Revolutionizing 5-Aminolevulinic Acid Production via a Novel Two-Step Synthetic Route
Introduction to Advanced 5-ALA Synthesis Technology
The pharmaceutical landscape for photodynamic therapy (PDT) agents is undergoing a significant transformation driven by the need for more efficient and safer manufacturing processes for key intermediates. Patent CN109796353B, published in April 2022, introduces a groundbreaking synthetic methodology for 5-aminolevulinic acid hydrochloride (5-ALA·HCl), a critical active pharmaceutical ingredient used in the diagnosis and treatment of various cancers including skin, bladder, and lung malignancies. This intellectual property represents a paradigm shift from the historically complex and hazardous multi-step syntheses toward a streamlined, two-step protocol that leverages the reactivity of succinic anhydride with deprotonated glycine derivatives. For R&D directors and process chemists, this patent offers a compelling solution to the long-standing challenges of yield optimization and impurity control, while simultaneously addressing the supply chain vulnerabilities associated with hazardous reagent sourcing. The technology underscores a move towards greener chemistry principles without compromising the stringent purity specifications required for clinical-grade intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 5-aminolevulinic acid has been plagued by inefficient reaction sequences that severely impact both economic viability and operational safety. Traditional routes, such as those originating from glycine via phthalic amidation or from furan methylamine through photooxidation, often necessitate four or more distinct chemical steps, each introducing potential yield losses and purification bottlenecks. More critically, several established methodologies rely on highly dangerous reagents; for instance, routes utilizing levulinic acid often require elemental bromine for alpha-bromination, posing severe corrosion and handling risks, while others employ sodium azide for nitrogen introduction, creating significant explosion hazards that complicate regulatory compliance and insurance logistics. Furthermore, methods involving transition metal catalysts like ruthenium or palladium introduce the risk of heavy metal contamination, necessitating expensive and time-consuming scavenging steps to meet pharmacopeial limits. These cumulative inefficiencies result in elevated production costs, extended lead times, and a fragile supply chain susceptible to disruptions in the availability of specialized catalysts or hazardous precursors.
The Novel Approach
In stark contrast to these legacy processes, the methodology disclosed in CN109796353B achieves the target molecule through a remarkably concise two-step sequence that bypasses the need for hazardous halogenation or explosive azide chemistry. The core innovation lies in the direct acylation of a protected glycine derivative (Compound A) with succinic anhydride under basic conditions, followed by a single acidic hydrolysis step to unveil the final amino-keto acid structure. This approach not only simplifies the operational workflow but also utilizes commodity chemicals that are abundantly available in the global fine chemical market, thereby insulating the production process from raw material volatility. By eliminating the need for transition metal catalysts and toxic halogens, the new route inherently produces a cleaner crude product profile, reducing the burden on downstream purification units. This strategic simplification translates directly into enhanced process robustness, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for high-volume output.
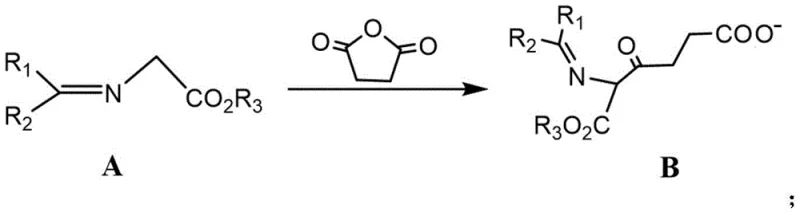
Mechanistic Insights into Base-Mediated Acylation and Hydrolysis
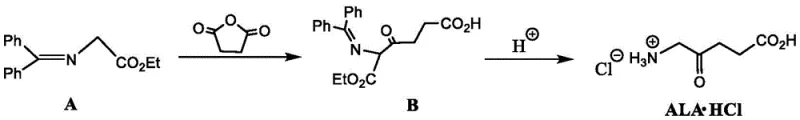
The chemical elegance of this synthesis is rooted in the precise control of nucleophilic reactivity during the initial carbon-carbon bond formation. In the first step, a strong base, such as sodium tert-butoxide, lithium diisopropylamide (LDA), or sodium hydride, is employed to quantitatively deprotonate the alpha-carbon of the glycine derivative (Compound A) in an aprotic polar solvent like DMF or THF at temperatures controlled below 0°C. This generates a highly reactive enolate species which subsequently attacks the electrophilic carbonyl carbon of succinic anhydride, inducing ring opening to form the keto-diester or keto-amide intermediate (Compound B). The choice of base and solvent is critical here, as it ensures high regioselectivity and prevents side reactions such as self-condensation or over-acylation, which are common pitfalls in less optimized protocols. The reaction is typically conducted under an inert atmosphere to prevent moisture interference, ensuring that the stoichiometry remains exact and the conversion to Intermediate B is maximized before proceeding to the next stage.
