Advanced Capecitabine Synthesis via Selective Amidation for Commercial Scale-up
Advanced Capecitabine Synthesis via Selective Amidation for Commercial Scale-up
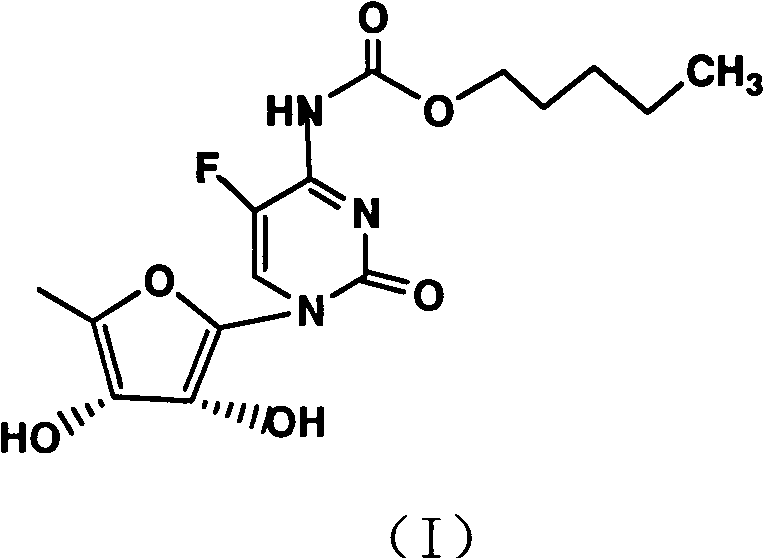
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for the production of critical oncology agents like Capecitabine. Patent CN101928314A discloses a groundbreaking preparation method that fundamentally reorders the synthetic sequence to maximize efficiency and purity. Unlike conventional routes that often struggle with low yields and complex purification due to unprotected reactive sites, this innovation introduces a strategic amidation step at the very beginning of the synthesis. By reacting 5-fluorocytosine with pentyl haloformate prior to glycosylation, the process effectively masks the reactive amino hydrogen, thereby preventing unwanted side reactions during subsequent silylation and coupling stages. This technical breakthrough not only elevates the total yield to an impressive 62-68% but also ensures a final purity exceeding 99.7%, addressing the stringent quality requirements of global regulatory bodies.
For procurement managers and supply chain directors, the implications of this patented technology are profound. The shift from expensive cytidine-based starting materials to readily available 5-fluorocytosine derivatives represents a significant optimization in raw material sourcing. Furthermore, the elimination of heavy metal catalysts, which are prevalent in older coupling methods, removes the need for costly and time-consuming metal scavenging steps. This streamlined approach translates directly into a more reliable capecitabine supplier capability, ensuring consistent batch-to-batch quality while simultaneously driving down the cost reduction in API manufacturing. The method's reliance on standard industrial solvents and mild reaction conditions further enhances its suitability for large-scale commercial production, mitigating supply chain risks associated with specialized reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for Capecitabine have long been plagued by inherent inefficiencies that hinder commercial viability. Historical methods, such as those described in US4966891, rely on 5-fluorouracil cytidine as a starting material, which is not only prohibitively expensive but also limits the scalability of the process due to supply constraints. Alternative routes involving furanoside hydroxyl protection followed by heavy metal-mediated coupling, as seen in US5453497, introduce significant environmental and safety liabilities. The use of heavy metal coupling agents necessitates rigorous downstream purification to meet residual metal specifications, adding substantial operational complexity and waste generation. Moreover, these conventional processes often suffer from poor stereocontrol, leading to the formation of difficult-to-separate anomers and reducing the overall yield to a mere 22-53%. The reliance on harsh mineral alkalis like sodium hydroxide for the final deprotection step can also compromise the configurational integrity of the molecule, resulting in lower purity profiles that require extensive recrystallization.
