Advanced Manufacturing of Aryl Thioesters for Next-Gen Liquid Crystal Displays
Advanced Manufacturing of Aryl Thioesters for Next-Gen Liquid Crystal Displays
The rapid evolution of the display industry demands increasingly sophisticated materials, particularly within the realm of liquid crystal (LCD) technology where molecular precision dictates performance. Patent CN103664721A introduces a transformative methodology for the synthesis of aryl esters of thiocarboxylic acids, a critical class of intermediates used in the formulation of advanced liquid crystal materials. This innovation addresses long-standing challenges in organic synthesis by providing a route that combines high chemical efficiency with operational safety. By leveraging specific organometallic coupling strategies, the process enables the production of complex thioester scaffolds under remarkably mild conditions, avoiding the extreme temperatures and hazardous reagents that have historically plagued this sector. For stakeholders in the electronic chemical supply chain, this represents a pivotal shift towards more sustainable and reliable manufacturing protocols.
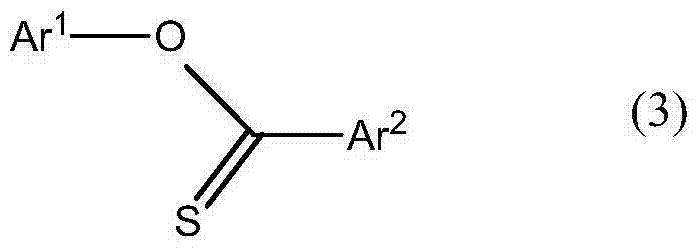
The core of this technological breakthrough lies in the strategic coupling of a halogenated thiocarbonyl compound with a specifically prepared arylated metal compound. Unlike conventional approaches that might struggle with selectivity or side reactions, this method ensures the formation of the desired carbon-sulfur bond with exceptional fidelity. The versatility of the process allows for a wide range of substituents on both the acyl and aryloxy portions of the molecule, granting chemists the flexibility to tune the physical properties of the final liquid crystal material. This adaptability is crucial for R&D teams aiming to optimize parameters such as birefringence and viscosity in next-generation display panels. Furthermore, the robustness of the reaction conditions suggests a high degree of reproducibility, a key metric for any reliable electronic chemical supplier aiming to secure long-term contracts with major panel manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl thioesters has been fraught with significant technical and safety hurdles that impede efficient commercial scale-up of complex electronic chemicals. Prior art methods, such as those involving the reaction of chlorothioformates with aromatic compounds or the treatment of carbanions with dithiocarbonates, often suffer from disappointingly low yields, leading to substantial material loss and increased waste disposal costs. More critically, several established pathways rely on the use of hydrogen sulfide, a gas with extreme toxicity that necessitates specialized containment infrastructure and rigorous safety protocols, thereby inflating capital expenditure. Other methods require reaction temperatures exceeding 100°C, which not only consumes excessive energy but also promotes the formation of unwanted by-products that complicate downstream purification. These inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for high-purity OLED material and LCD intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data offers a streamlined solution that bypasses these legacy issues through the use of relatively safe and accessible starting materials. By employing a halogenated thiocarbonyl electrophile and reacting it with an arylated metal species, the process achieves high yields without the need for dangerous gases or extreme thermal inputs. The reaction proceeds effectively in common proton-inert organic solvents like tetrahydrofuran (THF), which are readily available and easy to recover. This shift eliminates the need for high-pressure reactors or specialized gas scrubbing systems, significantly simplifying the plant engineering requirements. Moreover, the ability to operate at temperatures ranging from cryogenic levels up to merely 2°C allows for precise control over the reaction kinetics, minimizing thermal degradation and ensuring a cleaner crude product profile. This operational simplicity translates directly into cost reduction in liquid crystal material manufacturing by reducing both energy consumption and waste treatment burdens.
Mechanistic Insights into Organocuprate-Mediated Coupling
At the heart of this high-efficiency synthesis is a sophisticated understanding of organometallic reactivity, specifically the enhancement of nucleophilicity through transmetallation. The process typically begins with the generation of an arylated alkali metal compound, such as an aryllithium species, formed by treating an aryl halide or hydrogen derivative with an organolithium reagent. While this intermediate is reactive, the patent highlights a crucial optimization step: the optional conversion of this alkali metal species into an arylated copper compound. This transmetallation step involves reacting the aryllithium intermediate with a monovalent copper salt, such as copper chloride or copper cyanide. The resulting organocopper species exhibits superior chemoselectivity and reactivity towards the thiocarbonyl electrophile compared to its lithium counterpart. This mechanistic nuance is vital for R&D directors focused on purity, as it suppresses side reactions that could otherwise introduce difficult-to-remove impurities into the final batch.
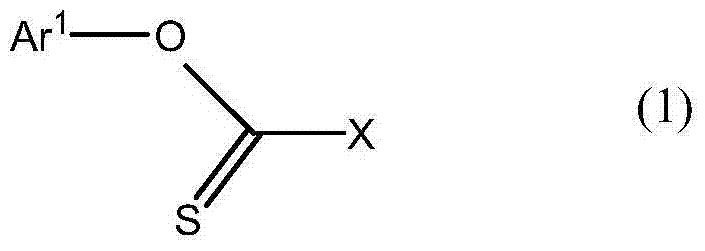
Furthermore, the presence of transition metal catalysts, specifically trivalent iron or divalent nickel compounds, can further augment the reaction efficiency when using direct organolithium coupling. The addition of catalytic amounts of species like tris(2,4-pentanedione)iron(III) facilitates the cross-coupling event, likely through a radical or oxidative addition-reductive elimination cycle that lowers the activation energy barrier. This catalytic enhancement ensures that even sterically hindered substrates can be coupled effectively, broadening the scope of accessible chemical space for material scientists. From an impurity control perspective, the mild conditions prevent the decomposition of sensitive functional groups that might be present on the aromatic rings, such as fluoro or cyano substituents which are common in liquid crystal mesogens. The result is a product with a narrow impurity profile, reducing the burden on purification steps like column chromatography or recrystallization and ensuring that the final material meets the stringent specifications required for display applications.
