Advanced Synthesis of Spiro[indazole-pyrazoline] Derivatives for Antitumor Drug Development
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer improved therapeutic indices, particularly in the challenging field of oncology. Patent CN110642864A discloses a sophisticated methodology for the preparation of spiro[indazole-pyrazoline] derivatives containing a pyrazole structure, representing a significant advancement in the design of antitumor agents. This technology leverages the principles of 1,3-dipolar cycloaddition to construct complex spirocyclic frameworks that are difficult to access through conventional linear synthesis. For R&D directors and procurement specialists, understanding the nuances of this synthetic pathway is crucial, as it offers a viable route to high-value intermediates with demonstrated biological activity against liver and colorectal cancer cell lines. The integration of triazole and pyrazole moieties within a single spiro architecture enhances the molecular diversity and potential binding affinity, making these compounds attractive candidates for further drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex spiro-heterocycles often suffer from poor regioselectivity and the requirement for harsh reaction conditions that can degrade sensitive functional groups. Many classical methods rely on multi-step sequences involving expensive transition metal catalysts, which introduce significant challenges in downstream processing, particularly regarding the removal of trace metal impurities to meet stringent pharmaceutical standards. Furthermore, conventional routes frequently struggle to control the stereochemistry at the spiro-center, leading to mixtures of diastereomers that complicate purification and reduce overall process efficiency. The reliance on unstable intermediates in older methodologies often necessitates cryogenic conditions that are difficult to maintain on a large industrial scale, thereby limiting the commercial feasibility of producing these high-potency scaffolds in metric ton quantities.
The Novel Approach
The methodology outlined in the patent overcomes these hurdles by utilizing a streamlined 1,3-dipolar cycloaddition strategy that exhibits excellent regio- and stereoselectivity. By generating a nitrile oxide intermediate in situ from an α-chloro-hydrazone precursor, the process facilitates a highly efficient coupling with an exocyclic double bond on the indazolone scaffold. This approach eliminates the need for precious metal catalysts, significantly simplifying the workup procedure and reducing the environmental footprint of the manufacturing process. The resulting spiro[indazole-pyrazoline] structure is formed with high fidelity, as evidenced by the clean reaction profiles and robust yields reported in the experimental data. ![Chemical structure of the target spiro[indazole-pyrazoline] derivative showing the complex fused ring system](/insights/img/spiro-indazole-pyrazoline-derivative-supplier-pharma-20260307051240-02.png)
Mechanistic Insights into 1,3-Dipolar Cycloaddition
The core of this synthetic innovation lies in the generation and trapping of the nitrile oxide dipole. The process begins with the chlorination of the pyrazole-carboxaldehyde hydrazone using tert-butyl hypochlorite at strictly controlled low temperatures, typically around -12°C. This step is critical, as the resulting α-chloro-hydrazone serves as the precursor to the nitrile oxide upon base-mediated dehydrohalogenation. The subsequent cycloaddition with the dipolarophile, 5-benzylidene-1-phenyl-6,7-dihydro-1H-indazol-4(5H)-one, proceeds through a concerted mechanism that ensures the oxygen atom of the nitrile oxide adds to the β-carbon of the exocyclic double bond. 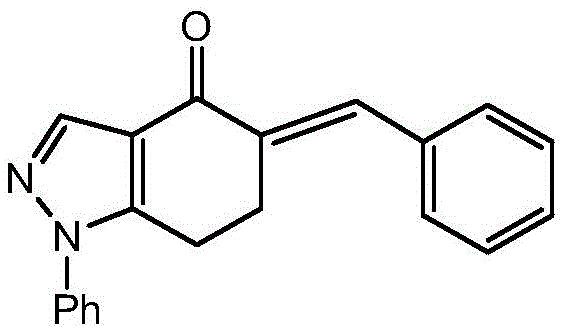
Impurity control is inherently built into this mechanism due to the high reactivity and short lifetime of the nitrile oxide species, which minimizes side reactions such as dimerization. The use of pyridine as a mild base in the final cyclization step further enhances selectivity by preventing the degradation of the sensitive triazole and pyrazole rings. From a quality control perspective, this mechanistic pathway ensures a consistent impurity profile, as the major byproducts are typically polar and easily removed via standard silica gel chromatography or recrystallization. The precise molar ratio of 3:2 between the nitrile oxide precursor and the dipolarophile is optimized to drive the reaction to completion while minimizing the accumulation of unreacted starting materials, thereby maximizing the space-time yield of the reactor.