The Novel Approach
The methodology outlined in CN101928314A offers a decisive break from these legacy constraints by implementing a protective group strategy at the earliest possible stage. By converting 5-fluorocytosine into its amide derivative (Formula II) using pentyl haloformate, the process creates a robust intermediate that withstands the rigors of subsequent silylation and glycosylation. This "amidation-first" logic effectively neutralizes the nucleophilic amino group, preventing it from interfering with the silylation of the ring nitrogen. Consequently, the consumption of hexamethyldisilazane (HMDS) is drastically minimized, as the reagent is no longer wasted on side reactions with the exocyclic amine. The subsequent glycosylation with B-acetyl furan glucosides proceeds with higher fidelity, yielding the diacetylated intermediate (Formula IV) in superior quantities. Finally, the adoption of a mild ammonia/methanol hydrolysis system replaces corrosive mineral bases, preserving the chiral center and simplifying the workup procedure to a straightforward extraction and crystallization.
Mechanistic Insights into Selective Amidation and Glycosylation
The core chemical innovation lies in the precise manipulation of reactivity on the pyrimidine ring. In the initial step, 5-fluorocytosine reacts with pentyl chloroformate or fluoroformate at elevated temperatures of 100-110°C. This reaction selectively targets the exocyclic amino group to form a urethane-like linkage, generating the 5-fluorocytosine amide derivative. This structural modification is critical because it reduces the electron density on the ring nitrogen, facilitating a cleaner silylation reaction in the subsequent step. When treated with hexamethyldisilazane and trimethylchlorosilane, the silyl group attaches exclusively to the ring nitrogen without competing N-silylation of the exocyclic amine, which would otherwise lead to decomposition or polymerization. This controlled activation sets the stage for a highly efficient Vorbrüggen-type glycosylation.
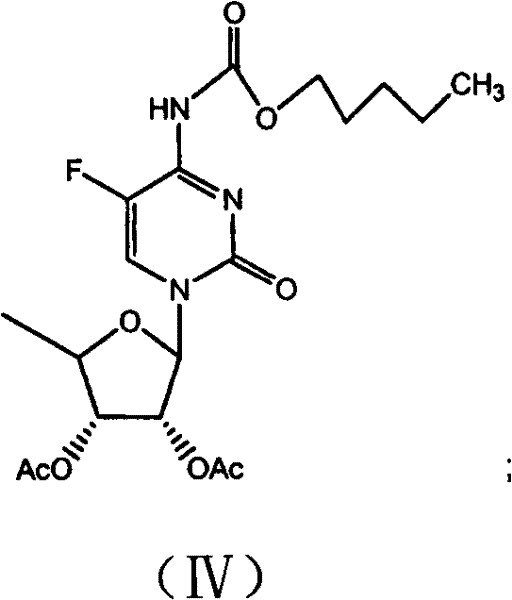
Following silylation, the activated cytosine species undergoes coupling with B-acetyl furan glucosides in an inert solvent such as acetonitrile or toluene. The presence of the amide protecting group ensures that the glycosidic bond forms with high regioselectivity at the N1 position of the pyrimidine ring. The reaction is typically conducted at 60-80°C, promoting the formation of the beta-anomer while suppressing the alpha-isomer. The resulting diacetylated cytidine amide derivative (Formula IV) is a stable crystalline solid that can be isolated or carried forward crude, demonstrating the robustness of the intermediate. The final hydrolysis step utilizes ammonia gas dissolved in methanol at low temperatures (0-5°C). This mild basic condition selectively cleaves the acetyl esters on the sugar moiety and the amide protecting group on the base without epimerizing the chiral centers on the ribose ring. This mechanistic finesse is what allows the process to achieve purity levels greater than 99.7% with minimal byproduct formation.
