Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A recent technological breakthrough documented in patent CN113105402B introduces a highly efficient preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, specifically those incorporating trifluoromethyl groups. This innovation addresses long-standing challenges in heterocyclic chemistry by eliminating the reliance on toxic heavy metal catalysts and complex anhydrous conditions. The significance of this chemical class cannot be overstated, as the 1,2,4-triazole motif is a privileged structure found in numerous blockbuster drugs, including Maraviroc, Sitagliptin, and Deferasirox, which are utilized for treating HIV, diabetes, and iron overload respectively. By integrating a trifluoromethyl group into this scaffold, manufacturers can significantly enhance the metabolic stability, lipophilicity, and bioavailability of the final drug candidate, thereby improving therapeutic efficacy.
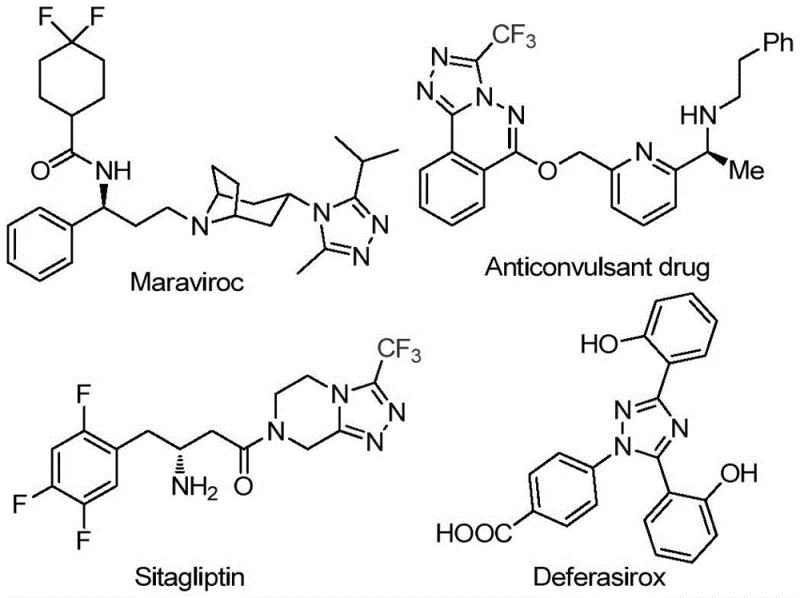
This novel methodology represents a paradigm shift for reliable pharmaceutical intermediate suppliers aiming to streamline their production pipelines. The process leverages a tandem reaction sequence involving iodine-promoted oxidation and cyclization in dimethyl sulfoxide (DMSO), offering a distinct advantage over conventional transition-metal catalyzed protocols. For R&D directors and process chemists, the ability to access these complex fluorinated heterocycles through a metal-free pathway simplifies purification workflows and reduces the risk of heavy metal contamination in the final API. Furthermore, the operational simplicity of the reaction, which proceeds under ambient atmospheric conditions rather than requiring inert gas protection, drastically lowers the barrier for technology transfer from laboratory discovery to commercial manufacturing. This report provides a deep technical analysis of the mechanism, operational parameters, and commercial implications of this invention for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those bearing both acyl and trifluoromethyl substituents, has been fraught with synthetic inefficiencies and environmental concerns. Traditional approaches often necessitate the use of expensive transition metal catalysts, such as copper or palladium complexes, which not only inflate the raw material costs but also introduce significant downstream processing burdens. The removal of trace heavy metals from the final product to meet stringent pharmacopeial limits requires additional purification steps, such as scavenging treatments or repeated recrystallizations, which inevitably erode the overall process yield. Moreover, many existing protocols demand rigorous anhydrous and oxygen-free environments, requiring specialized equipment like Schlenk lines or gloveboxes that are difficult to maintain on a multi-kilogram or tonne scale. These苛刻 conditions increase the operational complexity and safety risks associated with large-scale manufacturing, making cost reduction in API manufacturing a challenging objective for procurement teams managing tight budgets.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113105402B utilizes a non-metallic iodine promotion system that operates effectively in polar aprotic solvents like DMSO. This approach capitalizes on the dual functionality of iodine, which acts first as an oxidant to convert aryl ethyl ketones into reactive aryl diketones via a Kornblum-type oxidation, and subsequently as a promoter for the cyclization event. The reaction tolerates a wide range of functional groups on both the hydrazide and ketone substrates, allowing for the rapid generation of diverse chemical libraries without the need for protecting group strategies. By avoiding heavy metals entirely, the process inherently produces a cleaner crude reaction profile, minimizing