Scalable Synthesis of 5-Aminolevulinic Acid Hydrochloride via Meldrum's Acid Alkylation for Pharmaceutical Applications
The pharmaceutical landscape for photodynamic therapy (PDT) agents continues to evolve, driven by the critical demand for high-purity intermediates like 5-aminolevulinic acid hydrochloride (5-ALA HCl). As detailed in patent CN101462974A, a novel synthetic methodology has been developed that fundamentally shifts the production paradigm away from hazardous and low-yielding traditional routes. This innovation leverages the unique reactivity of propanedioic acid cyclic esters, commonly known as Meldrum's acid derivatives, to construct the carbon backbone of 5-ALA with exceptional precision. For R&D directors and procurement specialists alike, this represents a significant breakthrough in accessing reliable pharmaceutical intermediate supplier networks capable of delivering material that meets stringent purity specifications required for clinical applications such as the treatment of actinic keratosis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-ALA HCl has been plagued by significant technical barriers that compromise both cost efficiency and product quality. One of the most prevalent conventional strategies involves the direct bromination of levulinic acid, a pathway that suffers from severe regioselectivity issues. As illustrated in the reaction schemes of prior art, the bromination of levulinic acid inevitably generates a mixture of 5-bromo and 3-bromo isomers. Separating these isomers is notoriously difficult, often requiring energy-intensive high vacuum rectification or costly column chromatography, which drastically reduces the overall yield and increases the environmental footprint. Furthermore, the pungency and toxicity of the bromo-derivatives pose serious health risks to operators, complicating the safety profile of the manufacturing process.
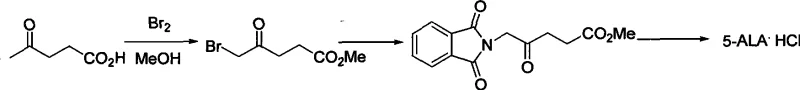
Alternative routes, such as those utilizing glycine or furfurylamine precursors, introduce their own set of complexities. Methods relying on phthalimide protection groups, while chemically sound on a small scale, involve multiple steps including protection, condensation, and harsh hydrolysis. These multi-step sequences accumulate impurities and result in lower overall throughput. Additionally, routes employing azide chemistry, while effective for nitrogen introduction, carry inherent safety risks due to the potential explosivity of organic azides, making them less desirable for large-scale commercial scale-up of complex pharmaceutical intermediates. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and prone to batch-to-batch variability.
The Novel Approach
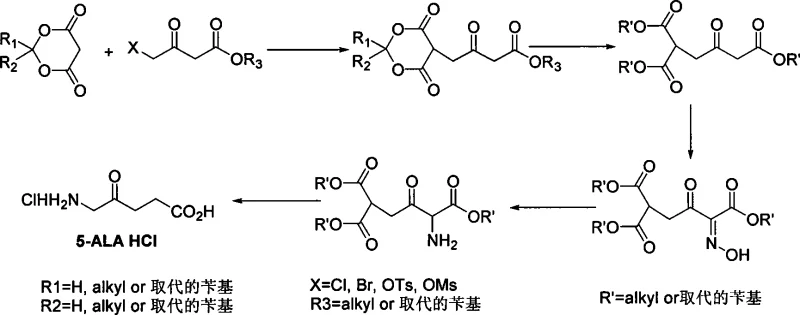
The inventive method disclosed in CN101462974A offers a robust alternative by utilizing a strategic alkylation of propanedioic acid cyclic esters. This approach bypasses the problematic direct functionalization of the levulinic acid backbone. Instead, it constructs the necessary carbon skeleton through the alkylation of the active methylene group in Meldrum's acid with a halo-keto ester. This reaction is highly selective and proceeds under relatively mild conditions. Following alkylation, the process employs a sequence of transesterification, oximation, and reduction to install the amino functionality. A key advantage of this route is that the intermediate products are predominantly solids, which facilitates purification through simple crystallization rather than complex distillation or chromatography. This shift from liquid to solid-phase intermediates is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it streamlines the isolation process and minimizes solvent consumption.
Mechanistic Insights into Meldrum's Acid Alkylation and Decarboxylation
The core of this synthetic strategy lies in the unique electronic properties of the propanedioic acid cyclic ester ring. The methylene protons at the C5 position of the Meldrum's acid ring are highly acidic due to the electron-withdrawing nature of the two flanking carbonyl groups and the ring strain. Upon treatment with a base, such as sodium methylate or potassium carbonate, a stable enolate is formed. This nucleophilic species attacks the electrophilic carbon of the halo-keto ester (where X is Cl, Br, OTs, or OMs) in a classic SN2 displacement. The resulting alkylated adduct retains the cyclic structure, which serves as a masked dicarboxylic acid equivalent. This masking effect is crucial because it prevents unwanted side reactions at the carboxyl positions during the subsequent transformation steps, effectively acting as a protecting group that is integral to the molecular framework.