The second mechanistic phase involves the global deprotection and hydrolysis of Intermediate B under vigorous acidic conditions, typically using concentrated hydrochloric acid under reflux. This step serves a dual purpose: it hydrolyzes the ester or amide protecting groups (such as benzyl or phenyl moieties) attached to the nitrogen and oxygen atoms, and simultaneously converts the terminal carboxylic acid derivatives into their free acid forms. The acidic environment also facilitates the formation of the stable hydrochloride salt of the amine, which precipitates or can be crystallized from the reaction mixture upon cooling. This tandem deprotection-salt-formation strategy is highly efficient, as it avoids the need for separate neutralization and salt-formation steps often seen in other routes. The resulting crude product can be further purified via recrystallization from methanol-ethyl acetate mixtures, yielding 5-ALA·HCl with high chemical purity and minimal residual solvent content, satisfying the rigorous quality standards demanded by regulatory bodies for oncology therapeutics.
How to Synthesize 5-Aminolevulinic Acid Hydrochloride Efficiently
Implementing this novel synthetic route requires careful attention to temperature control and reagent addition rates to manage the exothermic nature of the deprotonation and acylation steps. The process begins with the dissolution of the glycine derivative in a dry polar aprotic solvent, followed by the slow addition of the base at sub-zero temperatures to generate the nucleophile. Once the enolate is formed, a solution of succinic anhydride is added dropwise, maintaining the low temperature to suppress side reactions before allowing the mixture to warm to room temperature for completion. The subsequent hydrolysis step is straightforward but requires robust equipment capable of handling hot concentrated acid, followed by standard workup procedures involving extraction, decolorization with activated carbon, and crystallization.
- Deprotonate a glycine derivative (Compound A) using a strong base like sodium tert-butoxide in an organic solvent such as DMF at low temperature.
- React the deprotonated intermediate with succinic anhydride to form the protected keto-acid intermediate (Compound B).
- Hydrolyze Compound B under reflux with concentrated hydrochloric acid to remove protecting groups and yield the final 5-ALA hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this two-step synthesis offers profound strategic benefits that extend far beyond simple chemical efficiency. The elimination of hazardous reagents like sodium azide and elemental bromine removes significant logistical hurdles, including the need for specialized storage facilities, hazardous waste disposal contracts, and complex transportation permits, thereby streamlining the entire inbound and outbound logistics framework. Moreover, the reliance on succinic anhydride and simple glycine derivatives as starting materials ensures a stable and diversified supply base, as these are high-volume commodity chemicals produced by multiple global vendors, reducing the risk of single-source dependency. This resilience is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients who cannot afford interruptions in their API supply chains.
- Cost Reduction in Manufacturing: The drastic reduction in synthetic steps from the traditional four-to-six steps down to merely two creates a compounding effect on cost savings throughout the production lifecycle. By removing intermediate isolation, drying, and purification stages, the process significantly lowers solvent consumption, energy usage for heating and cooling, and labor hours required for reactor turnover. Furthermore, the avoidance of expensive transition metal catalysts eliminates the capital expenditure associated with metal scavenging technologies and the loss of valuable product during filtration steps. These operational efficiencies translate into a substantially lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market for photodynamic therapy agents while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The simplicity of the raw material profile enhances supply chain reliability by mitigating the risks associated with the procurement of specialty chemicals that often have long lead times or volatile pricing. Since the key reagents are widely available bulk chemicals, procurement teams can leverage volume purchasing power to negotiate better terms and secure long-term supply agreements. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures and the need for costly rework. This stability ensures a consistent flow of high-purity 5-aminolevulinic acid to downstream customers, reinforcing the manufacturer's reputation as a dependable partner in the pharmaceutical value chain.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns perfectly with modern green chemistry initiatives and increasingly stringent regulatory frameworks. The absence of heavy metals and explosive byproducts simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility, lowering compliance costs and minimizing the risk of regulatory fines. The process is inherently scalable, as the reaction kinetics and heat transfer profiles are manageable in large-scale reactors without requiring exotic engineering solutions. This ease of scale-up allows manufacturers to rapidly respond to surges in market demand, expanding capacity from pilot plant levels to multi-ton commercial production with minimal capital investment in new infrastructure, thus future-proofing the supply capability against market growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its adoption for commercial production. These insights are derived directly from the experimental data and process descriptions within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: Unlike traditional methods that utilize hazardous reagents such as elemental bromine or explosive sodium azide, this novel route relies on stable succinic anhydride and standard strong bases, significantly reducing operational risks and regulatory burdens associated with hazardous material handling.
Q: How does the two-step process impact overall production costs?
A: By condensing the synthesis into only two chemical transformations from readily available starting materials, the process eliminates multiple isolation and purification stages required in longer routes, thereby drastically reducing solvent consumption, energy usage, and labor costs while improving overall yield.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild, utilizing common organic solvents like DMF or THF and standard temperature controls, which facilitates easy scale-up from laboratory benchtop to multi-ton commercial production without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminolevulinic Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel two-step synthesis in reshaping the economics of 5-ALA production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with advanced reactor systems capable of handling the specific temperature and solvent requirements of this route, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Contact us today to discuss how we can accelerate your project timelines and reduce your overall manufacturing costs through the adoption of this superior synthetic methodology.
