Advanced Synthesis of Ortho-Chlorophenyl Spiro[indazole-pyrazoline] Derivatives for Oncology Applications
Introduction to Novel Antitumor Scaffolds
The pharmaceutical landscape is continuously evolving with the discovery of novel heterocyclic scaffolds that exhibit potent biological activities, particularly in the field of oncology. Patent CN110655522A discloses a significant advancement in the synthesis of ortho-chlorophenyl-substituted spiro[indazole-pyrazoline] derivatives containing a pyrazole structure. These complex molecules are not merely academic curiosities but represent high-value pharmaceutical intermediates with demonstrated inhibitory effects against aggressive tumor cell lines such as SW620, NCL-H460, and SKOV3. The strategic incorporation of a triazole moiety alongside the spiro-fused indazole-pyrazoline core enhances the molecule's ability to interact with biological enzymes and receptors through hydrogen bonding and aromatic stacking. For R&D directors and procurement specialists, understanding the synthetic accessibility of such privileged structures is paramount. This report analyzes the technical robustness of this patented methodology, highlighting its potential for cost reduction in API manufacturing and its suitability for reliable supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of spiro-heterocyclic systems has been fraught with challenges regarding regioselectivity and reaction efficiency. Conventional routes often rely on multi-step sequences that require harsh reaction conditions or expensive transition metal catalysts, which can introduce difficult-to-remove impurities into the final product. Furthermore, the synthesis of the necessary dipolarophiles and dipoles often involves unstable intermediates that degrade rapidly, leading to inconsistent yields and batch-to-batch variability. In an industrial setting, these factors translate to increased operational expenditures and extended lead times for high-purity pharmaceutical intermediates. The reliance on chromatographic purification for every step further exacerbates the cost burden, making large-scale production economically unviable for many potential drug candidates.
The Novel Approach
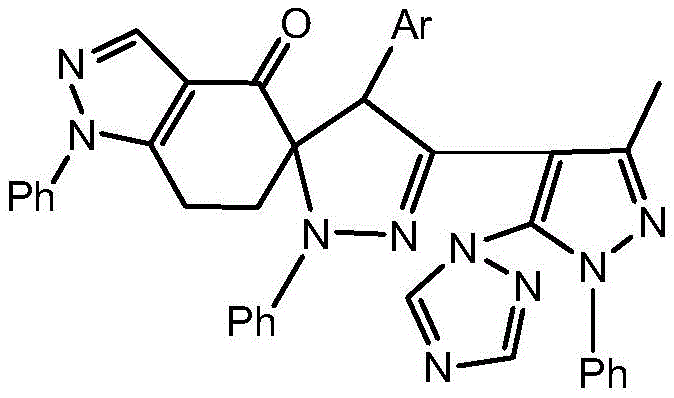
The methodology outlined in CN110655522A presents a streamlined alternative that circumvents these historical bottlenecks. By utilizing a specific 1,3-dipolar cycloaddition strategy, the process achieves excellent control over the stereochemistry and connectivity of the spiro center. The route begins with the efficient synthesis of a stable alpha-chloro hydrazone precursor, which serves as the nitrile imine dipole source upon treatment with base. This dipole then reacts seamlessly with an exocyclic double bond on the indazole-ketone framework. As illustrated in the reaction scheme below, the initial steps involve the preparation of the pyrazole-triazole hybrid aldehyde and its subsequent conversion to the hydrazone.
This modular approach allows for the independent optimization of the two main coupling partners before the final cyclization, ensuring that the commercial scale-up of complex polymer additives or pharmaceutical intermediates can be managed with greater predictability. The elimination of sensitive organometallic reagents simplifies the safety profile of the manufacturing process.
Mechanistic Insights into 1,3-Dipolar Cycloaddition
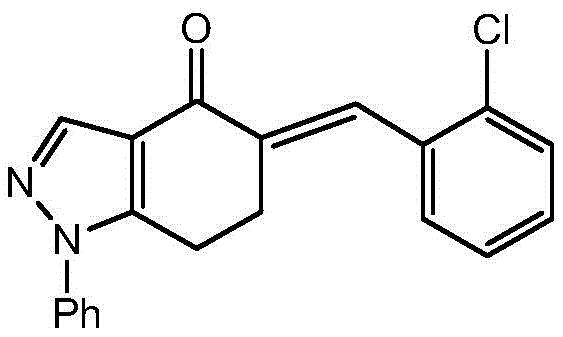
The core transformation in this synthesis is the 1,3-dipolar cycloaddition, a powerful tool for constructing five-membered heterocycles with high atom economy. In this specific embodiment, the alpha-chloro hydrazone derivative acts as a masked nitrile imine. Upon deprotonation and elimination of chloride, typically facilitated by a mild base like pyridine, the reactive 1,3-dipole is generated in situ. This dipole possesses a distinct electronic distribution that favors attack on the electron-deficient exocyclic double bond of the 5-(2-chlorobenzylidene)-1-phenyl-6,7-dihydro-1H-indazol-4(5H)-one. The presence of the ortho-chloro substituent on the benzylidene ring plays a crucial role; it not only modulates the electrophilicity of the dipolarophile but also enhances the lipophilicity of the final drug candidate, potentially improving its membrane permeability and bioavailability.
