Advanced Four-Step Synthesis of Piribedil: A Scalable Solution for Parkinson’s Disease API Manufacturing
Introduction to the Novel Piribedil Synthesis Technology
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for the production of active pharmaceutical ingredients (APIs), particularly for neurological treatments such as Parkinson's disease. Patent CN103373991A discloses a groundbreaking method for preparing Piribedil, a dopamine agonist, achieving exceptional purity levels exceeding 99% through a streamlined four-step sequence. This innovative approach departs from conventional high-pressure or noble-metal catalyzed routes, instead leveraging a strategic combination of Blanc chloromethylation and protected piperazine chemistry to ensure both high yield and operational simplicity. By utilizing piperonyl cyclonene as the foundational raw material, the process effectively mitigates the formation of complex impurity profiles often associated with direct alkylation methods. The technical significance of this patent lies in its ability to deliver a commercially attractive manufacturing protocol that balances rigorous quality control with practical engineering constraints, making it an ideal candidate for industrial adoption by forward-thinking chemical enterprises.
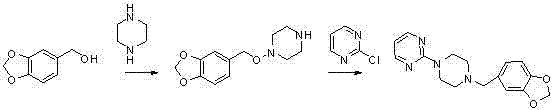
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Piribedil has been plagued by significant technical hurdles that impact both cost efficiency and supply chain reliability. Prior art, such as the method disclosed in Chinese patent CN1884280A, relies heavily on reductive amination using piperonylaldehyde and piperazine, a process that necessitates hazardous high-pressure hydrogenation conditions ranging from 20 to 60 bar. Such extreme operating parameters not only demand specialized, capital-intensive reactor infrastructure but also introduce substantial safety risks that can disrupt continuous manufacturing operations. Furthermore, alternative literature routes, including those utilizing Ruthenium catalysts for borrowing hydrogen transformations, introduce the complication of expensive noble metals which require rigorous removal protocols to meet stringent regulatory limits for residual metals in pharmaceutical products. These traditional pathways often suffer from lower overall yields, sometimes as low as 32% in older three-step sequences, and generate difficult-to-remove byproducts that compromise the final purity of the API, thereby increasing the cost of goods sold through extensive purification requirements.
The Novel Approach
In stark contrast, the methodology outlined in CN103373991A presents a paradigm shift by employing a mild, metal-free, and atmospheric pressure synthetic strategy that fundamentally alters the economic landscape of Piribedil production. This novel approach replaces the problematic reductive amination step with a highly controlled nucleophilic substitution using N-Boc protected piperazine, effectively eliminating the need for high-pressure hydrogenation equipment and the associated safety overheads. By introducing a temporary protecting group on the piperazine nitrogen, the process achieves superior regioselectivity, preventing the formation of bis-alkylated side products that typically plague unprotected amine reactions. The result is a cleaner reaction profile that facilitates easier downstream processing, such as simple crystallization or filtration, rather than complex chromatographic separations. This transition from harsh, metal-dependent chemistry to a benign, organic-synthetic workflow represents a significant technological upgrade, offering manufacturers a pathway to reduce lead time for high-purity APIs while simultaneously lowering the barrier to entry for commercial scale-up.
Mechanistic Insights into Blanc Chloromethylation and Protected Amine Coupling
The cornerstone of this improved synthesis lies in the initial Blanc chloromethylation step, where piperonyl cyclonene is transformed into 3,4-methylenedioxy benzyl chloride using paraformaldehyde and hydrochloric acid in the presence of a phase-transfer catalyst. This electrophilic aromatic substitution is meticulously optimized to proceed at mild temperatures between -10°C and 20°C, ensuring that the sensitive methylenedioxy ring remains intact while efficiently installing the reactive chloromethyl handle. The use of phase-transfer catalysts, such as tetrabutylammonium bromide, enhances the interfacial contact between the organic substrate and the aqueous acid phase, driving the reaction to completion with high conversion rates without the need for excessive thermal energy. Following this activation, the subsequent coupling with N-Boc-piperazine is a critical mechanistic maneuver; the bulky tert-butoxycarbonyl group sterically hinders the second nitrogen of the piperazine ring, forcing the alkylation to occur exclusively at the desired position. This steric control is vital for maintaining the structural integrity of the intermediate and preventing the generation of symmetrical bis-alkylated impurities that would otherwise require difficult separation techniques later in the process.
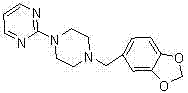
Furthermore, the final condensation step involves the reaction of the deprotected piperazine intermediate with 2-chloropyrimidine under alkaline conditions, a nucleophilic aromatic substitution that proceeds efficiently in polar protic solvents like ethanol or methanol. The choice of base, typically potassium carbonate or sodium bicarbonate, is crucial for neutralizing the hydrochloric acid byproduct generated during the displacement of the chloride ion, thereby shifting the equilibrium towards the formation of the final Piribedil product. The entire sequence is designed to minimize the generation of hazardous waste and avoid the use of toxic solvents, aligning with modern green chemistry principles that are increasingly demanded by global regulatory bodies. By understanding these mechanistic nuances, R&D directors can appreciate how each step is engineered not just for chemical conversion, but for the suppression of specific impurity vectors, ensuring that the final API meets the rigorous purity specifications required for neurological therapeutics without the need for exhaustive re-crystallization cycles.
