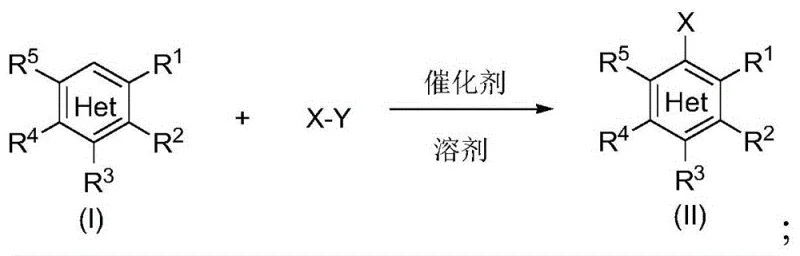
Revolutionizing Aryl Halide Production with Mild Organocatalytic Halogenation Technology
The chemical industry is constantly seeking more efficient and sustainable methods for synthesizing critical building blocks, and the recent disclosure in patent CN112573978B presents a transformative approach to aryl halide production. This technology addresses the long-standing challenges associated with traditional halogenation by utilizing organocatalysts such as sulfoxides and nitrogen oxides to activate halogenating agents. For R&D directors and process chemists, this represents a significant leap forward in achieving high-purity pharmaceutical intermediates under remarkably mild conditions. The method eliminates the need for hazardous elemental halogens while delivering superior regioselectivity and yield, positioning it as a cornerstone technique for modern fine chemical synthesis.
Furthermore, the versatility of this catalytic system allows for the functionalization of a broad spectrum of aromatic substrates, ranging from simple benzene derivatives to complex heterocyclic systems found in active pharmaceutical ingredients. By leveraging the unique electronic properties of catalysts like dimethyl sulfoxide (DMSO) or TEMPO salts, the reaction proceeds with exceptional efficiency at ambient temperatures. This capability is crucial for maintaining the integrity of sensitive functional groups that might otherwise decompose under the harsh thermal or acidic conditions typically required by legacy halogenation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl halides has relied heavily on the use of elemental halogens such as chlorine or bromine gas, which pose severe safety risks due to their high toxicity, corrosivity, and volatility. Even when safer N-halo reagents like N-chlorosuccinimide (NCS) are employed, they often exhibit insufficient reactivity towards electron-rich or sterically hindered aromatic rings without the aid of strong activators. Traditional activation strategies frequently involve stoichiometric amounts of Lewis acids like iron(III) chloride or phosphorus-based reagents, which can lead to significant waste generation and complicate downstream purification processes. Moreover, these conventional methods often suffer from poor selectivity, resulting in poly-halogenated byproducts that are difficult to separate and reduce the overall atom economy of the synthesis.
The Novel Approach
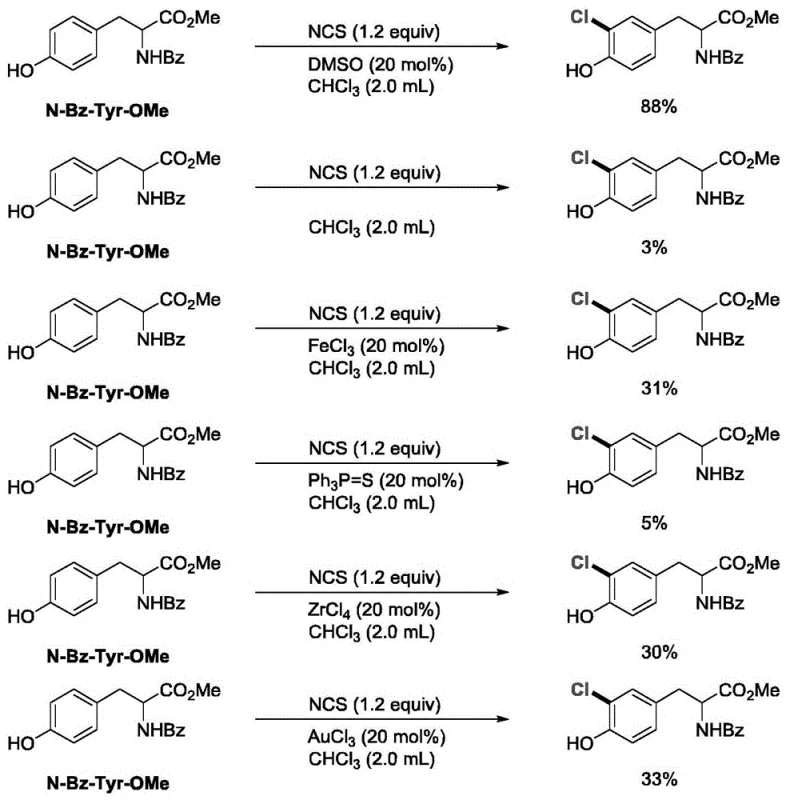
In stark contrast, the methodology described in CN112573978B utilizes catalytic amounts of sulfoxides or nitrogen oxides to dramatically enhance the electrophilicity of the halogen source, enabling rapid reaction kinetics at room temperature. Experimental data within the patent highlights the superiority of this approach, showing that DMSO-catalyzed chlorination of substrates like N-Bz-Tyr-OMe achieves yields of 86%, whereas traditional catalysts like FeCl3 or Ph3P=S struggle to reach even 33% under similar conditions. This substantial improvement in efficiency not only reduces raw material consumption but also simplifies the workup procedure, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The ability to achieve such high conversions with minimal catalyst loading underscores the robustness and practical utility of this novel organocatalytic strategy.
