Scalable Synthesis of 2-Methylpyridine Compounds via Triethylamine Cyclization for Industrial Applications
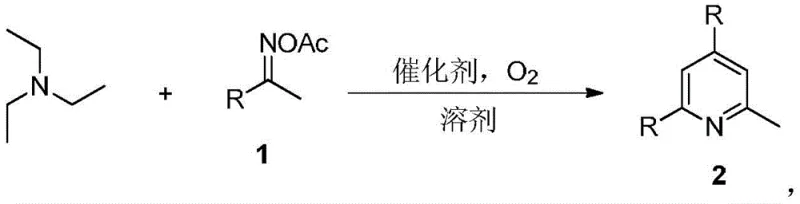
The pharmaceutical and agrochemical industries rely heavily on pyridine scaffolds, with 2-methylpyridine (2-picoline) serving as a critical building block for active pharmaceutical ingredients (APIs) such as long-acting sulfonamides and antihistamines like chlorpheniramine. Patent CN108314642B introduces a transformative synthetic methodology that addresses the longstanding inefficiencies in producing these valuable intermediates. By utilizing triethylamine and oxime acetates in a transition-metal-free, one-pot tandem reaction, this technology bypasses the hazardous high-pressure conditions associated with traditional acetylene or ethylene-based routes. The process operates under a mild oxygen atmosphere at temperatures ranging from 120°C to 160°C, utilizing inexpensive iodine-based catalysts to drive the cyclization. This innovation not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by minimizing waste and eliminating the need for costly transition metal removal steps, thereby presenting a compelling value proposition for manufacturers seeking reliable pharmaceutical intermediates supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-methylpyridine compounds has been dominated by methods involving acetaldehyde, acetylene, ethylene, or acrylonitrile. These conventional pathways are fraught with significant operational challenges that impact both economic viability and safety protocols. Specifically, the acetylene and ethylene methods necessitate extreme reaction environments characterized by high temperatures and high pressures, which inherently increase the risk of catastrophic equipment failure and require specialized, capital-intensive infrastructure. Furthermore, these processes are notorious for generating complex mixtures of byproducts due to uncontrolled side reactions, leading to low isolated yields and necessitating rigorous, multi-step purification procedures. The environmental burden of disposing of these chemical wastes, combined with the high energy consumption required to maintain severe reaction conditions, results in a substantially elevated cost of goods sold (COGS) that erodes profit margins for downstream drug manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN108314642B leverages triethylamine, a ubiquitous and inexpensive bulk chemical, as the primary nitrogen source and carbon skeleton provider. This novel approach facilitates a direct, one-step construction of the pyridine ring through a cascade reaction with oxime acetates, effectively collapsing what would traditionally be a multi-step synthesis into a single operational unit. The elimination of transition metal catalysts is particularly advantageous, as it removes the regulatory and technical hurdles associated with residual metal limits in final API products. By operating at atmospheric pressure with molecular oxygen as the terminal oxidant, the process drastically simplifies reactor requirements and enhances workplace safety. This shift from hazardous gas-phase chemistry to a more manageable liquid-phase reaction represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a cleaner, safer, and more economically efficient pathway to high-value heterocyclic compounds.
Mechanistic Insights into Iodine-Catalyzed Tandem Cyclization
The core of this synthetic breakthrough lies in the iodine-mediated oxidative cyclization mechanism, which orchestrates the assembly of the pyridine ring from simple precursors. The reaction initiates with the activation of the oxime acetate by the iodine catalyst, generating a reactive nitrile oxide or similar electrophilic species in situ. Triethylamine then participates in a complex sequence of C-H activation and condensation steps, where the ethyl groups serve as two-carbon synthons that integrate into the forming heterocycle. The presence of molecular oxygen is critical, acting as a green oxidant to regenerate the active iodine species and drive the aromatization of the intermediate dihydropyridine species to the final stable pyridine system. This tandem sequence avoids the isolation of unstable intermediates, thereby minimizing decomposition pathways and maximizing atom economy. The robustness of this catalytic cycle allows for the accommodation of diverse electronic environments on the aromatic ring, ensuring consistent performance across a broad substrate scope.
From an impurity control perspective, the simplicity of the one-pot design inherently reduces the generation of carry-over impurities that typically accumulate during multi-step syntheses. The use of iodine, N-iodosuccinimide, or ammonium iodide as catalysts ensures that the reaction proceeds with high chemoselectivity, minimizing the formation of polymeric tars or over-oxidized byproducts that plague radical-based oxidations. The workup procedure described in the patent, involving aqueous quenching and standard organic extraction, effectively removes inorganic iodine salts and unreacted starting materials. This streamlined purification profile is crucial for maintaining high purity specifications required for pharmaceutical grade intermediates. The ability to tolerate various functional groups, including halogens and electron-withdrawing substituents, without significant degradation further underscores the mechanistic stability of this iodine-catalyzed system, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Methylpyridine Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory or plant equipment without the need for specialized high-pressure autoclaves. The general procedure involves dissolving the oxime acetate substrate and triethylamine in a solvent such as toluene, acetonitrile, or 1,4-dioxane, followed by the addition of the iodine catalyst. The reaction mixture is then heated under an oxygen atmosphere, typically at 140°C, for a duration of approximately 12 hours. This standardized protocol has been validated across a wide array of substrates, demonstrating consistent reproducibility. For detailed operational parameters regarding specific substrates and optimization tips, please refer to the standardized synthesis steps outlined below.
- Dissolve triethylamine and the specific oxime acetate compound in a suitable solvent such as toluene, acetonitrile, or 1,4-dioxane within a reaction vessel.
