Advanced Manufacturing Protocol for High-Purity Piribedil API Intermediate via Optimized Alkylation
Introduction to Advanced Piribedil Manufacturing Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for dopamine agonists like Piribedil, a critical active pharmaceutical ingredient used in treating Parkinson's disease. Patent CN101735201B introduces a transformative preparation method that addresses longstanding inefficiencies in existing manufacturing protocols by utilizing piperonyl butoxide derivatives and optimized alkylation strategies. This technical breakthrough enables the production of high-purity intermediates through a sequence that eliminates the need for high-pressure hydrogenation or precious metal catalysts, thereby streamlining the supply chain for global API manufacturers. The structural integrity of the final product is maintained through mild reaction conditions that preserve the sensitive heterocyclic moieties essential for biological activity. 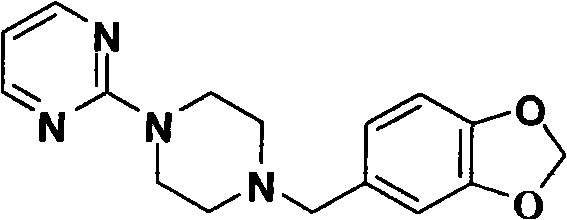 As a reliable pharmaceutical intermediate supplier, understanding these nuanced process improvements is vital for ensuring consistent quality and regulatory compliance in large-scale production environments.
As a reliable pharmaceutical intermediate supplier, understanding these nuanced process improvements is vital for ensuring consistent quality and regulatory compliance in large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Piribedil has been plagued by significant operational hurdles that impede efficient commercial scale-up of complex pharmaceutical intermediates. Conventional Route 1 necessitates reductive amination under high hydrogen pressures ranging from 2 to 60 bar, imposing severe safety constraints and requiring specialized high-pressure reactor infrastructure that increases capital expenditure. Furthermore, alternative pathways such as Route 2 rely heavily on expensive ruthenium catalysts like [Ru(p-cymene)Cl2]2, which not only inflate raw material costs but also introduce complex purification challenges to remove trace heavy metals from the final API. Other methods suffer from poor atom economy and low yields, with some literature reporting total yields as low as 32.4% due to the formation of persistent di-alkylated byproducts that are difficult to separate. The reliance on toxic, high-boiling solvents like xylene or DMF in these legacy processes further complicates waste management and solvent recovery, creating substantial environmental liabilities for manufacturing facilities.
The Novel Approach
The innovative methodology outlined in the patent data presents a paradigm shift by decoupling the synthesis into three distinct, highly controlled stages that maximize yield while minimizing hazard. 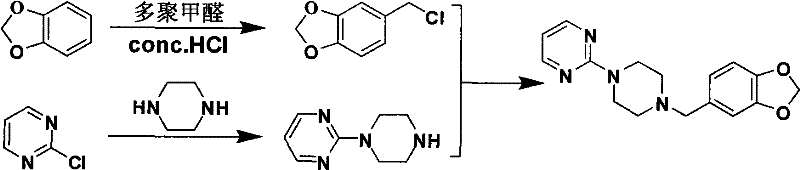 This novel approach initiates with a Blanc chloromethylation of the piperonyl ring under mild acidic conditions, bypassing the need for hazardous chlorinating agents typically used in older protocols. The subsequent mono-alkylation of piperazine is meticulously managed through controlled dropwise addition of 2-chloropyrimidine, a technique that kinetically favors the formation of the desired mono-substituted intermediate over unwanted di-substituted impurities. Finally, the coupling reaction utilizes inexpensive organic bases in low-toxicity alcohol solvents, facilitating easy product isolation through simple crystallization rather than complex chromatography. This strategic redesign results in a total yield improvement to between 45% and 75%, representing a substantial cost reduction in API manufacturing while ensuring the process remains suitable for industrial production without specialized high-pressure equipment.