the formation of metal-complexed impurities that are notoriously difficult to separate. This streamlined workflow not only accelerates the timeline for process development but also aligns perfectly with modern green chemistry principles, offering substantial cost savings through reduced waste generation and simplified regulatory compliance for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic innovation lies in a sophisticated tandem reaction mechanism that seamlessly integrates oxidation and heterocyclization into a single pot. The process initiates with the reaction of an aryl ethyl ketone with elemental iodine in DMSO at temperatures between 90 °C and 110 °C. Under these thermal conditions, the iodine facilitates the alpha-iodination of the ketone, followed by nucleophilic substitution by DMSO and subsequent elimination to generate an aryl 1,2-diketone intermediate in situ. This oxidative transformation is critical as it installs the necessary carbonyl functionality required for the subsequent ring closure. Once the diketone species is formed, the addition of trifluoroethylimide hydrazide triggers a condensation reaction, likely proceeding through a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine serves to buffer the reaction medium and scavenge the hydrogen iodide byproduct, driving the equilibrium forward. Finally, the intramolecular cyclization occurs under continued heating at 110 °C to 130 °C, where the nucleophilic nitrogen attacks the adjacent carbonyl carbon, closing the five-membered triazole ring and expelling water to yield the final 3,4,5-trisubstituted product.
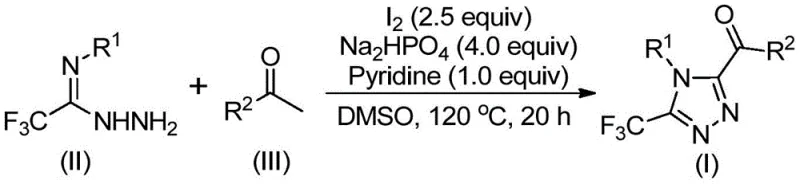
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. The absence of transition metals eliminates the risk of metal-induced side reactions, such as homocoupling or over-oxidation, which often plague conventional syntheses. The use of stoichiometric iodine and base ensures that the reaction proceeds through well-defined ionic or polar intermediates, resulting in a predictable impurity profile that is easier to characterize and control. Furthermore, the high functional group tolerance observed in the substrate scope suggests that the reaction conditions are mild enough to preserve sensitive moieties like halogens, ethers, and esters, which might otherwise be degraded under harsher catalytic conditions. This mechanistic robustness is paramount for R&D teams focused on developing high-purity pharmaceutical intermediates, as it ensures batch-to-batch consistency and minimizes the risk of unexpected genotoxic impurities forming during the manufacturing process.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
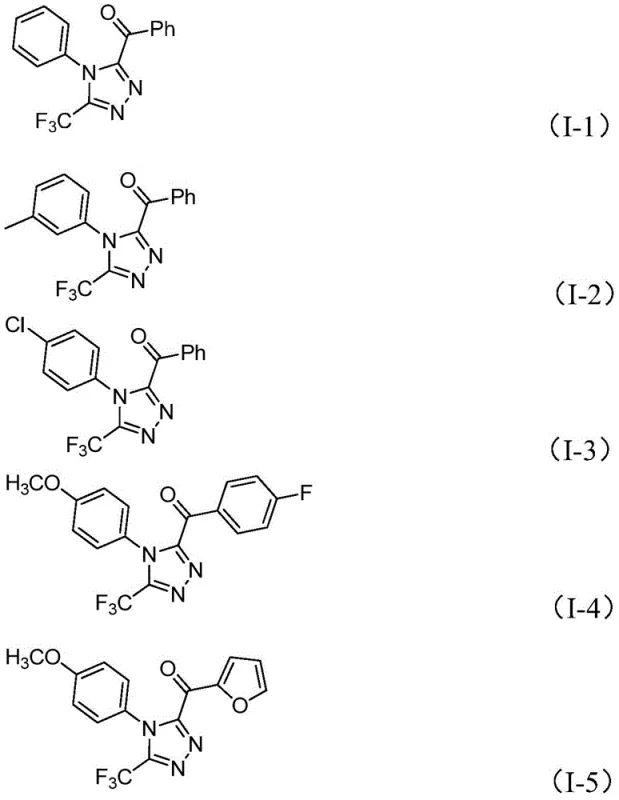
The practical execution of this synthesis is designed for maximum operational ease, making it highly attractive for process chemists looking to implement new routes quickly. The standard protocol involves a two-stage heating sequence in a single reaction vessel, eliminating the need for intermediate isolation of the unstable diketone species. Typically, the aryl ketone and a portion of iodine are heated in DMSO, followed by the sequential addition of the hydrazide, base, and remaining iodine. The reaction is then maintained at elevated temperatures for an extended period to ensure complete conversion. Detailed standardized operating procedures, including precise stoichiometric ratios and workup instructions, are essential for reproducing the high yields reported in the patent data. For a comprehensive guide on the specific molar equivalents and purification techniques required to achieve optimal results, please refer to the technical documentation below.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110 °C for 4-6 hours to initiate Kornblum oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture.