How to Synthesize Aryl Thioesters Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the organometallic coupling. The standard protocol involves dissolving the aryl precursor in an aprotic solvent and cooling the mixture significantly before the addition of the lithiating agent to prevent runaway exotherms. Once the arylated metal intermediate is formed, the introduction of the copper salt serves as a pivotal turning point in the reaction trajectory, converting a highly basic species into a softer nucleophile better suited for attacking the thiocarbonyl center. The subsequent addition of the halogenated thiocarbonyl compound must be managed to maintain the low-temperature regime, ensuring that the reaction proceeds smoothly without generating excessive heat that could degrade the product. Detailed standardized synthesis steps see the guide below for exact parameters regarding molar ratios and quenching procedures.
- Prepare the arylated metal compound by reacting an aryl halide or aryl hydrogen derivative with an organoalkali metal reagent like n-BuLi in an aprotic solvent such as THF at low temperatures.
- Optionally perform transmetallation by adding a monovalent copper salt to generate a more reactive aryl-copper species, which significantly enhances coupling efficiency.
- Add the halogenated thiocarbonyl electrophile to the reaction mixture, maintaining temperatures between -30°C and 2°C to ensure high selectivity and yield before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of toxic hydrogen sulfide from the manufacturing workflow removes a significant regulatory and safety liability, simplifying compliance with environmental health and safety (EHS) standards across different global jurisdictions. This reduction in hazard profile facilitates smoother logistics and storage of raw materials, as the supply chain no longer needs to accommodate high-risk gas cylinders or complex scrubbing infrastructure. Additionally, the use of common solvents and commercially available metal salts ensures that the raw material base is robust and less susceptible to market shortages, enhancing supply chain reliability for critical display components. The overall simplification of the process flow reduces the number of unit operations required, which in turn lowers the operational expenditure associated with labor and equipment maintenance.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in reaction yield and the elimination of expensive safety measures associated with toxic gases. By achieving high conversion rates under mild conditions, the process minimizes the loss of valuable starting materials, which is particularly important when dealing with complex, multi-substituted aromatic precursors. The avoidance of high-temperature heating cycles significantly reduces energy consumption per kilogram of product, contributing to a lower carbon footprint and reduced utility costs. Furthermore, the cleaner reaction profile means that less solvent and adsorbent are required during the purification stage, driving down the cost of goods sold (COGS) through reduced consumable usage. These factors combine to create a more economically viable production model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: The reliance on stable, solid, or liquid reagents rather than hazardous gases fundamentally de-risks the supply chain. Sourcing halogenated thiocarbonyl compounds and copper salts is straightforward from a wide network of chemical suppliers, preventing single-source bottlenecks. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures related to high-pressure or high-temperature systems. This stability allows for more accurate forecasting and inventory management, ensuring that downstream customers in the display industry receive their materials on time. The ability to scale this chemistry from gram to ton quantities without changing the fundamental reaction parameters provides a seamless path from pilot plant to full commercial production, securing long-term supply continuity.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the mild nature of this synthesis mitigates many typical scale-up risks. The exothermicity of the reaction is manageable even at large volumes due to the low operating temperatures, reducing the risk of thermal runaways that can shut down production lines. From an environmental standpoint, the absence of sulfur-containing gas emissions simplifies waste gas treatment, and the use of recyclable solvents aligns with green chemistry principles. This alignment with sustainability goals is increasingly important for multinational corporations seeking to reduce their environmental impact. The process generates less hazardous waste overall, lowering disposal costs and simplifying the permitting process for new manufacturing facilities, thereby accelerating time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aryl thioester synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The focus is on practical considerations such as reagent availability, safety protocols, and product quality assurance.
Q: Why is this new method safer than traditional thioester synthesis?
A: Traditional methods often rely on toxic hydrogen sulfide gas or require hazardous high-temperature conditions exceeding 100°C. This patented process utilizes stable halogenated thiocarbonyl compounds and operates at mild temperatures near room temperature or below, eliminating the need for specialized gas handling equipment and reducing thermal risks.
Q: How does the use of copper reagents impact the reaction yield?
A: The patent data demonstrates that converting the initial organolithium intermediate into an organocopper species prior to coupling drastically improves reaction efficiency. While direct coupling may result in moderate yields, the copper-mediated pathway consistently achieves high conversion rates, minimizing waste and maximizing output per batch.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for industrial viability. It uses common aprotic solvents like THF, avoids exotic catalysts, and operates under atmospheric pressure. The mild reaction conditions simplify temperature control requirements, making the scale-up from laboratory to commercial tonnage straightforward and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Thioester Supplier
As the demand for high-performance liquid crystal materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in advanced organic synthesis is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge methodologies like the one described in CN103664721A to deliver superior intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global display giants without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aryl thioester meets the exacting standards required for electronic applications. Our commitment to process excellence ensures that our clients receive materials that enable the highest performance in their final devices.
We invite procurement leaders and technical directors to engage with us to explore how this optimized synthesis route can benefit your specific supply chain needs. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current material requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project milestones. Let us be your partner in driving innovation and efficiency in the electronic chemical sector.
