Advanced Piribedil Synthesis: Scalable Routes for Pharmaceutical Intermediate Supply Chains
Advanced Piribedil Synthesis: Scalable Routes for Pharmaceutical Intermediate Supply Chains
The pharmaceutical industry constantly seeks robust manufacturing pathways for dopaminergic agonists like Piribedil, essential for treating Parkinson's disease. Patent CN101735201A introduces a transformative preparation method that addresses critical bottlenecks in existing synthetic routes. This technology leverages piperidine as a cost-effective starting material and employs a Blanc reaction to generate piperonyl chloride, followed by selective alkylation steps. The innovation lies in its ability to operate under moderate conditions without high temperature or pressure, ensuring safety and scalability. By achieving HPLC purity levels not lower than 99.8 percent, this method sets a new benchmark for quality in pharmaceutical intermediate production. For global supply chain leaders, this represents a viable strategy to secure high-purity API intermediates with reduced operational risk.
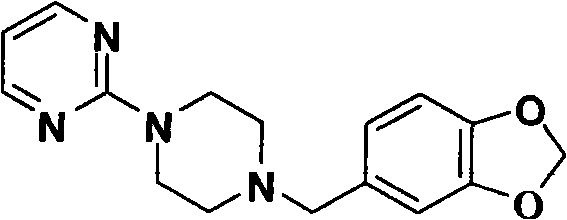
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, Piribedil synthesis has been plagued by inefficient routes that hinder commercial viability. Route 1, for instance, relies on reduction amination under hydrogen pressure of 2 to 60 bar, demanding specialized equipment and posing significant safety hazards. Furthermore, subsequent N-hydrocarbonylation steps often utilize toxic solvents like DMF or tetrahydrofuran, yielding only 40 percent to 44 percent, which is economically unsustainable. Route 2 employs expensive ruthenium catalysts such as Ru[(p-cymene)Cl2]2, drastically inflating raw material costs and introducing heavy metal contamination risks that require complex removal processes. Other methods involve tedious ion exchange chromatography or produce excessive double alkylation byproducts, resulting in total recoveries as low as 32.4 percent. These technical deficiencies create substantial barriers for procurement teams seeking reliable pharmaceutical intermediates supplier partnerships.
The Novel Approach
The patented methodology offers a decisive break from these constraints by optimizing reaction kinetics and reagent stoichiometry. By utilizing piperonyl cyclonene in a Blanc reaction at 15 to 30°C, the process avoids harsh conditions while achieving yields above 85 percent. The synthesis of the key intermediate, 1-(2-pyrimidyl)piperazine, is refined by controlling the feed ratio of piperazine to 2-chloropyrimide at 1:2.7 to 7, effectively suppressing double alkylation side reactions. This strategic adjustment boosts intermediate yields to over 90 percent. The final N-alkylation step replaces toxic xylene with lower boiling point solvents like ethanol or isopropanol, facilitating easier solvent recovery and recycling. This holistic optimization ensures cost reduction in API manufacturing while maintaining stringent environmental compliance standards.
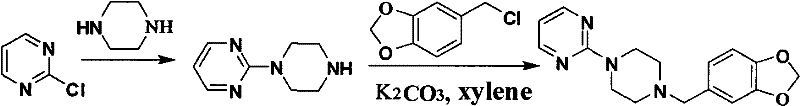
Mechanistic Insights into N-Alkylation and Blanc Reaction
The core of this synthesis lies in the precise control of nucleophilic substitution and electrophilic aromatic substitution mechanisms. In the first step, the Blanc reaction facilitates the chloromethylation of piperonyl cyclonene. The reaction mechanism involves the generation of a chloromethyl carbocation intermediate from paraformaldehyde and concentrated hydrochloric acid, which then attacks the electron-rich aromatic ring. Maintaining the temperature between 15 to 30°C is critical to prevent polymerization of the formaldehyde and ensure selective mono-chloromethylation. The molar ratio of piperonyl cyclonene to paraformaldehyde to hydrochloric acid is strictly controlled at 1:1.4 to 1.8:2.5 to 5, ensuring complete conversion while minimizing waste. This step establishes the electrophilic center required for the final coupling, demonstrating how fundamental organic transformations can be tuned for industrial efficiency.
Impurity control is paramount in the subsequent alkylation steps, particularly during the formation of 1-(2-pyrimidyl)piperazine. The mechanism involves the nucleophilic attack of piperazine on the 2-chloropyrimide. A common pitfall in this reaction is the formation of bis-alkylated byproducts where both nitrogen atoms of the piperazine ring react. The patent mitigates this by employing a slow addition technique, dripping the 2-chloropyrimide solution into an excess of piperazine over 2 to 5 hours. This maintains a high local concentration of the nucleophile, favoring mono-alkylation. The use of solvents like water, acetone, or alcohols further modulates the reaction polarity, enhancing selectivity. The resulting intermediate, shown below, is isolated as a faint yellow oily substance with high purity, ready for the final coupling without extensive chromatographic purification.
