amidation
The success of this synthesis hinges on the precise coordination chemistry between the cuprous salt and the diamine ligand. The reaction mechanism involves the oxidative addition of the aryl bromide to the copper center, facilitated by the electron-donating properties of the diamine ligand. The patent highlights that not all ligands are created equal; while simple N,N'-dimethyl-1,2-ethylenediamine is effective, sterically hindered or electron-deficient aryl-substituted diamines (such as N,N'-bis(4-cyanophenylmethyl)-1,2-ethylenediamine) can further enhance catalytic activity and suppress side reactions. The choice of ligand directly influences the stability of the copper-amide complex, preventing the precipitation of inactive copper species that often stall traditional Goldberg reactions. Additionally, the use of potassium carbonate as a base is critical; it is sufficiently strong to deprotonate the amide nitrogen to form the reactive nucleophile but mild enough to prevent the degradation of the sensitive fluorinated aromatic ring or the piperidine moiety. This delicate balance ensures that the catalytic cycle turns over efficiently, minimizing the formation of homocoupling byproducts or dehalogenated impurities.
Impurity control is another cornerstone of this mechanistic design. In previous routes, the use of acyl chlorides led to over-acylation, creating difficult-to-remove di-amide impurities. By utilizing the pre-formed 2,4,6-trifluorobenzamide directly in the coupling reaction, this risk is entirely eliminated. The reaction conditions are tuned to favor the mono-coupled product, and the mild temperature profile prevents the thermal rearrangement or decomposition of the trifluorophenyl group. Furthermore, the workup procedure described involves washing with sodium sulfide to chelate and remove residual copper, followed by acid-base extraction. This ensures that the final API intermediate meets stringent heavy metal specifications required by regulatory bodies. The ability to control the impurity profile at the molecular level through ligand and solvent selection demonstrates a deep understanding of the reaction kinetics, providing a robust platform for consistent batch-to-batch quality.
How to Synthesize Lasmiditan Efficiently
The synthesis of Lasmiditan via this copper-catalyzed route offers a practical and scalable solution for manufacturing teams looking to optimize their production lines. The process begins with the preparation of the key starting materials, 2,4,6-trifluorobenzamide and the bromopyridine ketone derivative, which are commercially accessible or easily synthesized. The core coupling reaction is performed by suspending the reactants in a suitable organic solvent such as THF or ethyl acetate, adding the copper catalyst and ligand, and stirring at ambient temperature. Detailed standard operating procedures regarding stoichiometry, addition rates, and specific workup protocols are essential for maximizing yield and purity. For a comprehensive guide on the exact experimental conditions and purification steps validated in the patent examples, please refer to the standardized synthesis instructions below.
- Prepare the reaction mixture by combining 2,4,6-trifluorobenzamide, (6-bromopyridin-2-yl)(1-methylpiperidin-4-yl)methanone, cuprous salt (CuI/CuBr/CuCl), and a diamine ligand in an organic solvent like THF or ethyl acetate.
- Add a weak inorganic solid base such as potassium carbonate (K2CO3) and stir the mixture at mild temperatures between 0°C and 60°C, optimally at room temperature (20-30°C).
- Upon completion, filter the reaction mass, wash with sodium sulfide to remove copper residues, and perform acid-base extraction to isolate high-purity Lasmiditan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this copper-catalyzed process offers tangible strategic benefits that extend beyond simple chemistry. The elimination of palladium catalysts and phosphine ligands removes a significant cost driver and supply risk, as these materials are subject to volatile market pricing and geopolitical supply constraints. By switching to earth-abundant copper salts and simple diamine ligands, manufacturers can achieve substantial cost savings in raw material procurement. Moreover, the avoidance of trifluorobenzoyl chloride distillation reduces the need for specialized corrosion-resistant equipment and lowers maintenance costs associated with handling hazardous acidic vapors. This simplification of the process infrastructure allows for faster turnaround times and more flexible production scheduling, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with economical copper salts drastically lowers the direct material cost per kilogram of the API. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs. The simplified workup procedure minimizes solvent usage and waste disposal fees, further improving the economic profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like cuprous iodide and potassium carbonate ensures a stable and diversified supply base, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction at room temperature also means that the process is less sensitive to minor fluctuations in utility availability, ensuring consistent output even in varying operational environments.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to acyl chloride-based routes, simplifying environmental permitting and waste treatment operations. The absence of heavy metal contamination risks facilitates easier regulatory approval and reduces the burden on quality control laboratories for extensive metal scavenging validation, accelerating the path to commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on feasibility and performance. Understanding these nuances is crucial for R&D teams evaluating the technology for technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why is the copper-catalyzed route superior to palladium methods for Lasmiditan?
A: The copper-catalyzed route described in CN111943930A eliminates the need for expensive noble metal catalysts like Pd2dba3 and ligands like BINAP. It operates under significantly milder conditions (room temperature vs. high heat) and avoids the low yields associated with previous palladium-mediated Goldberg reactions.
Q: What are the critical parameters for solvent selection in this synthesis?
A: Solvent selection is critical because 2,4,6-trifluorobenzamide has poor solubility in many common coupling solvents at room temperature. The patent identifies low molecular weight esters, ethers, and nitriles (e.g., ethyl acetate, THF, acetonitrile) as optimal, whereas traditional solvents like toluene or DMF result in poor conversion or decomposition.
Q: How does this process improve supply chain reliability for API manufacturers?
A: By utilizing readily available cuprous salts and simple diamine ligands instead of scarce palladium complexes, the process reduces raw material procurement risks. Furthermore, the mild reaction conditions minimize equipment corrosion and safety hazards associated with acyl chloride distillation, ensuring more consistent production cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lasmiditan Supplier
As the global demand for migraine therapeutics continues to rise, securing a dependable source of high-quality Lasmiditan intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the copper-mediated coupling described in CN111943930A to deliver superior products. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Lasmiditan intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative synthesis capabilities can drive value for your organization.
