Advanced Negishi Coupling Strategy for High-Purity Sitagliptin Intermediate Manufacturing
Advanced Negishi Coupling Strategy for High-Purity Sitagliptin Intermediate Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways to synthesize complex amino acid derivatives, particularly those serving as critical building blocks for integrin inhibitors. Patent CN111471003A, published on July 31, 2020, introduces a groundbreaking preparation method for a specific amino acid compound designated as formula (IV), which serves as a pivotal intermediate in the synthesis of Lifitegrast (also known as Sarilumab's companion small molecule or simply the integrin inhibitor marketed as Xiidra). This novel approach leverages transition metal catalysis to construct the carbon-carbon bond between an aromatic sulfone moiety and a chiral amino acid backbone, bypassing the limitations of classical substitution reactions. The technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to optimize their portfolio for ophthalmic and autoimmune therapeutic markets.
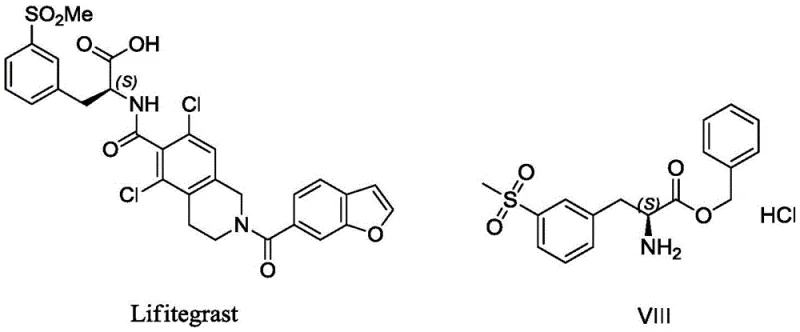
The target molecule, specifically when R1 is methyl and R2 is benzyl, corresponds to (S)-2-amino-3-methylsulfonyl-benzyl phenylpropionate hydrochloride (Compound VIII). This compound is not merely a laboratory curiosity but a commercially vital precursor for Lifitegrast, a drug approved by the FDA for treating dry eye disease. The ability to produce this intermediate with high stereochemical integrity and minimal impurities is paramount for downstream drug safety. The patent outlines a robust methodology that utilizes readily available starting materials, positioning it as a prime candidate for cost reduction in API manufacturing where raw material availability often dictates production timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Compound VIII relied heavily on a linear, multi-step sequence starting from L-3-bromophenylalanine. As illustrated in the prior art reaction scheme, this traditional pathway involves at least four distinct synthetic operations: protection of the amino group with a Boc anhydride, introduction of the methyl sulfone functionality via copper-catalyzed substitution, esterification with benzyl alcohol, and finally, deprotection of the Boc group. This lengthy sequence inherently accumulates impurities at each stage, leading to lower overall yields and requiring extensive purification efforts between steps. Furthermore, the introduction of the sulfone group onto a pre-existing amino acid scaffold often requires harsh conditions or expensive reagents, which complicates the commercial scale-up of complex pharmaceutical intermediates.
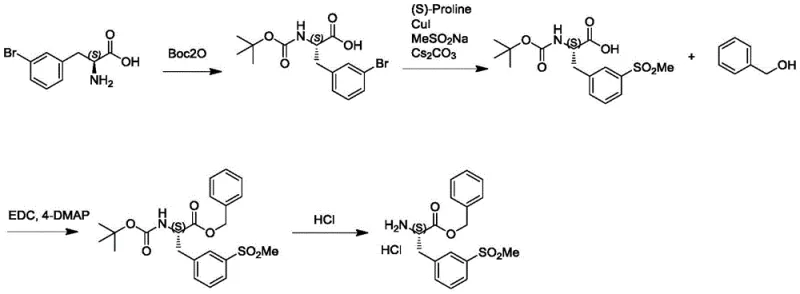
The reliance on L-3-bromophenylalanine as a starting material also presents supply chain vulnerabilities, as chiral halogenated amino acids can be cost-prohibitive and subject to availability fluctuations. Each additional step in the conventional route increases the consumption of solvents, reagents, and energy, directly impacting the environmental footprint and the final cost of goods sold (COGS). For procurement managers, this translates to higher purchase prices and longer lead times, as the cumulative time required for four separate reactions and workups significantly delays batch completion. The need to manage multiple hazardous waste streams from copper catalysis and various coupling agents further burdens the operational overhead of manufacturing facilities.
