Advanced Capecitabine Synthesis: Eliminating Toxic Reagents for Scalable API Manufacturing
Advanced Capecitabine Synthesis: Eliminating Toxic Reagents for Scalable API Manufacturing
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, safer, and more economically viable synthetic routes for critical oncology therapeutics. A pivotal development in this landscape is documented in patent CN102199180A, which discloses a novel preparation method for capecitabine, a widely used oral cytotoxic agent for treating breast and colorectal cancers. This technology fundamentally reengineers the N-4 carbamylation step, moving away from hazardous phosgene equivalents toward a sophisticated triazole-mediated activation strategy. For R&D directors and procurement leaders, this represents not merely a chemical optimization but a strategic supply chain advantage, offering a pathway to high-purity intermediates with drastically reduced environmental and safety liabilities. The ability to produce such complex nucleoside derivatives without relying on controlled toxic substances positions manufacturers to navigate regulatory landscapes more efficiently while ensuring consistent product quality.
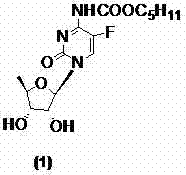
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of capecitabine has been plagued by significant safety and efficiency bottlenecks inherent to traditional carbamylation chemistries. As illustrated in prior art methodologies, the standard approach often relies on n-amyl chloroformate or triphosgene as the acylating agent to introduce the critical carbamate functionality at the N-4 position of the cytidine scaffold. These reagents are notoriously hazardous; triphosgene, for instance, can decompose to release phosgene gas, a chemical warfare agent requiring extreme containment measures and specialized ventilation infrastructure. Furthermore, alternative routes utilizing activated esters like pentyloxy formyl radical p-nitrophenyl phenolic ester introduce downstream processing nightmares, often necessitating silica gel chromatography for purification. Such purification steps are economically prohibitive at a commercial scale, leading to substantial solvent consumption, extended batch cycles, and increased operational expenditures that erode profit margins for generic API manufacturers.
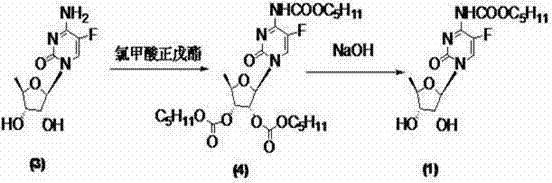
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a transformative solution by employing 1,1'-carbonyl di(1,2,4-triazole) as the key activating agent. This reagent functions as a highly efficient carbonyl source that reacts selectively with the amino group of the protected cytidine intermediate under mild conditions. The genius of this approach lies in its one-pot potential; the activated intermediate reacts directly with n-amyl alcohol without the need for isolation, streamlining the workflow significantly. By circumventing the use of chloro-formic esters and phosgene classes entirely, the process eliminates the generation of corrosive hydrochloric acid byproducts and toxic gas emissions. This results in a cleaner reaction profile where the crude product can often be purified through simple aqueous washing and recrystallization, thereby drastically reducing the environmental footprint and enhancing the overall safety profile of the manufacturing facility.
Mechanistic Insights into Triazole-Mediated N-4 Carbamylation
To fully appreciate the technical superiority of this route, one must delve into the mechanistic nuances of the triazole activation system. The reaction initiates with the nucleophilic attack of the N-4 amino group of the 5'-deoxy-5-fluorocytidine derivative onto the carbonyl carbon of the 1,1'-carbonyl di(1,2,4-triazole). This forms a highly reactive urea-type intermediate where the triazole ring acts as an excellent leaving group due to its electron-deficient nature and aromatic stability upon departure. Unlike traditional acyl chlorides which react violently and indiscriminately, this triazole-activated species offers superior chemoselectivity, ensuring that the sensitive hydroxyl groups on the ribose sugar ring remain intact without requiring excessive protecting group manipulation. The subsequent addition of n-amyl alcohol triggers a transesterification-like displacement, where the alcohol attacks the activated carbonyl, displacing the triazole moiety and establishing the stable N-4 amyl carbamate bond essential for the drug's bioactivation pathway in vivo.
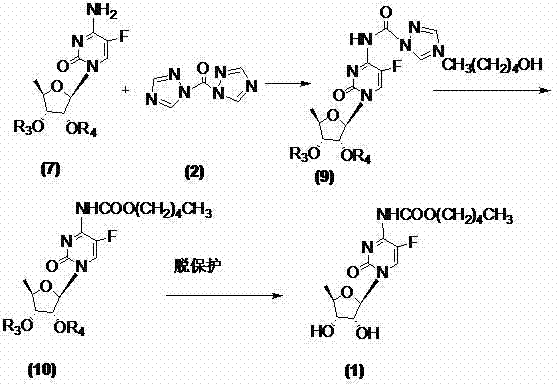
Beyond the primary coupling mechanism, the impurity control profile of this synthesis is exceptionally robust, addressing a primary concern for R&D directors focused on regulatory compliance. In conventional phosgene-based routes, over-acylation of the sugar hydroxyls or formation of symmetrical urea byproducts are common persistent impurities that are difficult to purge. The triazole method, operating typically at temperatures between 0°C and 40°C, kinetically favors the formation of the desired carbamate while minimizing thermal degradation of the fluorinated nucleoside core. Furthermore, the byproducts generated, primarily triazole derivatives, are generally more polar and water-soluble than the target lipophilic intermediate, facilitating their removal during the aqueous workup phase. This inherent selectivity translates directly into a higher crude purity, often exceeding 99.7% after simple recrystallization from ethyl acetate, thereby reducing the burden on analytical quality control teams and accelerating the release of batches for clinical or commercial use.