From an impurity control perspective, the mechanism offers inherent advantages. The concerted nature of the cycloaddition minimizes the formation of side products that typically arise from stepwise radical pathways. The subsequent aromatization or stabilization of the pyrazoline ring locks the spiro-configuration, preventing retro-cycloaddition under standard workup conditions. The preparation of the dipolarophile itself, shown in the following scheme, involves a straightforward aldol-type condensation between an indazole ketone and 2-chlorobenzaldehyde.
This step is highly robust, utilizing aqueous sodium hydroxide in ethanol, a solvent system that is both environmentally benign and easy to recover. The convergence of these two well-defined fragments in the final step ensures that the critical quality attributes of the final high-purity OLED material or pharmaceutical intermediate are maintained throughout the synthesis.
How to Synthesize Spiro[indazole-pyrazoline] Derivatives Efficiently
The synthesis of these valuable derivatives follows a logical five-step sequence that balances chemical complexity with operational simplicity. The process begins with the nucleophilic substitution of a chloropyrazole with 1,2,4-triazole, followed by hydrazone formation and controlled chlorination. The parallel preparation of the indazole ketone dipolarophile allows for flexible scheduling in a multipurpose plant. The final cycloaddition is performed under mild conditions, avoiding the need for high pressure or extreme temperatures. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Synthesize 1-phenyl-3-methyl-5-(1,2,4-triazolyl)-4-pyrazolecarboxaldehyde via nucleophilic substitution in DMSO.
- Convert the aldehyde to its phenylhydrazone derivative using phenylhydrazine in refluxing ethanol.
- Perform controlled chlorination with tert-butyl hypochlorite at -12°C to generate the alpha-chloro hydrazone dipole precursor.
- Prepare the dipolarophile, 5-(2-chlorobenzylidene)-1-phenyl-6,7-dihydro-1H-indazol-4(5H)-one, via base-catalyzed condensation.
- Execute the final 1,3-dipolar cycloaddition between the alpha-chloro hydrazone and the exocyclic double bond to form the spiro core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from a laboratory curiosity to a commercially viable ingredient hinges on cost, availability, and scalability. This patented process addresses these concerns directly by leveraging commodity chemicals and avoiding supply-chain-constrained reagents. The absence of precious metal catalysts such as palladium or rhodium eliminates the need for expensive scavenging resins and rigorous heavy metal testing, which are significant cost drivers in API manufacturing. Furthermore, the solvents employed—ethanol, DMSO, and petroleum ether—are globally available in bulk quantities, insulating the production schedule from regional supply shocks.
- Cost Reduction in Manufacturing: The primary economic driver of this process is the elimination of transition metal catalysts. In traditional cross-coupling approaches, the cost of the catalyst and the subsequent purification steps to meet residual metal specifications can account for a substantial portion of the COGS. By utilizing an organocatalytic or base-mediated cycloaddition, this route drastically reduces raw material costs. Additionally, the high selectivity of the reaction minimizes the loss of valuable intermediates to side reactions, improving the overall mass balance and yield. The use of simple recrystallization for purification, rather than preparative HPLC, further lowers the operational expenditure associated with solvent consumption and waste disposal.
- Enhanced Supply Chain Reliability: The starting materials, including 2-chlorobenzaldehyde and phenylhydrazine derivatives, are produced on a multi-ton scale by numerous global suppliers. This commoditization ensures that there is no single point of failure in the supply chain. Unlike specialized building blocks that may have long lead times or limited sources, these precursors can be sourced competitively, allowing for strategic stockpiling and just-in-time delivery models. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, reducing the risk of batch failures and ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling a 1,3-dipolar cycloaddition from grams to kilograms requires careful thermal management, but the exothermicity of this specific reaction is manageable with standard jacketed reactors. The process avoids the generation of hazardous gaseous byproducts, simplifying the venting and scrubbing requirements of the manufacturing facility. Moreover, the aqueous workup steps allow for the effective separation of inorganic salts, which can be treated in standard wastewater facilities. This alignment with green chemistry principles facilitates regulatory approval and reduces the environmental compliance burden, making it an attractive option for manufacturers aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro-compounds. The answers are derived from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is critical for evaluating the feasibility of integrating this chemistry into your existing pipeline.
Q: What are the key advantages of this 1,3-dipolar cycloaddition route for spiro compounds?
A: This route offers superior regioselectivity and avoids the use of expensive transition metal catalysts, significantly simplifying downstream purification and reducing heavy metal impurities in the final API intermediate.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The protocol utilizes precise temperature control (-12°C) during the critical chlorination step and employs recrystallization techniques, ensuring a clean impurity profile suitable for stringent pharmacopeial standards.
Q: Is this synthesis scalable for commercial production?
A: Yes, the reaction conditions utilize common industrial solvents like ethanol and DMSO, and the reagents are commercially available, making the transition from lab-scale to multi-ton production feasible without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[indazole-pyrazoline] Derivatives Supplier
The development of novel antitumor agents requires a partner who understands both the scientific intricacies and the commercial realities of fine chemical production. NINGBO INNO PHARMCHEM stands at the forefront of this industry, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of 1,3-dipolar cycloadditions, ensuring that your project moves from bench to plant without interruption. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for clinical and commercial applications.
We invite you to discuss how our expertise can accelerate your drug development timeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