The Novel Approach
In stark contrast, the method disclosed in CN111471003A revolutionizes the synthesis by employing a convergent strategy centered on palladium-catalyzed Negishi coupling. Instead of modifying a completed amino acid, this approach constructs the core skeleton by coupling an aryl bromide (Compound I) with an organozinc reagent derived from an iodo-alanine derivative (Compound II). This strategic shift reduces the synthesis to effectively two main stages: the formation of the organozinc species and the subsequent cross-coupling, followed by a simple deprotection. By assembling the molecule from simpler, achiral or easily resolved fragments, the process achieves superior atom economy and operational simplicity. This streamlined workflow is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand.
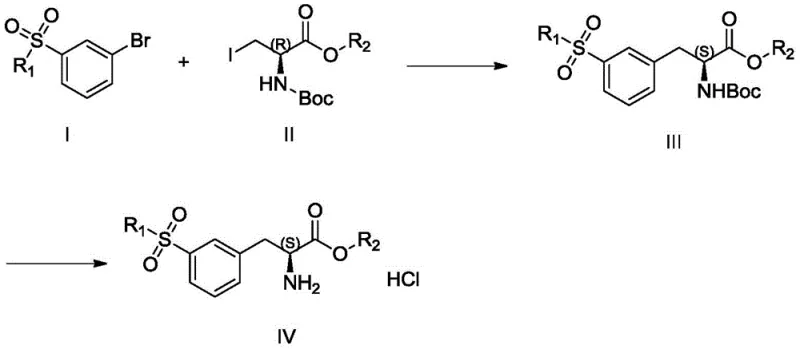
The versatility of this new route is underscored by the broad scope of substituents allowed for R1 and R2, ranging from simple alkyl groups like methyl and ethyl to more complex moieties like benzyl and cyclohexyl. This flexibility suggests that the platform technology can be adapted to synthesize a library of analogues, providing R&D teams with valuable tools for structure-activity relationship (SAR) studies without needing to develop entirely new synthetic routes for each variant. The use of standard palladium catalysts and common phosphine ligands ensures that the chemistry is transferable to existing manufacturing infrastructure without requiring specialized equipment, facilitating a smoother transition from bench-scale discovery to pilot plant production.
Mechanistic Insights into Pd-Catalyzed Negishi Coupling
The core of this technological advancement lies in the precise execution of the Negishi cross-coupling reaction. The process begins with the activation of metallic zinc powder in a polar aprotic solvent, typically N,N-dimethylacetamide (DMAC), often aided by a catalytic amount of methanesulfonic acid. This activation step is critical for generating the reactive organozinc species from N-Boc-3-iodo-L-alanine esters. Unlike Grignard reagents, organozinc compounds are milder and tolerate a wider range of functional groups, including the ester and protected amine functionalities present in the substrate. The subsequent transmetallation to the palladium center allows for the selective formation of the C(sp2)-C(sp3) bond with retention of configuration at the chiral alpha-carbon, a crucial factor for maintaining the biological activity of the final drug substance.
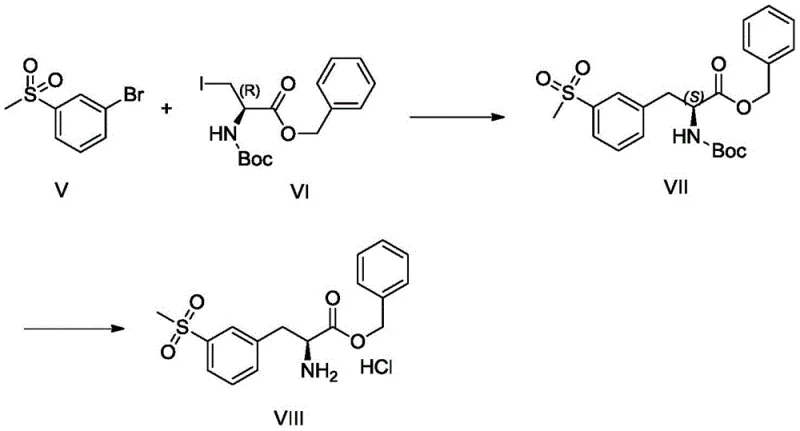
Impurity control is meticulously managed through the selection of ligands and reaction conditions. The patent highlights the efficacy of bulky phosphine ligands, such as tris(o-methylphenyl)phosphine, in conjunction with palladium sources like tris(dibenzylideneacetone)dipalladium (Pd2(dba)3). These ligands create a steric environment around the palladium center that favors the desired reductive elimination step while suppressing side reactions like beta-hydride elimination, which could otherwise lead to racemization or alkene byproducts. Furthermore, the reaction temperature is carefully controlled between 70°C and 80°C, a range that provides sufficient energy for the coupling to proceed to completion while minimizing thermal degradation of the sensitive amino acid ester. This balance ensures that the crude product profile is clean, simplifying downstream purification and ensuring the final hydrochloride salt meets stringent purity specifications exceeding 99%.
