Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
The synthesis of thioester compounds represents a critical capability in modern organic chemistry, particularly for the production of complex pharmaceutical intermediates and fine chemicals. These structural motifs serve as versatile acyl donors in native chemical ligation and are pivotal in the biosynthesis of valuable proteins and metabolic pathways involving acetyl-CoA. However, traditional synthetic routes have long been plagued by significant operational hazards and efficiency bottlenecks. A groundbreaking approach detailed in patent CN113004181A introduces a transformative methodology that leverages transition metal-catalyzed carbonylation to construct these valuable bonds with unprecedented safety and efficiency. This innovation shifts the paradigm from hazardous thiol-based couplings to a robust system utilizing readily available benzyl chlorides and sulfonyl chlorides. For R&D directors and procurement specialists alike, this technology offers a pathway to high-purity intermediates while drastically simplifying the supply chain logistics associated with noxious sulfur sources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of thioester compounds has relied heavily on the direct acylation of thiols with carboxylic acids or their activated derivatives, a process fraught with substantial industrial disadvantages. Thiols are notoriously malodorous, toxic, and prone to oxidative dimerization, creating severe challenges for worker safety and environmental compliance in large-scale manufacturing facilities. Furthermore, the sulfur atoms in free thiols possess a high affinity for transition metals, frequently leading to catalyst poisoning that necessitates excessive catalyst loading or results in stalled reactions with poor yields. Alternative methods such as the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often require harsh reaction conditions, stoichiometric oxidants, or generate significant amounts of waste byproducts. These legacy processes increase the cost of goods sold (COGS) through expensive waste treatment protocols and lower overall atom economy, making them increasingly unviable for the cost-sensitive production of agrochemical and pharmaceutical intermediates in a competitive global market.
The Novel Approach
The novel carbonylation strategy disclosed in the patent data fundamentally re-engineers the synthetic route by replacing problematic thiols with stable and odorless sulfonyl chlorides as the sulfur source. This method employs a sophisticated palladium-catalyzed system where benzyl chloride compounds react with sulfonyl chlorides in the presence of tungsten carbonyl, which uniquely serves as both the carbonyl source and the reducing agent. By eliminating the need for external reducing agents and gaseous carbon monoxide cylinders, the process significantly enhances operational safety and simplifies reactor setup. The reaction proceeds efficiently at moderate temperatures around 100°C in common organic solvents like acetonitrile, demonstrating exceptional functional group tolerance across a wide range of substrates. This approach not only mitigates the health risks associated with volatile sulfur compounds but also streamlines the purification process, allowing for the isolation of high-purity thioester products through standard filtration and chromatography techniques without complex downstream processing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this technological advancement lies in the intricate interplay between the palladium catalyst system and the dual-function tungsten carbonyl reagent. The reaction initiates with the reduction of the palladium(II) precursor, typically palladium acetate, to the active palladium(0) species by tungsten hexacarbonyl, which simultaneously releases carbon monoxide into the reaction medium. This in situ generation of CO ensures a steady concentration of the carbonylating agent without the safety hazards of high-pressure gas handling. The active Pd(0) complex then undergoes oxidative addition with the benzyl chloride substrate, forming a benzyl-palladium intermediate that is primed for carbonyl insertion. Subsequent coordination and insertion of the carbon monoxide generate an acyl-palladium species, which then reacts with the sulfonyl chloride. The presence of water and triethylamine plays a crucial promotional role, likely facilitating the activation of the sulfonyl chloride and stabilizing the transition states involved in the sulfur-carbon bond formation. This mechanistic pathway avoids the formation of free thiol intermediates entirely, thereby preventing catalyst deactivation and ensuring high turnover numbers even with electron-deficient or sterically hindered substrates.
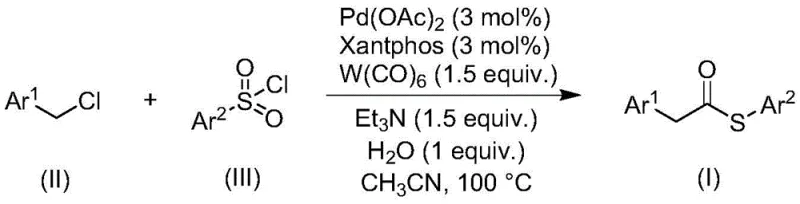
Impurity control in this system is inherently superior due to the specificity of the catalytic cycle and the stability of the starting materials. Unlike thiol-based methods where disulfide formation is a pervasive side reaction that complicates purification and lowers yield, this carbonylation route produces minimal sulfur-containing byproducts. The use of Xantphos as a bidentate ligand further enhances selectivity by stabilizing the palladium center and preventing the formation of palladium black, a common cause of reaction failure in carbonylation chemistry. The broad substrate scope allows for the incorporation of various substituents such as methyl, methoxy, halogens, and ester groups on both the benzyl and sulfonyl aromatic rings without significant loss in efficiency. This predictability in reaction outcome is vital for process chemists aiming to lock in a robust manufacturing route early in the drug development lifecycle, ensuring that the impurity profile remains consistent and manageable from gram-scale optimization to multi-ton commercial production.