The final transformation involves a hydrolytic decarboxylation that elegantly reveals the target 5-aminolevulinic acid structure. Under acidic hydrolysis conditions, typically involving reflux with hydrochloric acid, the cyclic ester ring opens to form the corresponding malonic acid derivative. Malonic acid derivatives are thermally unstable and readily undergo decarboxylation upon heating, losing a molecule of carbon dioxide to generate the mono-carboxylic acid chain. This spontaneous loss of CO2 drives the equilibrium forward, ensuring high conversion rates. Simultaneously, any protecting groups on the ester side chains are hydrolyzed, and the amine is protonated to form the stable hydrochloride salt. This cascade of hydrolysis and decarboxylation is highly efficient, minimizing the formation of byproducts and ensuring that the final high-purity pharmaceutical intermediate is obtained with a clean impurity profile, free from the isomeric contaminants that plague direct bromination routes.
How to Synthesize 5-Aminolevulinic Acid Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing 5-ALA HCl with high fidelity. The process begins with the alkylation of the cyclic ester in a polar aprotic solvent, followed by a transesterification step to adjust the ester groups for optimal solubility and reactivity in downstream steps. The introduction of the nitrogen atom is achieved via oximation using sodium nitrite, followed by a reduction step that can utilize either metal powders like zinc or catalytic hydrogenation. The final step is a rigorous hydrolysis that cleaves the ester and cyclic structures while forming the hydrochloride salt. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Perform alkylation reaction using propanedioic acid cyclic ester and a halo-keto ester derivative in the presence of a base.
- Execute transesterification followed by oximation with sodium nitrite to form the oxime intermediate.
- Conduct reduction using metal powder or catalytic hydrogenation, followed by hydrolysis and decarboxylation to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the purification train. By generating solid intermediates that can be purified via crystallization, the process eliminates the need for expensive and time-consuming column chromatography or high-vacuum distillation. This reduction in processing complexity translates directly into lower operational expenditures and shorter cycle times. Furthermore, the avoidance of hazardous reagents like organic azides and the mitigation of toxic bromo-derivative exposure significantly lowers the compliance burden and insurance costs associated with manufacturing, creating a more resilient and cost-effective supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of costly separation technologies. Traditional routes often suffer from low yields due to the difficulty in separating regioisomers, forcing manufacturers to process larger volumes of raw materials to achieve the same output. In contrast, the high selectivity of the Meldrum's acid alkylation ensures that a greater proportion of the starting material is converted into the desired product. Additionally, the use of commodity chemicals such as Meldrum's acid and simple halo-keto esters as starting materials avoids the price volatility associated with specialized reagents like potassium phthalimide. The ability to recycle solvents and the reduced need for exotic catalysts further contribute to substantial cost savings, making the final API intermediate more competitively priced in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturers, and this synthetic route enhances reliability by diversifying the raw material base. The key starting materials are bulk chemicals produced by multiple vendors globally, reducing the risk of supply bottlenecks that can occur with niche reagents. Moreover, the robustness of the reaction conditions—operating at moderate temperatures and pressures—means that the process is less susceptible to equipment failures or strict environmental controls that might halt production. The scalability of the crystallization steps ensures that production capacity can be ramped up quickly to meet surges in demand for photodynamic therapy drugs, providing a secure source of reliable pharmaceutical intermediate supplier capability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a significant improvement over legacy technologies. The avoidance of azide chemistry removes the risk of explosive incidents, a major concern in large-scale organic synthesis. Furthermore, the reduction in solvent usage and the elimination of heavy metal catalysts or difficult-to-treat waste streams align with modern green chemistry principles. The solid waste generated is minimal and manageable, and the aqueous waste streams are less contaminated with toxic organic halides compared to bromination routes. This cleaner profile facilitates easier permitting and regulatory approval, accelerating the time to market for new drug formulations and ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-ALA HCl using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, offering clarity on why this route is becoming the preferred choice for forward-thinking chemical manufacturers. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: What are the primary advantages of the Meldrum's acid route over traditional levulinic acid bromination?
A: The Meldrum's acid route avoids the formation of difficult-to-separate 3-bromo isomers common in direct levulinic acid bromination, significantly simplifying purification and improving overall yield without requiring column chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes classical reactions with readily available starting materials and produces solid intermediates that are easily purified by crystallization, making it highly amenable to industrial scale-up.
Q: How does this method address safety concerns associated with azide-based routes?
A: By utilizing an oximation-reduction sequence instead of azide substitution, the process eliminates the handling of explosive organic azides, thereby enhancing operational safety and reducing regulatory hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminolevulinic Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final therapeutic agent is inextricably linked to the purity of its precursors. Our technical team has extensively analyzed the synthetic pathways described in CN101462974A and has optimized them for commercial viability. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at the ppm level, guaranteeing that every batch of 5-ALA HCl we deliver supports the efficacy and safety of your photodynamic therapy formulations.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthetic technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development programs.