How to Synthesize Capecitabine Efficiently
The synthesis of Capecitabine via this patented route is designed for operational simplicity and high throughput. The process begins with the thermal reaction of 5-fluorocytosine and pentyl haloformate, followed by filtration and washing to isolate the pure amide intermediate. This solid is then dissolved in an inert solvent and treated with silylating agents under nitrogen to generate the reactive silyl ether in situ. Without isolation, the sugar donor is added to effect the coupling, and the mixture is worked up via pH adjustment and extraction. The final deprotection is a homogeneous reaction in methanol, followed by solvent removal and recrystallization from ethyl acetate. For detailed standard operating procedures and specific molar ratios, please refer to the technical guide below.
- React 5-fluorocytosine with pentyl haloformate at 100-110°C to form the protected amide derivative.
- Treat the amide derivative with hexamethyldisilazane (HMDS) and trimethylchlorosilane under nitrogen protection.
- Couple the silylated intermediate with B-acetyl furan glucosides in inert solvent at 60-80°C.
- Hydrolyze the diacetylated intermediate using ammonia in methanol at 0-5°C to obtain the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages that directly impact the bottom line and supply security. The primary driver of value is the substantial reduction in raw material costs achieved by eliminating the need for expensive cytidine starters and minimizing the usage of hexamethyldisilazane. By optimizing the stoichiometry and preventing side reactions, the process ensures that every kilogram of reagent contributes to the final product, thereby enhancing overall atom economy. This efficiency translates into a more competitive pricing structure for the final API, allowing manufacturers to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on quality standards. Additionally, the simplified workup procedures reduce the demand for specialized labor and equipment maintenance, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and the reduction in HMDS consumption remove significant cost centers from the production budget. Traditional methods often require expensive scavengers to remove metal residues, a step that is entirely unnecessary in this metal-free protocol. Furthermore, the higher yield of 62-68% means that less starting material is required to produce the same amount of active ingredient, effectively lowering the cost of goods sold (COGS). The use of common industrial solvents like acetonitrile and ethyl acetate ensures that solvent recovery and recycling can be easily implemented, contributing to long-term financial sustainability.
- Enhanced Supply Chain Reliability: The reliance on 5-fluorocytosine, a commodity chemical with a stable global supply, mitigates the risk of raw material shortages that often plague cytidine-dependent routes. The robustness of the intermediates, particularly the amide derivative, allows for potential storage and batching flexibility, enabling manufacturers to build strategic inventory buffers. The simplified purification steps reduce the lead time for high-purity capecitabine batches, ensuring that delivery schedules can be met consistently even during periods of high market demand. This reliability is crucial for maintaining uninterrupted production lines for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily replicated in multi-ton reactors without significant heat transfer or mixing issues. The replacement of harsh mineral alkalis with ammonia/methanol significantly reduces the generation of saline wastewater, aligning with modern green chemistry principles and reducing waste disposal costs. The absence of heavy metals simplifies environmental permitting and compliance reporting, making the facility more attractive to auditors and regulators. This environmental stewardship not only reduces liability but also enhances the brand reputation of the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield improvements, purity profiles, and operational benefits. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing supplier capabilities.
Q: How does the new amidation-first method improve yield compared to traditional routes?
A: The novel method increases total recovery to 62-68% from the traditional 22-53% by minimizing side reactions through early amino group protection.
Q: What are the advantages of using ammonia/methanol for hydrolysis?
A: Using ammonia/methanol saponification simplifies post-treatment significantly compared to harsh mineral alkalis, reducing processing difficulty and improving configurational stability.
Q: Does this process reduce the consumption of expensive reagents?
A: Yes, by performing amidation in advance, the consumption of hexamethyldisilazane (HMDS) during the alkylation step is drastically reduced, lowering overall material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the oncology market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented method are fully realized in a GMP environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Capecitabine meets the highest international standards. Our commitment to process excellence allows us to deliver high-purity capecitabine that is ready for immediate formulation, minimizing your time to market.
We invite you to collaborate with us to leverage this superior manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your project's economic and operational performance.