How to Synthesize S-Aryl Thioesters Efficiently
Implementing this synthesis protocol requires precise attention to reagent ratios and reaction parameters to maximize yield and purity. The process begins by combining the palladium catalyst, ligand, tungsten carbonyl, base, and water with the specific benzyl chloride and sulfonyl chloride substrates in an anhydrous organic solvent. The reaction mixture is then heated to promote the carbonylation cascade, followed by a straightforward workup procedure involving filtration and silica gel treatment. While the general procedure is robust, optimizing the specific stoichiometry for new substrates may be necessary to achieve the highest possible efficiency. For detailed operational parameters and specific molar ratios validated across multiple examples, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten carbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in an organic solvent like acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 24 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this carbonylation technology offers compelling advantages that directly address the pain points of cost volatility and raw material availability in the fine chemical sector. By shifting away from thiols, manufacturers can bypass the complex logistics and premium pricing associated with handling hazardous, smelly sulfur reagents that often require specialized storage and transport infrastructure. The reliance on benzyl chlorides and sulfonyl chlorides leverages a vast existing supply chain of commodity chemicals, ensuring consistent availability and shielding production schedules from the disruptions often seen with niche reagents. Furthermore, the dual functionality of tungsten carbonyl reduces the total number of distinct raw materials required, simplifying inventory management and reducing the working capital tied up in reagent stock. This streamlined input profile translates into a more resilient supply chain capable of sustaining continuous manufacturing operations with reduced risk of stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of external reducing agents and the use of inexpensive palladium acetate combined with tungsten carbonyl significantly lowers the direct material costs per kilogram of product. The avoidance of thiols removes the need for expensive scrubbing systems and specialized personal protective equipment (PPE) required for toxic vapor containment, leading to substantial operational expenditure (OPEX) savings. Additionally, the high reaction efficiency and simplified workup reduce solvent consumption and waste disposal fees, contributing to a leaner and more cost-effective production model that enhances overall profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available benzyl chlorides and sulfonyl chlorides ensures that the production of these thioester intermediates is not bottlenecked by the supply of exotic or difficult-to-source reagents. The robustness of the reaction conditions allows for flexible scheduling and reduces the sensitivity to minor variations in raw material quality, which is critical for maintaining consistent output in a multi-vendor supply network. This reliability enables procurement managers to negotiate better long-term contracts and secure stable pricing, knowing that the underlying chemistry is not dependent on fragile or single-source supply lines that could jeopardize project timelines.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure with respect to gas inputs (due to in situ CO generation) and at moderate temperatures, making it highly amenable to scale-up in standard glass-lined or stainless steel reactors without requiring high-pressure autoclaves. The absence of volatile thiols greatly simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the risk of regulatory fines related to odor complaints or toxic emissions. This green chemistry profile aligns perfectly with the increasing sustainability mandates of global pharmaceutical clients, positioning suppliers who adopt this technology as preferred partners for eco-conscious manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific projects. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What is the primary advantage of using sulfonyl chlorides over thiols in this synthesis?
A: The use of sulfonyl chlorides eliminates the need for handling volatile, unpleasant-smelling, and toxic thiol compounds, which often poison catalysts and complicate industrial hygiene protocols.
Q: How does tungsten carbonyl function in this reaction system?
A: Tungsten carbonyl serves a dual role as both the carbon monoxide source for the carbonylation step and as a reducing agent to regenerate the active palladium(0) species, removing the need for external reductants.
Q: Is this method suitable for large-scale production of diverse thioesters?
A: Yes, the method demonstrates wide substrate tolerance for various aryl and heteroaryl groups, uses commercially available starting materials, and operates under relatively mild conditions suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Aryl Thioesters Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our technical team has extensively evaluated this palladium-catalyzed carbonylation route and confirmed its potential for delivering high-purity thioester intermediates with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis and advanced material applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in carbonylation chemistry can drive value and reliability in your supply chain.