How to Synthesize Spiro[indazole-pyrazoline] Derivatives Efficiently
The synthesis involves a five-step sequence starting from readily available pyrazole and indazole precursors. The process requires careful attention to temperature control during the chlorination step and precise stoichiometry during the final cycloaddition. Detailed standard operating procedures for scaling this route from gram to kilogram scale are essential for maintaining product quality.
- Synthesize 1-phenyl-3-methyl-5-(1,2,4-triazolyl)-4-pyrazolecarboxaldehyde via nucleophilic substitution in DMSO.
- Convert the aldehyde to its hydrazone derivative using phenylhydrazine under reflux conditions.
- Generate the nitrile oxide precursor by chlorinating the hydrazone with tert-butyl hypochlorite at严格控制 temperatures below -5°C.
- Prepare the dipolarophile, 5-benzylidene-1-phenyl-6,7-dihydro-1H-indazol-4(5H)-one, via aldol condensation.
- Perform the final 1,3-dipolar cycloaddition between the nitrile oxide and the exocyclic double bond to form the spiro cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and raw material security. The elimination of transition metal catalysts not only reduces the direct cost of goods sold but also removes the need for specialized scavenging resins and extensive analytical testing for heavy metals, which are significant cost drivers in API manufacturing. The starting materials, such as benzaldehyde and phenylhydrazine derivatives, are commodity chemicals with robust global supply chains, ensuring that production schedules are not disrupted by the scarcity of exotic reagents. This reliability is paramount for maintaining continuous supply to downstream drug manufacturers who require just-in-time delivery of critical intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive organic oxidants like tert-butyl hypochlorite instead of costly metal-based reagents. The high atom economy of the cycloaddition step means that a larger proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs. Furthermore, the simplified purification protocol, which relies on standard filtration and recrystallization rather than complex preparative HPLC, drastically lowers the operational expenditure associated with downstream processing. These factors combine to create a highly competitive cost structure for the production of these complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: The synthetic route is designed with supply chain resilience in mind, utilizing reagents that are widely available from multiple qualified vendors globally. The robustness of the reaction conditions, particularly the tolerance of the final step to ambient temperatures after the initial mixing, allows for flexible manufacturing scheduling without the need for specialized cryogenic infrastructure beyond the specific chlorination stage. This flexibility enables manufacturers to respond quickly to fluctuations in demand, ensuring that lead times for high-purity pharmaceutical intermediates remain short and predictable even during periods of market volatility.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of common solvents like ethanol and ethyl acetate, which are easier to recover and recycle compared to chlorinated solvents often used in alternative routes. The absence of heavy metals simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. The high yields achieved under optimized conditions, such as the controlled addition of oxidants, ensure that the process generates minimal chemical waste, aligning with modern green chemistry principles and helping pharmaceutical partners meet their sustainability goals without compromising on output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro-compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and relevance for industry stakeholders.
Q: What is the biological activity profile of the synthesized spiro[indazole-pyrazoline] derivatives?
A: According to patent CN110642864A, the target Compound 5 demonstrates significant inhibitory activity against HepG2 (liver cancer) and SW620 (colorectal adenocarcinoma) cell lines, with IC50 values indicating strong potency, while showing lower activity against other tested lines like A549 and SKOV3.
Q: Why is temperature control critical in the chlorination step of this synthesis?
A: The formation of the α-chloro-hydrazone intermediate requires strict temperature control below -5°C, ideally around -12°C. Experimental data indicates that raising the temperature to 0°C results in a complete loss of yield (0%), highlighting the thermal instability of the reactive intermediates involved.
Q: How does the 1,3-dipolar cycloaddition improve the synthetic route?
A: This reaction allows for the efficient construction of the complex spiro-isoxazoline ring system with high regioselectivity. It avoids the need for transition metal catalysts, simplifying purification and reducing the risk of heavy metal contamination in the final pharmaceutical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[indazole-pyrazoline] Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of new drug candidates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of spiro[indazole-pyrazoline] derivatives meets the exacting standards required by global regulatory agencies. Our commitment to technical excellence ensures that the complex stereochemistry and structural integrity of these molecules are preserved throughout the manufacturing process.
We invite you to contact our technical procurement team to discuss how we can support your specific development needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic route can reduce your overall project costs. We are ready to provide specific COA data and route feasibility assessments tailored to your timeline, ensuring that you have all the necessary information to make informed sourcing decisions for your antitumor drug pipeline.