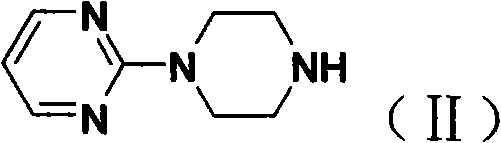
How to Synthesize Piribedil Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and safety. The process is divided into three distinct stages: preparation of piperonyl chloride, synthesis of the pyrimidine-piperazine intermediate, and the final coupling reaction. Each stage demands precise temperature control and stoichiometric balance to prevent side reactions. The use of triethylamine as a catalyst in the final step is particularly effective in scavenging hydrochloric acid byproducts, driving the equilibrium towards product formation. Detailed standard operating procedures are essential for reproducibility at scale. The following guide outlines the critical steps for technical teams to replicate this high-efficiency pathway in a GMP-compliant environment.
- Prepare piperonyl chloride via Blanc reaction using piperonyl cyclonene, paraformaldehyde, and concentrated hydrochloric acid at 15-30°C.
- Synthesize 1-(2-pyrimidyl)piperazine by reacting 2-chloropyrimide with excess piperazine in alcohol solvents at 40-60°C to minimize double alkylation.
- Perform final N-alkylation between piperonyl chloride and 1-(2-pyrimidyl)piperazine using triethylamine catalysis in ethanol or isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented route offers substantial strategic benefits beyond mere technical metrics. The elimination of high-pressure hydrogenation and expensive transition metal catalysts directly translates to reduced capital expenditure and lower operating costs. By avoiding solvents like DMF and xylene, which are increasingly regulated due to toxicity, manufacturers can streamline waste treatment processes and reduce environmental compliance burdens. The use of readily available raw materials like piperidine and 2-chloropyrimide ensures supply continuity, mitigating the risk of shortages that often plague specialty chemical markets. This stability is crucial for maintaining consistent production schedules and meeting the demanding lead times of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by removing the need for precious metal catalysts such as ruthenium, which are subject to volatile market pricing. Additionally, the replacement of high-boiling toxic solvents with recoverable alcohols like ethanol reduces solvent purchase and disposal expenses. The high yield of intermediates minimizes raw material waste, ensuring that every kilogram of input contributes effectively to the final output. These cumulative efficiencies result in substantial cost savings without compromising product quality, making it an economically superior choice for large-scale production.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals that are widely available from multiple global vendors, reducing dependency on single-source suppliers. The moderate reaction conditions eliminate the need for specialized high-pressure reactors, allowing production to be flexible across various manufacturing sites. This flexibility ensures that supply disruptions at one facility can be quickly mitigated by shifting production to another. Furthermore, the simplified purification steps reduce the overall cycle time, enabling faster turnaround from raw material intake to finished goods delivery.
- Scalability and Environmental Compliance: Scalability is inherent in the design, as the reaction exotherms are manageable and do not require complex cooling systems typically needed for cryogenic or high-temperature processes. The reduction in hazardous waste generation aligns with modern green chemistry principles, facilitating easier permitting and regulatory approval in strict jurisdictions. The ability to recycle solvents further diminishes the environmental footprint, supporting corporate sustainability goals. This makes the technology not only commercially viable but also environmentally responsible for long-term operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Piribedil synthesis technology. These answers are derived directly from the patent data and practical manufacturing considerations. They provide clarity on yield expectations, purity standards, and operational requirements. Understanding these aspects is vital for R&D directors and procurement specialists evaluating the feasibility of adopting this route for their supply chains.
Q: How does this patent improve upon conventional Piribedil synthesis routes?
A: The patent overcomes limitations of prior art by avoiding high-pressure hydrogenation and expensive ruthenium catalysts. It utilizes a low-temperature Blanc reaction and optimized alkylation conditions to achieve yields exceeding 90% for intermediates and overall yields of 45-75%, significantly reducing operational complexity and raw material costs compared to Route 1 and Route 2.
Q: What purity levels can be achieved with this manufacturing process?
A: The described method ensures exceptional product quality with HPLC purity content not lower than 99.8%. The simplified purification process, involving recrystallization from ethanol or isopropanol, effectively removes impurities without requiring complex chromatography, making it highly suitable for stringent pharmaceutical grade requirements.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It operates at moderate temperatures (20-150°C) without high pressure, uses low-toxicity solvents like ethanol instead of DMF or xylene, and employs readily available raw materials like piperidine and 2-chloropyrimide, ensuring stable quality and supply continuity for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piribedil Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies into commercial reality. Our CDMO expertise allows us to adapt routes like CN101735201A to meet specific client needs, ensuring seamless technology transfer. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision. Our facilities are equipped with rigorous QC labs to verify stringent purity specifications, ensuring every batch meets the 99.8 percent purity benchmark required for pharmaceutical applications. Partnering with us means securing a supply chain that is both robust and compliant with international standards.
We invite you to explore how this optimized synthesis can enhance your product portfolio and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediates supplier dedicated to driving innovation and efficiency in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