- Add the iodine-based catalyst (Iodine, N-iodosuccinimide, or Ammonium Iodide) to the mixture and establish an oxygen atmosphere.
- Heat the reaction mixture to a temperature between 120°C and 160°C, typically 140°C, and stir for approximately 12 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this triethylamine-based route offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary driver for cost optimization is the substitution of expensive, hazardous gases like acetylene with triethylamine, a commodity chemical with a stable and abundant global supply chain. This shift mitigates the volatility associated with specialty gas pricing and logistics, ensuring a more predictable raw material cost structure. Furthermore, the elimination of transition metal catalysts removes the necessity for expensive scavenging resins or complex filtration systems designed to meet strict heavy metal residue limits, directly translating to substantial cost savings in downstream processing. The simplified workflow also reduces labor hours and utility consumption, contributing to a leaner manufacturing overhead.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of inexpensive reagents and the avoidance of high-pressure infrastructure. By removing the need for precious metal catalysts, the process eliminates a significant line item in the bill of materials. Additionally, the one-pot nature of the reaction reduces solvent usage and waste disposal costs, as there are no intermediate isolation steps requiring additional drying and purification. This holistic reduction in operational complexity allows for a more competitive pricing strategy for the final high-purity pharmaceutical intermediates, enhancing margin potential for partners.
- Enhanced Supply Chain Reliability: Relying on triethylamine and simple oxime acetates diversifies the raw material base away from petrochemical gases that are subject to refinery outages and transportation bottlenecks. Triethylamine is produced at massive scales globally, ensuring continuity of supply even during market disruptions. The mild reaction conditions also reduce equipment maintenance frequency and downtime, as reactors are not subjected to the corrosive and stressful environments of high-pressure acid or gas chemistry. This reliability is critical for maintaining just-in-time delivery schedules for key clients in the pharmaceutical sector.
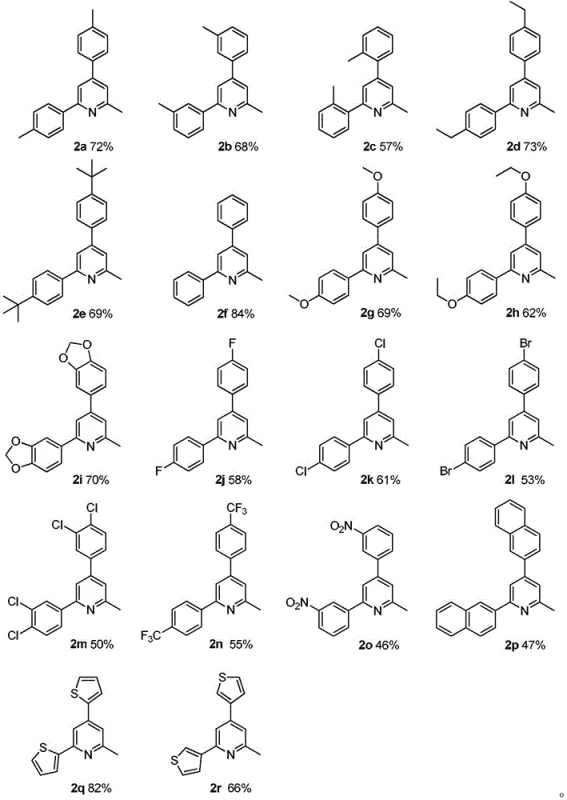
- Scalability and Environmental Compliance: The transition from batch processes involving hazardous gases to a liquid-phase oxidation significantly lowers the environmental footprint of the manufacturing site. The absence of heavy metal waste simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. The process is inherently scalable, as demonstrated by the successful synthesis of various derivatives in the patent examples, moving seamlessly from gram-scale optimization to kilogram and ton-scale production. This scalability ensures that supply can be ramped up quickly to meet surging demand for APIs without requiring prohibitive capital investment in new high-pressure facilities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the reaction's scope and operational parameters. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for assessing the fit of this methodology for specific target molecules and for planning the necessary quality control measures.
Q: What are the advantages of using triethylamine over traditional acetylene methods?
A: Traditional methods often require high temperature and high pressure environments with acetylene or ethylene, posing significant safety risks and yielding numerous side products. The triethylamine route operates under milder conditions (120-160°C) without high pressure, significantly improving operational safety and reducing purification costs associated with complex byproduct mixtures.
Q: Which catalysts are effective for this iodine-mediated cyclization?
A: The patent specifies that elemental iodine, N-iodosuccinimide (NIS), and ammonium iodide are all effective catalysts for this transformation. Elemental iodine generally provides robust yields across a wide range of substrates, while NIS offers a convenient solid alternative that can simplify handling in certain manufacturing setups.
Q: Does this method tolerate electron-withdrawing groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. Substrates containing electron-withdrawing groups such as nitro, trifluoromethyl, fluoro, chloro, and bromo substituents successfully undergo cyclization to form the corresponding 2-methylpyridine derivatives, although yields may vary slightly depending on the specific electronic nature and position of the substituent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylpyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain. Our team of expert chemists has extensively evaluated the triethylamine cyclization route described in CN108314642B and confirmed its potential for robust industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-methylpyridine derivatives delivered meets the exacting standards required for API synthesis. We are committed to leveraging this advanced chemistry to provide our partners with a secure, high-quality supply of essential heterocyclic building blocks.
We invite forward-thinking pharmaceutical companies to collaborate with us to unlock the full commercial potential of this innovative synthesis. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our pilot batches and to discuss route feasibility assessments for your upcoming projects. Let us help you optimize your supply chain and reduce your time-to-market with our superior manufacturing capabilities.