Mechanistic Insights into Sulfoxide and N-Oxide Catalyzed Halogenation
The core mechanism driving this high-efficiency transformation involves the coordination of the oxygen atom in the sulfoxide or nitrogen oxide catalyst with the halogen atom of the N-halo reagent. This interaction polarizes the nitrogen-halogen bond, effectively increasing the positive charge density on the halogen and transforming it into a potent electrophile capable of attacking the aromatic ring. Unlike metal-based Lewis acids which coordinate with the substrate, this organocatalytic mode of action activates the reagent directly, thereby minimizing side reactions such as substrate oxidation or polymerization that are common with transition metals. The catalytic cycle is regenerated efficiently, allowing for turnover numbers that make the process economically viable for large-scale operations while maintaining stringent purity specifications required for GMP production environments.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in suppressing the formation of undesired byproducts. Traditional harsh conditions often promote over-halogenation or degradation of labile protecting groups, leading to complex impurity profiles that require extensive chromatographic purification. By operating at temperatures as low as 25°C and utilizing neutral or weakly basic catalytic species, this new method preserves the structural fidelity of complex molecules like indoles, pyrazoles, and fused heterocycles. This enhanced selectivity ensures that the final aryl halide product meets the rigorous quality standards demanded by regulatory bodies, reducing the burden on quality control labs and accelerating the timeline from bench-scale discovery to commercial supply chain integration.
How to Synthesize Aryl Halides Efficiently
The implementation of this synthetic route is straightforward and relies on standard laboratory equipment, making it accessible for both research and production settings. The process typically involves dissolving the aromatic substrate and the N-halo reagent in a common organic solvent such as chloroform or dichloromethane, followed by the addition of a catalytic quantity of DMSO or a specific N-oxide derivative. The reaction mixture is then stirred at ambient temperature for a period ranging from 2 to 12 hours, depending on the electronic nature of the substrate, until full conversion is observed. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield across different batches.
- Prepare the reaction mixture by combining the aromatic substrate, N-halosuccinimide (NCS/NBS/NIS), and a catalytic amount of dimethyl sulfoxide (DMSO) or a nitrogen oxide derivative in a suitable solvent like chloroform.
- Stir the reaction mixture at mild temperatures ranging from 0°C to 25°C for a duration of 2 to 12 hours, monitoring progress via TLC or HPLC to ensure complete conversion.
