Advanced Synthesis of Spiro[indazole-pyrazoline] Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer improved pharmacokinetic profiles and potent biological activity, particularly in the realm of oncology. Patent CN110642864A introduces a sophisticated synthetic methodology for producing spiro[indazole-pyrazoline] derivatives containing a pyrazole structure, which have demonstrated significant potential as antitumor agents. This technology represents a substantial leap forward in medicinal chemistry, offering a robust route to complex spirocyclic systems that are notoriously difficult to synthesize with high regioselectivity. By leveraging a strategic 1,3-dipolar cycloaddition approach, this process enables the efficient construction of a molecular architecture that integrates multiple pharmacophores, including triazole, pyrazole, and indazole moieties, into a single rigid framework. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is critical for securing a reliable supply of high-purity pharmaceutical intermediates needed for next-generation cancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing spiro-heterocycles often suffer from poor regiocontrol and harsh reaction conditions that can degrade sensitive functional groups. In many conventional pathways, the formation of the spiro center requires high temperatures or strong acidic conditions that lead to polymerization or decomposition of the starting materials. Furthermore, achieving the specific substitution pattern required for optimal biological activity—such as the integration of a 1,2,4-triazole ring alongside the spiro core—is frequently inefficient, resulting in complex mixtures that are difficult to separate. These inefficiencies not only drive up the cost of goods but also introduce significant variability in the quality of the final intermediate, posing a risk to downstream drug development timelines. The inability to precisely control the stereochemistry and regiochemistry at the spiro junction remains a persistent bottleneck in the scalable manufacturing of these valuable compounds.
The Novel Approach
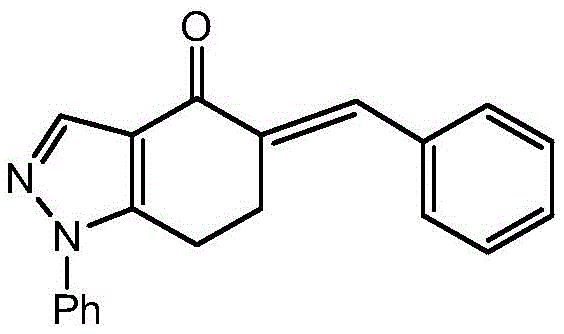
The methodology outlined in CN110642864A overcomes these historical challenges by employing a mild and highly selective 1,3-dipolar cycloaddition strategy. Instead of forcing the ring closure under drastic conditions, this novel approach generates a reactive nitrile oxide intermediate in situ from an alpha-chloro hydrazone precursor at low temperatures. This allows for a controlled reaction with an exocyclic double bond on an indazolone scaffold, ensuring that the spiro ring forms exclusively at the desired position. The process utilizes readily available starting materials and avoids the need for expensive transition metal catalysts, which simplifies the purification process and reduces the burden of heavy metal removal. By optimizing the stoichiometry and reaction parameters, this route achieves superior yields and purity profiles compared to legacy methods, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into 1,3-Dipolar Cycloaddition
The core of this synthetic innovation lies in the precise generation and trapping of the nitrile oxide dipole. The process begins with the conversion of a pyrazole aldehyde into a hydrazone, which is subsequently chlorinated using tert-butyl hypochlorite. This chlorination step is mechanistically critical; it must be conducted at strictly controlled low temperatures, specifically around -12°C, to prevent the premature decomposition of the unstable alpha-chloro hydrazone. Upon treatment with a base such as pyridine in the final step, the alpha-chloro hydrazone undergoes dehydrohalogenation to generate the reactive nitrile oxide species. This 1,3-dipole then immediately engages in a concerted cycloaddition with the electron-deficient exocyclic double bond of the 5-benzylidene-indazolone acceptor. The orbital symmetry allowed nature of this reaction ensures high stereoselectivity, locking the new isoxazoline ring into the spiro configuration with minimal formation of regioisomeric byproducts.
Impurity control is paramount in this mechanism, particularly regarding the stability of the nitrile oxide intermediate. If the temperature rises above -5°C during the chlorination phase, the yield drops precipitously, indicating competitive side reactions or decomposition pathways become dominant. The patent data explicitly shows that maintaining the reaction at -12°C results in a yield of 92%, whereas running the same reaction at 0°C leads to a complete failure with 0% yield. This sharp sensitivity underscores the importance of precise thermal management in the reactor design. Additionally, the use of a specific molar excess of the nitrile oxide precursor (3:2 ratio relative to the dipolarophile) helps to drive the reaction to completion, compensating for any minor losses due to dimerization of the nitrile oxide, thereby ensuring a clean reaction profile that facilitates downstream isolation.