This novel approach initiates with a Blanc chloromethylation of the piperonyl ring under mild acidic conditions, bypassing the need for hazardous chlorinating agents typically used in older protocols. The subsequent mono-alkylation of piperazine is meticulously managed through controlled dropwise addition of 2-chloropyrimidine, a technique that kinetically favors the formation of the desired mono-substituted intermediate over unwanted di-substituted impurities. Finally, the coupling reaction utilizes inexpensive organic bases in low-toxicity alcohol solvents, facilitating easy product isolation through simple crystallization rather than complex chromatography. This strategic redesign results in a total yield improvement to between 45% and 75%, representing a substantial cost reduction in API manufacturing while ensuring the process remains suitable for industrial production without specialized high-pressure equipment.
Mechanistic Insights into Optimized N-Alkylation and Chloromethylation
The core chemical innovation lies in the precise control of nucleophilic substitution dynamics during the formation of the 1-(2-pyrimidyl)piperazine intermediate. By dissolving piperazine and 2-chloropyrimidine separately and maintaining the reaction temperature between 40°C and 60°C, the process ensures that the nucleophilic attack occurs selectively at the primary amine nitrogen. The slow dropwise addition of the electrophile (2-chloropyrimidine) into the excess piperazine solution creates a concentration gradient that statistically minimizes the probability of a second alkylation event on the same piperazine molecule. 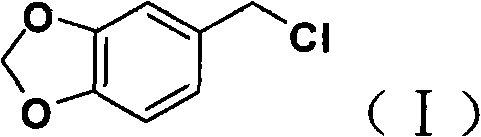 This kinetic control is critical because di-alkylated byproducts possess similar polarity to the target molecule, making their removal via standard purification techniques energetically and economically prohibitive. Furthermore, the initial Blanc reaction leverages the electron-rich nature of the piperonyl ring to facilitate electrophilic aromatic substitution with formaldehyde and hydrochloric acid, generating the reactive benzyl chloride species in situ with high efficiency. The use of concentrated hydrochloric acid serves a dual purpose as both a catalyst and a source of chloride ions, driving the equilibrium towards the chloromethylated product without generating excessive hazardous waste streams associated with thionyl chloride methods.
This kinetic control is critical because di-alkylated byproducts possess similar polarity to the target molecule, making their removal via standard purification techniques energetically and economically prohibitive. Furthermore, the initial Blanc reaction leverages the electron-rich nature of the piperonyl ring to facilitate electrophilic aromatic substitution with formaldehyde and hydrochloric acid, generating the reactive benzyl chloride species in situ with high efficiency. The use of concentrated hydrochloric acid serves a dual purpose as both a catalyst and a source of chloride ions, driving the equilibrium towards the chloromethylated product without generating excessive hazardous waste streams associated with thionyl chloride methods.
Impurity control is further enhanced by the selection of solvent systems that promote selective precipitation of byproducts during the workup phase. In the final N-alkylation step, the use of triethylamine as a base scavenges the hydrochloric acid byproduct effectively, preventing the protonation of the piperazine nitrogen which would otherwise deactivate the nucleophile and stall the reaction. The choice of ethanol or isopropanol as solvents allows for the differential solubility of the final Piribedil product versus unreacted starting materials, enabling a purification strategy based on slurry washing and recrystallization rather than column chromatography. This mechanistic understanding ensures that the HPLC purity of the final product consistently exceeds 99.8%, meeting the stringent requirements for high-purity pharmaceutical intermediates destined for human therapeutic use. The absence of transition metals in the final steps guarantees that the heavy metal profile of the API remains well within ICH Q3D guidelines, reducing the burden on downstream quality control laboratories.
How to Synthesize Piribedil Efficiently
The synthesis protocol described herein offers a streamlined pathway for producing Piribedil that balances chemical efficiency with operational simplicity, making it ideal for both pilot and commercial scales. The process begins with the preparation of piperonyl chloride, followed by the synthesis of the pyrimidyl-piperazine core, and concludes with the coupling of these two fragments. Detailed standard operating procedures regarding specific molar ratios, stirring rates, and crystallization parameters are essential for replicating the high yields reported in the patent data.
