Advanced Biocatalytic Synthesis of Sitagliptin Intermediates Using Engineered (R)-ω-Transaminase Mutants
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for the production of high-value Active Pharmaceutical Ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in Chinese Patent CN111411094B, which discloses a novel (R)-ω-transaminase mutant and its specific application in the biocatalytic synthesis of Sitagliptin intermediates. This patent represents a pivotal advancement in protein engineering, addressing the longstanding challenges of substrate inhibition and low catalytic efficiency that have historically plagued the enzymatic production of chiral amines. By leveraging gene mining technologies to identify wild-type enzymes from Aspergillus terreus and subsequently applying rigorous multi-site directed mutagenesis, the inventors have developed a biocatalyst capable of operating under industrially favorable conditions. This innovation is particularly critical for the manufacturing of Sitagliptin, a blockbuster DPP-4 inhibitor, where the demand for high-purity chiral intermediates continues to surge globally. The technology described offers a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to diversify their sourcing strategies and reduce dependency on single-source proprietary technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biocatalytic preparation of Sitagliptin has been dominated by proprietary technologies, such as those developed by Merck and Codexis, which utilize specific (R)-ω-transaminase variants. While effective, these established methods often face limitations regarding substrate loading capacities and operational flexibility. For instance, prior art processes, such as those described in Chinese Patent CN108586346A, typically employ substrate concentrations around 367 mM to maintain acceptable conversion rates, which inherently limits the volumetric productivity of the reactor. Operating at lower substrate concentrations necessitates the use of larger reaction volumes and increased amounts of solvents, leading to higher downstream processing costs and a larger environmental footprint. Furthermore, the reliance on a narrow range of enzyme sources creates supply chain vulnerabilities, as manufacturers are often locked into specific licensing agreements or dependent on the availability of a single engineered strain. The conventional chemical routes, involving asymmetric hydrogenation or chiral resolution, often require expensive transition metal catalysts and rigorous purification steps to remove trace metal impurities, adding complexity and cost to the overall manufacturing process.
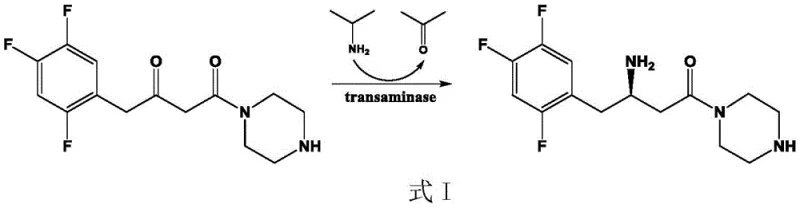
The Novel Approach
In stark contrast to these limitations, the technology disclosed in CN111411094B introduces a highly engineered (R)-ω-transaminase mutant that exhibits superior substrate tolerance and catalytic activity. Through a systematic process of molecular modification involving six specific point mutations (L182M, R79A, Q51S, V149D, L235E, and G216A), the resulting enzyme variant, designated as AtTA6, demonstrates the ability to efficiently catalyze the amination of the precursor ketone analogue 1-(3-oxopyrrolidin-1-yl)-4-(2,4,5-trifluorophenyl)-1,3-butanedione. This novel biocatalyst allows for substrate concentrations ranging from 600 mM to 900 mM, with optimal performance observed at 800 mM, achieving conversion rates as high as 95.4%. This substantial increase in substrate loading capacity directly translates to improved process economics by maximizing the output per batch and minimizing solvent usage. Moreover, the mutant enzyme maintains excellent stereoselectivity, producing the desired (R)-configuration with an e.e. value exceeding 99%, which is essential for meeting the stringent quality standards required for pharmaceutical intermediates. The ability to operate effectively at elevated temperatures (up to 50°C) further enhances the reaction kinetics, driving the equilibrium towards product formation and reducing the overall reaction time.