How to Synthesize (S)-2-amino-3-methylsulfonyl-benzyl phenylpropionate hydrochloride Efficiently
The practical implementation of this synthesis involves a sequential addition protocol designed to maximize safety and yield. Initially, the organozinc reagent is prepared in a separate vessel to ensure complete consumption of the iodide starting material before introduction to the palladium catalyst. This separation prevents potential side reactions between the zinc and the catalyst precursors. Once the organozinc species is confirmed ready via TLC or GC analysis, it is slowly added to the heated mixture of the aryl bromide, palladium catalyst, and ligand. The reaction progress is monitored closely using LC-MS to determine the exact endpoint, preventing over-reaction or decomposition. Following the coupling, the workup involves a straightforward extraction and concentration, followed by acid-mediated deprotection to yield the final ammonium salt.
- Generate the organozinc reagent by reacting N-Boc-3-iodo-L-alanine benzyl ester with activated zinc powder in DMAC at 60-70°C.
- Perform the Negishi coupling reaction between the organozinc reagent and 3-bromophenyl methyl sulfone using a palladium catalyst and phosphine ligand at 70-80°C.
- Remove the Boc protecting group and isolate the final hydrochloride salt product through acidification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical elegance. The most immediate impact is observed in the simplification of the raw material basket. By shifting away from expensive, chiral halogenated amino acids like L-3-bromophenylalanine towards commodity chemicals like 3-bromophenyl methyl sulfone and generic iodo-alanine derivatives, the process decouples production costs from the volatility of niche chiral pool pricing. This substitution effect drives substantial cost savings, as the new starting materials are synthesized in much larger volumes globally, ensuring stable pricing and consistent quality. Additionally, the reduction in step count directly correlates to reduced labor hours and utility consumption per kilogram of output.
- Cost Reduction in Manufacturing: The elimination of two full synthetic steps (Boc protection and separate sulfone introduction) removes the associated reagent costs, solvent usage, and waste disposal fees for those specific operations. Without needing to purchase costly copper catalysts or perform multiple chromatographic purifications, the overall cost of goods is significantly lowered. The use of recyclable solvents like DMAC and ethyl acetate further enhances the economic profile, allowing for a leaner manufacturing budget that can be passed on as competitive pricing to downstream partners.
- Enhanced Supply Chain Reliability: Diversifying the supply base for starting materials mitigates the risk of single-source bottlenecks. Since the key aryl bromide and iodo-alanine components are produced by multiple chemical manufacturers worldwide, the risk of supply disruption is drastically minimized compared to relying on a sole supplier for a complex chiral intermediate. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry, also means that batch failure rates are reduced, ensuring a more predictable and continuous flow of material to meet production schedules.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful scale-up in a 50L reactor, proving that the chemistry translates well from the laboratory to pilot and commercial scales. The process avoids the use of highly toxic heavy metals other than the trace palladium, which can be efficiently scavenged, and eliminates the generation of copper-containing waste streams associated with the old method. This alignment with green chemistry principles simplifies regulatory compliance and environmental permitting, accelerating the timeline for facility approval and commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains.
Q: What are the key advantages of this Negishi coupling route over traditional methods?
A: This method significantly shortens the synthetic route from four steps to effectively two major transformations, eliminating the need for harsh sulfone introduction conditions on a chiral amino acid backbone, thereby improving overall yield and optical purity.
Q: Which catalyst system is preferred for this transformation?
A: The patent specifies the use of palladium sources such as Pd2(dba)3 or Pd(OAc)2 combined with bulky phosphine ligands like tris(o-methylphenyl)phosphine to ensure efficient cross-coupling of the sterically hindered substrates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up in a 50L reactor (Example 7), achieving consistent yields around 45% and purity exceeding 99.5%, confirming its viability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-amino-3-methylsulfonyl-benzyl phenylpropionate hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving therapies like Lifitegrast. Our team of expert chemists has thoroughly analyzed the methodology described in CN111471003A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to fruition. We are equipped with state-of-the-art reactors capable of handling air-sensitive organometallic reactions and rigorous QC labs that enforce stringent purity specifications to ensure every batch meets the highest international standards. Our commitment to technical excellence ensures that we can deliver this complex intermediate with the consistency and reliability your projects demand.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall project costs.