How to Synthesize Capecitabine Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and solvent selection to maximize yield and operational safety. The process begins by dissolving the protected cytidine starting material, such as 2',3'-di-O-acetyl-5'-deoxy-5-fluorocytidine, in an aprotic solvent like methylene dichloride or toluene. To this solution, 1,1'-carbonyl di(1,2,4-triazole) is added in a molar ratio ranging from 1:1.5 to 1:3.0 relative to the nucleoside, ensuring complete activation of the amino group. Following the formation of the activated intermediate, n-amyl alcohol is introduced to the reaction mixture, driving the carbamylation to completion. The detailed standardized synthetic steps, including specific temperature ramps, quenching protocols, and crystallization parameters, are outlined in the technical guide below for process engineers to review.
- Activate the N-4 position of protected 5'-deoxy-5-fluorocytidine using 1,1'-carbonyl di(1,2,4-triazole) in an aprotic solvent.
- React the activated intermediate directly with n-amyl alcohol to form the N-4 amyl carbamate derivative without isolation.
- Perform deprotection under alkaline conditions followed by recrystallization to achieve high-purity capecitabine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this triazole-based synthesis protocol offers compelling economic and logistical advantages that extend far beyond simple reagent substitution. The elimination of highly regulated toxic precursors like phosgene and chloroformates removes a significant layer of regulatory overhead, reducing the costs associated with hazardous material storage, transportation, and disposal. This simplification of the chemical inventory allows for more flexible sourcing strategies and mitigates the risk of supply disruptions caused by strict environmental regulations on toxic chemicals. Moreover, the streamlined workup procedure, which avoids energy-intensive chromatographic separations, significantly shortens the manufacturing cycle time. This increased throughput capacity enables suppliers to respond more agilely to market demand fluctuations, ensuring a more reliable and continuous supply of this critical oncology intermediate to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream processing train. By eliminating the need for silica gel chromatography and reducing the number of unit operations required for purification, manufacturers can achieve substantial savings in solvent consumption and labor costs. The high selectivity of the triazole reagent minimizes the formation of hard-to-remove impurities, which in turn reduces product loss during purification steps, leading to an improved overall mass balance. Additionally, the ability to use commercially available, stable reagents rather than custom-synthesized or highly controlled toxic agents stabilizes raw material pricing and reduces procurement volatility.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, non-hazardous reagents that are less susceptible to shipping restrictions and regulatory bans. Traditional phosgene-based routes often face logistical bottlenecks due to the specialized containment required for transport; the triazole method circumvents these issues entirely. The mild reaction conditions also reduce the strain on reactor equipment, lowering maintenance frequency and extending the operational lifespan of manufacturing assets. This reliability ensures that production schedules can be maintained consistently, reducing the risk of stockouts for downstream partners who depend on a steady flow of high-quality intermediates for their own formulation processes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a best-in-class approach for modern chemical manufacturing. The absence of toxic gas evolution simplifies the engineering controls required for scale-up, allowing for larger batch sizes without proportional increases in safety infrastructure investment. The waste stream generated is significantly less hazardous, facilitating easier treatment and disposal in compliance with increasingly stringent global environmental standards. This 'green chemistry' alignment not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly critical in vendor selection criteria for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this route for commercial production, as it highlights the specific operational parameters that drive success.
Q: Why is the triazole-based activation method safer than traditional phosgene routes?
A: Traditional methods utilize n-amyl chloroformate or triphosgene, which release toxic phosgene gas and require stringent safety containment. The triazole-based method employs solid, stable reagents that eliminate the risk of gas leakage and reduce hazardous waste generation.
Q: Does this novel synthesis route impact the purity profile of the final API?
A: Yes, positively. By avoiding harsh acylating agents and simplifying the workup to simple washing and recrystallization, the process minimizes side reactions and metal contamination, consistently achieving HPLC purity levels exceeding 99.7%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The reaction operates at mild temperatures (0-40°C) and uses common aprotic solvents like methylene dichloride. The elimination of chromatographic purification steps significantly enhances throughput and reduces production costs for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term viability of the pharmaceutical supply chain. Our CDMO division possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the triazole-activated synthesis of capecitabine are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle complex nucleoside analysis, guaranteeing that every batch meets the exacting standards required for global regulatory submissions. Our commitment to technical excellence ensures that we can deliver high-purity intermediates that facilitate smoother downstream API synthesis for our partners.
We invite you to collaborate with us to optimize your supply chain for oncology therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate how our advanced manufacturing capabilities can enhance your product's competitiveness in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