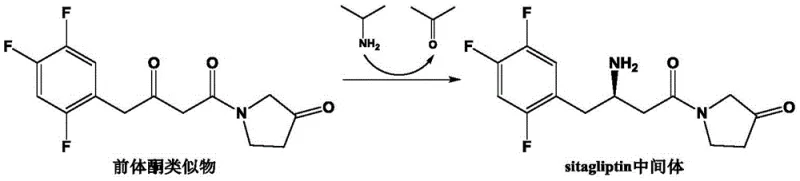
Mechanistic Insights into (R)-ω-Transaminase Catalyzed Asymmetric Amination
The core of this technological advancement lies in the precise manipulation of the enzyme's active site to accommodate the bulky and sterically demanding precursor ketone analogue. Transaminases function via a ping-pong bi-bi mechanism involving the cofactor pyridoxal 5'-phosphate (PLP). In the first half-reaction, the amino group from the donor, typically isopropylamine in this industrial application, is transferred to the PLP to form pyridoxamine 5'-phosphate (PMP). In the second half-reaction, the amino group is transferred from PMP to the carbonyl acceptor, the precursor ketone, generating the chiral amine product and regenerating PLP. The specific mutations introduced in the AtTA sequence, such as the substitution of Leucine with Methionine at position 182 and Arginine with Alanine at position 79, are strategically located within the substrate binding pocket. These alterations likely expand the hydrophobic volume of the active site and reduce steric hindrance, allowing the trifluorophenyl moiety of the substrate to bind more favorably. Additionally, the mutation of Valine to Aspartic Acid at position 149 may introduce new electrostatic interactions that stabilize the transition state, thereby lowering the activation energy of the rate-limiting step. This rational design approach ensures that the enzyme not only accepts the non-natural substrate but does so with high fidelity, strictly enforcing the formation of the (R)-enantiomer through precise spatial orientation of the substrate within the catalytic cleft.
Controlling the impurity profile is another critical aspect of this mechanistic framework. In biocatalytic processes, side reactions such as non-specific amination or the formation of by-products due to enzyme instability can compromise the purity of the final API. The enhanced thermal stability of the AtTA6 mutant, evidenced by its optimal activity at 50°C compared to the wild type, suggests a more rigid and robust protein structure that resists denaturation under process conditions. This structural integrity minimizes the release of host cell proteins and other biological contaminants into the reaction mixture. Furthermore, the high stereoselectivity (>99% e.e.) effectively eliminates the formation of the unwanted (S)-enantiomer, which would otherwise act as a difficult-to-remove impurity requiring costly chiral chromatography for separation. By ensuring that the reaction proceeds almost exclusively through the desired stereochemical pathway, the process simplifies the downstream purification train, potentially allowing for direct crystallization of the product from the reaction mixture. This level of control over the reaction trajectory is paramount for pharmaceutical manufacturers who must adhere to strict regulatory guidelines regarding impurity thresholds and batch-to-batch consistency.
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this biocatalytic route requires a structured approach to fermentation and bioconversion to maximize the yield and quality of the Sitagliptin intermediate. The process begins with the cultivation of recombinant E. coli strains harboring the plasmid encoding the optimized transaminase mutant, followed by induction of enzyme expression and harvesting of the whole cells. These wet cells serve as the biocatalyst in a buffered aqueous system containing the ketone substrate and amine donor. The detailed standardized synthesis steps, including specific buffer compositions, pH controls, and work-up procedures, are outlined in the comprehensive guide below.
- Preparation of the biocatalyst involves cultivating recombinant E. coli strains expressing the specific (R)-ω-transaminase mutant (e.g., AtTA6) and harvesting the wet cells via centrifugation.
- The biocatalytic reaction is conducted in a triethanolamine-HCl buffer system containing the precursor ketone analogue substrate, isopropylamine as the amino donor, and PLP as a coenzyme at optimized temperatures around 50°C.
- Post-reaction processing includes stopping the reaction with acid, separating the supernatant, and isolating the chiral amine product through crystallization or extraction to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel biocatalytic technology offers substantial strategic benefits that extend beyond mere technical feasibility. The ability to utilize higher substrate concentrations fundamentally alters the cost structure of the manufacturing process. By processing more material in the same reactor volume, companies can significantly reduce the consumption of solvents, water, and energy associated with heating and cooling large liquid volumes. This intensification of the process leads to a drastic simplification of the waste treatment requirements, as there is less effluent to manage per kilogram of product produced. Furthermore, the elimination of transition metal catalysts, which are often required in competing chemical synthesis routes, removes the need for expensive and time-consuming metal scavenging steps. This not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions related to the availability of precious metals. The robust nature of the enzyme also implies a longer operational lifespan and potential for reuse, further enhancing the economic viability of the process on a commercial scale.