How to Synthesize Spiro[indazole-pyrazoline] Derivatives Efficiently
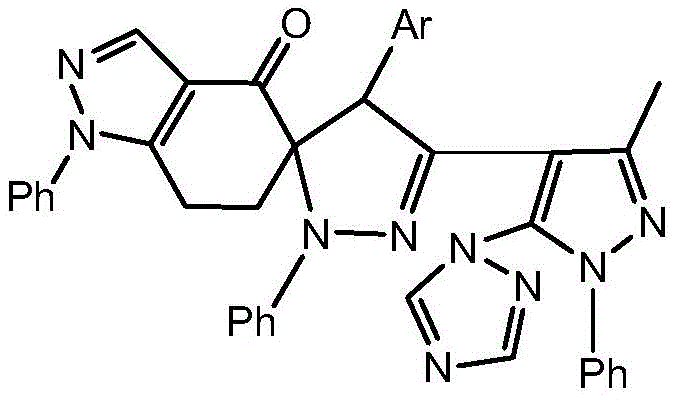
The synthesis of these high-value derivatives follows a logical five-step sequence that balances chemical efficiency with operational simplicity. The process begins with the nucleophilic substitution to install the triazole ring, followed by hydrazone formation and the critical low-temperature chlorination. The parallel synthesis of the indazolone ketone acceptor via aldol condensation provides the necessary dipolarophile. The convergence of these two fragments in the final cycloaddition step creates the complex spiro architecture in a single operation. For detailed operational parameters, safety data, and specific workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Synthesize 1-phenyl-3-methyl-5-(1,2,4-triazolyl)-4-pyrazolecarboxaldehyde via nucleophilic substitution.
- Convert the aldehyde to a hydrazone using phenylhydrazine under reflux conditions.
- Generate the nitrile oxide precursor by chlorinating the hydrazone with tert-butyl hypochlorite at -12°C.
- Prepare the dipolarophile 5-benzylidene-1-phenyl-6,7-dihydro-1H-indazol-4(5H)-one via aldol condensation.
- Perform the final 1,3-dipolar cycloaddition between the nitrile oxide and the exocyclic double bond to form the spiro core.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented methodology offers distinct advantages that translate directly into cost reduction in pharmaceutical intermediates manufacturing. By eliminating the need for precious metal catalysts and utilizing common organic solvents like ethanol and dichloroethane, the process significantly lowers the raw material costs and simplifies the waste stream management. The high selectivity of the reaction means that fewer purification steps are required, reducing the consumption of silica gel and eluents, which are major cost drivers in intermediate production. Furthermore, the robustness of the reaction conditions, provided that temperature controls are respected, ensures consistent batch-to-batch quality, reducing the risk of costly batch failures that can disrupt supply continuity for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for specialized scavenging resins and extensive heavy metal testing, leading to substantial cost savings. The high yield achieved through optimized stoichiometry minimizes raw material waste, ensuring that every kilogram of input translates effectively into output. Additionally, the use of standard unit operations such as filtration and recrystallization allows for processing in existing multipurpose facilities without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including benzaldehyde, phenylhydrazine, and common heterocyclic building blocks, are commodity chemicals with stable global supply chains. This reduces the risk of supply bottlenecks associated with exotic or proprietary reagents. The process scalability has been demonstrated through careful optimization of addition rates and thermal profiles, ensuring that the technology can be transferred from laboratory to pilot plant with minimal technical risk, thereby securing long-term availability for clients.
- Scalability and Environmental Compliance: The solvent system employed is compatible with standard recovery and recycling protocols, aligning with modern green chemistry principles and reducing the environmental footprint of production. The avoidance of hazardous reagents and the generation of manageable byproducts simplify the regulatory compliance landscape for waste disposal. This environmental efficiency not only lowers disposal costs but also enhances the sustainability profile of the final product, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on critical process parameters. Understanding these details is essential for process chemists aiming to adapt this methodology for large-scale production environments.
Q: What is the critical temperature control parameter for the chlorination step?
A: The chlorination of the hydrazone intermediate with tert-butyl hypochlorite must be strictly maintained below -5°C, ideally at -12°C. Deviation to 0°C results in a complete loss of yield (0%), while -12°C ensures optimal conversion up to 92%.
Q: Why is the molar ratio of reactants significant in the final cycloaddition?
A: The patent specifies a molar ratio of 3:2 between the nitrile oxide precursor (Compound 3) and the indazolone ketone (Compound 4). This excess of the dipole generator drives the equilibrium forward, maximizing the formation of the spiro cycle while minimizing unreacted starting material.
Q: What purification method is recommended for the final spiro derivative?
A: The final product is best purified using silica gel column chromatography with a specific eluent system of ethyl acetate and petroleum ether in a 1:8 volume ratio, ensuring high purity suitable for biological testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[indazole-pyrazoline] Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of spiro[indazole-pyrazoline] derivatives meets the exacting standards required for clinical and commercial applications. Our commitment to technical excellence allows us to navigate the complexities of spiro-cycle synthesis with confidence.
We invite you to collaborate with us to leverage this advanced technology for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain strategy and accelerate your path to market.