- Prepare piperonyl chloride via Blanc reaction using piperonyl ring, paraformaldehyde, and concentrated hydrochloric acid at 15-30°C.
- Synthesize 1-(2-pyrimidyl)piperazine by slowly adding 2-chloropyrimidine to a piperazine solution at 40-60°C to suppress di-alkylation byproducts.
- Perform final N-alkylation between piperonyl chloride and 1-(2-pyrimidyl)piperazine using triethylamine catalyst in ethanol or isopropanol at 50°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this manufacturing route offers significant advantages by fundamentally altering the cost structure of Piribedil production through raw material optimization. The elimination of expensive ruthenium catalysts and high-pressure hydrogenation equipment translates directly into reduced capital expenditure and lower operating costs per kilogram of finished product. By utilizing commodity chemicals such as piperonyl ring, paraformaldehyde, and piperazine, the supply chain becomes more resilient to fluctuations in the pricing of specialty reagents, ensuring stable availability for long-term contracts. The shift away from toxic solvents like xylene to greener alternatives like ethanol simplifies regulatory compliance and reduces the costs associated with hazardous waste disposal and solvent recovery systems. These factors collectively contribute to a more sustainable and economically viable manufacturing model that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by replacing precious metal catalysts with inexpensive organic bases and avoiding energy-intensive high-pressure reactions. The improved yield profile means less raw material is wasted on byproducts, effectively lowering the cost of goods sold (COGS) for the final API. Additionally, the ability to recover and reuse excess piperazine from the aqueous layer further enhances the overall material efficiency of the process. This economic efficiency allows manufacturers to offer more competitive pricing structures to downstream pharmaceutical clients without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key starting materials are bulk commodities with established global supply networks, reducing the risk of single-source bottlenecks. The mild reaction conditions allow for production in standard glass-lined or stainless steel reactors, increasing the number of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This flexibility ensures continuity of supply even during regional disruptions, as the technology can be easily transferred between different manufacturing sites. The robustness of the process against minor variations in temperature or mixing rates further guarantees consistent batch-to-batch quality.
- Scalability and Environmental Compliance: The absence of high-pressure steps and the use of low-boiling, recyclable solvents make this process inherently safer and easier to scale from laboratory to multi-ton production. Environmental compliance is significantly improved due to the reduction in volatile organic compound (VOC) emissions and the elimination of heavy metal contaminants from the waste stream. The simplified purification workflow reduces water consumption and energy usage associated with extensive chromatographic separations. These attributes position the technology as a preferred choice for manufacturers aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method for Piribedil. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to assist technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does this patent improve the yield compared to conventional Route 5?
A: The patent improves total yield from 32.4% in conventional Route 5 to between 45% and 75% by optimizing the molar ratio of piperazine to 2-chloropyrimidine and controlling the addition rate to minimize di-alkylation byproducts.
Q: What are the safety advantages of the solvents used in this new method?
A: Unlike conventional methods that utilize toxic high-boiling solvents like xylene or DMF, this process employs lower toxicity solvents such as ethanol and isopropanol, which are easier to recover and recycle, significantly reducing environmental hazards.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, this method avoids the use of expensive ruthenium catalysts required in other routes (like Route 2), relying instead on inexpensive inorganic or organic bases like triethylamine, which drastically lowers raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piribedil Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to transform complex patent methodologies into robust, commercial-grade manufacturing processes for our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Piribedil meets or exceeds the 99.8% purity benchmark established in the patent. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, providing peace of mind for regulatory filings and patient safety.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior manufacturing protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality pharmaceutical intermediates reliably. Let us collaborate to enhance your supply chain efficiency and drive innovation in your drug development pipeline.