- Cost Reduction in Manufacturing: The implementation of this high-efficiency biocatalyst drives down manufacturing costs through process intensification and waste minimization. By enabling substrate concentrations up to 900 mM, the technology maximizes reactor throughput, meaning fewer batches are required to meet production targets, which optimizes labor and utility usage. The high stereoselectivity eliminates the need for chiral resolution steps, which are traditionally capital-intensive and yield-loss prone. Additionally, the use of whole-cell biocatalysts avoids the costs associated with enzyme purification, as the crude cell lysate or intact cells can often be used directly. The removal of heavy metal catalysts from the process flow further reduces the cost burden associated with specialized filtration media and analytical testing for metal residues, resulting in a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Diversifying the enzyme source is a critical strategy for mitigating supply chain risks in the pharmaceutical sector. This technology utilizes a novel enzyme scaffold derived from Aspergillus terreus, which is distinct from the proprietary enzymes currently dominating the market. This independence provides manufacturers with a secure alternative supply route, reducing vulnerability to licensing disputes or single-vendor bottlenecks. The genetic stability of the recombinant strains ensures consistent enzyme production over time, guaranteeing a steady supply of the biocatalyst. Moreover, the simplified reaction conditions and robustness of the mutant enzyme facilitate technology transfer between different manufacturing sites, allowing for a more flexible and resilient global supply network that can adapt to regional demand fluctuations without compromising product quality.
- Scalability and Environmental Compliance: Scaling biocatalytic processes from the laboratory to industrial production is often hindered by mass transfer limitations and enzyme instability, but this mutant addresses those challenges directly. Its enhanced thermal stability allows for operation at 50°C, which improves substrate solubility and reaction kinetics, making scale-up more predictable and manageable. The aqueous nature of the biocatalytic reaction aligns perfectly with green chemistry principles, significantly reducing the reliance on volatile organic compounds (VOCs) compared to traditional organic synthesis. This environmental advantage simplifies regulatory compliance regarding emissions and waste disposal, positioning the manufacturer as a leader in sustainable pharmaceutical production. The high conversion rates minimize the amount of unreacted starting material that needs to be recovered or disposed of, further streamlining the environmental management of the facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this (R)-ω-transaminase technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing infrastructure.
Q: What represents the primary advantage of this new transaminase mutant over existing commercial enzymes?
A: The primary advantage lies in its significantly enhanced substrate tolerance and thermal stability. Unlike previous generations that were limited to lower substrate concentrations (e.g., ~367 mM), this mutant operates efficiently at concentrations up to 900 mM, drastically improving space-time yield and reducing solvent waste in large-scale manufacturing.
Q: How does the stereoselectivity of this biocatalytic route compare to chemical asymmetric synthesis?
A: The engineered (R)-ω-transaminase mutant demonstrates exceptional stereoselectivity, consistently achieving an enantiomeric excess (e.e.) value of greater than 99%. This eliminates the need for complex chiral resolution steps often required in traditional chemical synthesis, thereby simplifying the downstream purification process.
Q: Is this technology suitable for breaking the supply chain monopoly on Sitagliptin intermediates?
A: Yes, by utilizing a novel enzyme source derived from Aspergillus terreus and optimizing it through multi-site directed mutagenesis, this technology provides a distinct, non-infringing pathway for producing key Sitagliptin intermediates, offering pharmaceutical manufacturers a strategic alternative to established proprietary processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of complex pharmaceutical intermediates like those for Sitagliptin. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art fermentation suites and rigorous QC labs capable of handling sensitive biocatalytic processes while maintaining stringent purity specifications. We understand the critical importance of supply continuity and quality assurance in the pharmaceutical value chain, and our team is prepared to deploy this novel transaminase technology to secure your supply of high-purity Sitagliptin intermediates.
We invite you to engage with our technical procurement team to discuss how this cutting-edge biocatalytic route can be tailored to your specific production needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-efficiency enzymatic process. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our engineered strains against your current benchmarks. Let us collaborate to optimize your supply chain and drive down costs while ensuring the highest standards of quality and sustainability in your API manufacturing operations.