- Heat the mixture to 110-130 °C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The primary driver of value is the drastic simplification of the raw material portfolio; the process relies on aryl ethyl ketones and trifluoroethylimide hydrazides, which are commodity chemicals available from multiple global vendors. This abundance ensures a resilient supply chain that is less susceptible to the bottlenecks often associated with specialized organometallic reagents or custom-synthesized ligands. Additionally, the elimination of heavy metal catalysts removes the need for expensive metal scavengers and the associated disposal costs for hazardous metal waste, leading to significant operational expenditure reductions. The ability to run the reaction without strict inert atmosphere protection further lowers capital expenditure requirements, as standard glass-lined reactors can be utilized instead of specialized pressure vessels equipped for nitrogen or argon purging.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the replacement of costly transition metal catalysts with inexpensive elemental iodine. Since iodine is a bulk commodity chemical, its price volatility is significantly lower than that of precious metals like palladium or rhodium, providing greater budget predictability for long-term production contracts. Furthermore, the simplified downstream processing—specifically the absence of metal removal steps—reduces the consumption of silica gel, solvents, and labor hours required for purification. This cumulative effect results in a leaner manufacturing process with a lower cost of goods sold (COGS), allowing pharmaceutical companies to maintain healthier margins even in competitive generic markets. The high atom economy of the tandem reaction also means less waste is generated per kilogram of product, further reducing waste treatment fees.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of key starting materials, and this synthesis excels by utilizing widely accessible substrates. Aryl ketones and hydrazides are produced on a massive scale for various industries, ensuring that supply disruptions are rare and lead times are short. This reliability is crucial for meeting the just-in-time delivery demands of modern pharmaceutical manufacturing, where delays in intermediate supply can halt entire production lines. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, reducing the frequency of batch failures. This stability allows supply chain planners to optimize inventory levels with greater confidence, knowing that the manufacturing process is forgiving and consistent.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this method is explicitly designed for easy expansion. The patent data confirms successful execution on gram scales with straightforward exotherms that can be managed in standard reactor configurations. From an environmental standpoint, the avoidance of toxic heavy metals aligns perfectly with increasingly stringent global regulations regarding residual metals in drug substances. This compliance advantage accelerates the regulatory filing process, as there is no need to validate complex metal clearance studies. Additionally, the use of DMSO, a solvent with a favorable safety profile and high boiling point, facilitates safer handling at high temperatures compared to volatile organic solvents, enhancing overall plant safety and reducing the risk of solvent-related incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent literature, providing clarity on reaction conditions, substrate scope, and purification requirements. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The responses cover critical aspects such as catalyst loading, solvent selection, and the versatility of the method across different substitution patterns.
Q: What catalyst is used in this triazole synthesis method?
A: The process utilizes elemental iodine as a non-metallic promoter, avoiding the need for expensive or toxic transition metal catalysts typically found in traditional cyclization reactions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded from gram-level laboratory synthesis to industrial scale production due to its simple operation and lack of stringent anhydrous or oxygen-free requirements.
Q: What are the key starting materials required?
A: The synthesis relies on commercially available and inexpensive aryl ethyl ketones and trifluoroethylimide hydrazides, which ensures a stable and cost-effective supply chain for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with an experienced CDMO partner is essential for navigating the complexities of process development and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of detecting impurities at ppm levels, guaranteeing that all delivered materials meet stringent purity specifications required by major regulatory agencies. We understand the critical nature of supply chain security and are committed to providing a stable, high-quality source of these valuable intermediates to support your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative iodine-promoted synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this metal-free route for your specific molecule. We encourage you to contact us today to obtain specific COA data for our existing catalog of triazole derivatives or to initiate a discussion on route feasibility assessments for your proprietary compounds. Let us help you optimize your supply chain and accelerate your path to market with our advanced synthetic capabilities.