How to Synthesize Piribedil Efficiently
The synthesis of Piribedil via this patented route offers a clear, step-by-step protocol that transitions seamlessly from laboratory bench scale to multi-ton commercial production. The process begins with the activation of the aromatic core followed by the strategic assembly of the piperazine-pyrimidine scaffold, utilizing standard unit operations familiar to any competent chemical manufacturing facility. Detailed below is the procedural framework derived from the patent examples, which highlights the specific reagents, stoichiometry, and workup procedures necessary to achieve the reported high yields and purity profiles. Operators should note that while the conditions are mild, precise control of pH during the extraction phases and temperature during the reflux steps is essential to maximize the recovery of the intermediate species.
- Perform Blanc chloromethylation on piperonyl cyclonene using paraformaldehyde and hydrochloric acid with a phase-transfer catalyst to generate 3,4-methylenedioxy benzyl chloride.
- Condense the benzyl chloride intermediate with N-Boc protected piperazine under alkaline conditions to form the protected piperazine derivative, minimizing bis-alkylation side reactions.
- Execute acidic deprotection to remove the Boc group, followed by condensation with 2-chloropyrimidine to yield the final Piribedil API intermediate with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies that strengthen the overall supply network. The elimination of high-pressure hydrogenation steps removes a major bottleneck in production scheduling, as facilities no longer need to allocate scarce high-pressure reactor capacity or manage the complex logistics of hydrogen gas supply. Additionally, the avoidance of noble metal catalysts like Ruthenium eradicates the volatility associated with precious metal pricing and supply shortages, creating a more stable and predictable cost structure for raw materials. The simplified workup procedures, which rely on standard extractions and crystallizations rather than complex chromatography, significantly reduce the consumption of silica gel and organic solvents, leading to a drastic reduction in waste disposal costs and environmental compliance burdens. These factors combined create a resilient manufacturing model that is less susceptible to external market shocks and regulatory changes regarding heavy metal residues.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive noble metal catalysts and high-pressure equipment requirements, which traditionally inflate the capital expenditure and operating costs of API production. By utilizing commodity chemicals such as paraformaldehyde, hydrochloric acid, and protected piperazines, the raw material cost profile is significantly flattened, allowing for substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the high selectivity of the protected amine coupling reduces the loss of valuable starting materials to side reactions, improving the overall mass balance and atom economy of the process. This efficiency gain means that less raw material is required per kilogram of finished product, directly enhancing the gross margin potential for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available, bulk commodity chemicals ensures that the supply chain is robust and less prone to disruptions caused by the scarcity of specialized reagents. Unlike routes dependent on custom-synthesized aldehydes or rare earth catalysts, the inputs for this process are sourced from mature, high-volume chemical markets, guaranteeing consistent availability and competitive pricing. The mild reaction conditions also imply that the process can be executed in a wider range of manufacturing facilities, including those without specialized high-pressure capabilities, thereby expanding the pool of potential contract manufacturing organizations (CMOs) and diversifying the supply base. This flexibility is crucial for maintaining continuity of supply in the face of geopolitical or logistical challenges that might affect specialized chemical suppliers.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a cleaner profile by avoiding the generation of heavy metal waste streams that require costly and energy-intensive treatment before discharge. The use of common solvents like ethanol and methanol, which can be easily recovered and recycled through distillation, further minimizes the environmental footprint and aligns with increasingly strict global sustainability mandates. The scalability of the process is evidenced by the use of standard batch reactor operations, which can be linearly scaled from pilot plants to hundred-ton production lines without the need for complex process intensification technologies. This ease of scale-up reduces the time-to-market for new generic formulations and allows manufacturers to respond rapidly to fluctuations in market demand for Parkinson's disease medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Piribedil synthesis technology, providing clarity on its advantages over legacy methods. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, offering a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: How does this new synthesis route improve upon traditional reductive amination methods?
A: Traditional methods often require high-pressure hydrogenation (20-60 bar) for reductive amination, posing significant safety risks and equipment costs. This novel route utilizes mild nucleophilic substitution conditions at atmospheric pressure, drastically simplifying the engineering requirements and enhancing operational safety.
Q: What is the primary advantage of using N-Boc protected piperazine in this process?
A: Using N-Boc protected piperazine prevents the formation of bis-alkylated impurities, which are common when using unprotected piperazine. This strategic protection ensures higher regioselectivity during the alkylation step, leading to a cleaner crude product and significantly reducing the burden on downstream purification processes.
Q: Does this process rely on expensive noble metal catalysts?
A: No, unlike alternative routes that utilize Ruthenium (Ru) catalysts for borrowing hydrogen methodologies, this synthesis is entirely metal-free. This eliminates the risk of heavy metal contamination in the final API and removes the costly and complex step of metal scavenging, resulting in substantial cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piribedil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to secure a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering high-purity Piribedil intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in process optimization and impurity control, we can guarantee a supply of material that not only meets but exceeds the quality expectations of top-tier pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. Request a Customized Cost-Saving Analysis today to understand the full economic potential of switching to this metal-free, high-yield process. Our experts are ready to provide specific COA data and route feasibility assessments to support your next project milestone, ensuring that your supply chain remains robust, compliant, and cost-effective in the long term.