- Upon completion, remove the solvent via rotary evaporation and purify the resulting aryl halide product using standard column chromatography techniques to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic halogenation technology offers compelling strategic benefits that extend beyond mere chemical efficiency. The elimination of hazardous elemental halogens and corrosive metal salts significantly reduces the safety risks associated with storage and handling, thereby lowering insurance costs and simplifying regulatory compliance regarding hazardous waste disposal. Furthermore, the use of inexpensive and widely available catalysts like DMSO ensures a stable supply chain for raw materials, mitigating the risk of production delays caused by the scarcity of specialized reagents. This reliability is essential for maintaining continuous manufacturing operations and meeting the tight delivery schedules expected by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The high catalytic efficiency and exceptional yields demonstrated in the patent data translate directly into lower cost of goods sold by maximizing the output per unit of raw material input. By avoiding the need for expensive transition metal catalysts and the subsequent costly removal steps required to meet residual metal limits in APIs, manufacturers can achieve substantial cost savings. Additionally, the simplified purification process reduces solvent consumption and energy usage during distillation and drying, further enhancing the overall economic viability of the production process.
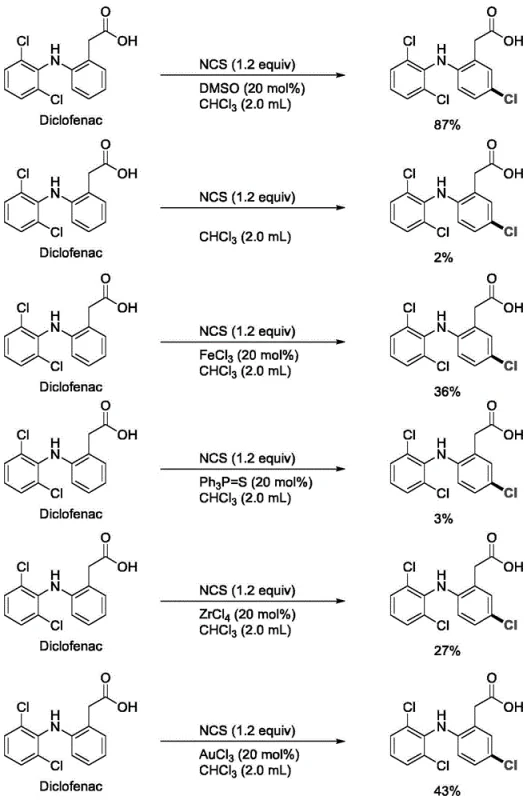
- Enhanced Supply Chain Reliability: The robustness of this method across a wide variety of substrates, including complex drug molecules like Diclofenac and Naproxen, ensures consistent product quality and supply continuity. The mild reaction conditions minimize the risk of batch failures due to thermal runaway or equipment corrosion, which are common pitfalls in traditional halogenation processes. This operational stability allows supply chain planners to forecast production capacities with greater accuracy and respond more agilely to fluctuations in market demand for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards associated with elemental halogen addition, making it safer for large reactor vessels. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations and corporate sustainability goals, enhancing the company's reputation as a responsible manufacturer. This green chemistry profile not only future-proofs the manufacturing asset but also appeals to environmentally conscious partners seeking to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced halogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation to provide clarity for potential partners and stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of using DMSO as a catalyst for halogenation compared to traditional Lewis acids?
A: According to patent CN112573978B, DMSO catalysis offers significantly higher yields (up to 87% vs 43% with FeCl3) and operates under much milder conditions, avoiding the corrosive nature and harsh handling requirements of traditional metal-based activators.
Q: Is this halogenation method compatible with sensitive functional groups found in API intermediates?
A: Yes, the method demonstrates excellent chemoselectivity, successfully halogenating complex molecules like Diclofenac, Naproxen, and Clopidogrel without degrading sensitive ester, amide, or ether functionalities present in the structure.
Q: Can this process be scaled for commercial manufacturing of pharmaceutical intermediates?
A: The process utilizes inexpensive, commercially available reagents like NCS and DMSO in common solvents like chloroform, making it highly amenable to scale-up for cost reduction in pharmaceutical intermediate manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic halogenation technology in advancing the synthesis of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aryl halide delivered meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project, optimizing your supply chain for both performance and cost-efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this technology can enhance your production capabilities and reduce lead time for high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
