Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, which serve as critical scaffolds in numerous bioactive molecules. Recent advancements documented in patent CN113105402B highlight a transformative preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses longstanding challenges in process chemistry. This innovation is particularly relevant for the production of high-value active pharmaceutical ingredients (APIs) such as antiviral and antidiabetic agents, where structural integrity and purity are paramount. The significance of this chemical class is underscored by its presence in major drugs like Maraviroc and Sitagliptin, demonstrating the broad therapeutic potential of this core structure. By leveraging a non-metal promoted pathway, this technology offers a compelling alternative to traditional methods that often rely on scarce resources.
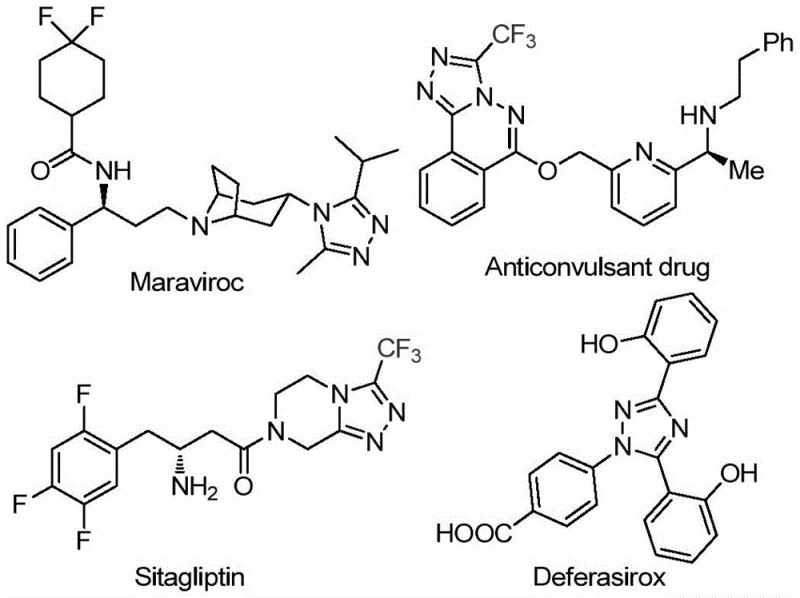
For procurement managers and supply chain directors, the implications of adopting this synthesis route extend far beyond the laboratory bench, offering a strategic advantage in cost reduction in pharmaceutical intermediates manufacturing. The elimination of heavy metal catalysts not only reduces the direct cost of reagents but also drastically simplifies the downstream purification process, which is often a bottleneck in commercial production. Furthermore, the use of dimethyl sulfoxide (DMSO) as a solvent and elemental iodine as a promoter utilizes commodity chemicals that are globally accessible, thereby mitigating supply chain risks associated with specialized reagents. As a reliable pharmaceutical intermediates supplier, understanding these process efficiencies allows us to offer more competitive pricing models while maintaining rigorous quality standards. The ability to operate without stringent anhydrous or oxygen-free conditions further lowers the barrier for entry for manufacturing partners, facilitating faster technology transfer and scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has been plagued by reliance on transition metal catalysts, which introduce significant complexity and cost into the manufacturing workflow. Traditional pathways often require expensive palladium or copper complexes that necessitate rigorous removal steps to meet regulatory limits for residual metals in final drug substances. These purification stages, typically involving specialized scavengers or extensive chromatography, increase both the processing time and the overall environmental footprint of the production cycle. Additionally, many conventional methods demand strictly anhydrous and anaerobic environments, requiring specialized equipment and inert gas manifolds that drive up capital expenditure and operational overhead. The sensitivity of these reactions to moisture and oxygen often leads to batch-to-batch variability, complicating quality control efforts and potentially resulting in lower overall yields. For large-scale operations, these factors combine to create a fragile supply chain that is vulnerable to disruptions and cost overruns.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105402B introduces a streamlined, iodine-promoted protocol that circumvents the need for toxic heavy metals while maintaining high efficiency and selectivity. This novel approach utilizes a tandem reaction sequence where aryl ethanones undergo iodination and Kornblum oxidation in situ, followed by condensation with trifluoroethylimide hydrazides to form the target triazole ring. The operational simplicity is remarkable, as the reaction proceeds effectively in air without the need for glovebox techniques or dried solvents, making it highly amenable to standard reactor setups. The versatility of this method is evidenced by its tolerance for a wide range of functional groups, allowing for the synthesis of diverse derivatives including those with electron-withdrawing or electron-donating substituents. This flexibility is crucial for medicinal chemists aiming to explore structure-activity relationships without being constrained by synthetic limitations.
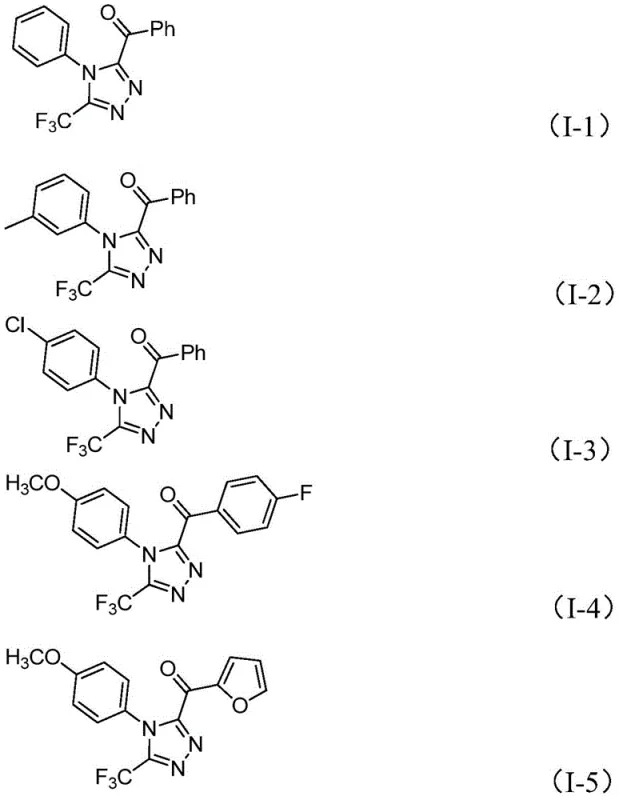
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation steps driven by the unique reactivity of iodine in dimethyl sulfoxide. Initially, the aryl ethanone substrate undergoes alpha-iodination facilitated by molecular iodine, generating an reactive intermediate that is subsequently oxidized by DMSO to form an aryl diketone species. This oxidative step is critical as it activates the carbonyl carbon for nucleophilic attack, setting the stage for the subsequent ring-closing event. The introduction of trifluoroethylimide hydrazide then triggers a dehydration condensation, forming a hydrazone intermediate that possesses the necessary connectivity for cyclization. Under the influence of the base (pyridine) and continued heating, an intramolecular nucleophilic attack occurs, closing the five-membered triazole ring and expelling water as the only byproduct. This elegant mechanism avoids the formation of complex organometallic intermediates, thereby reducing the risk of side reactions and impurity generation.
From a quality control perspective, the impurity profile of this reaction is significantly cleaner compared to metal-catalyzed alternatives, primarily due to the absence of metal-ligand complexes that can degrade or participate in off-cycle reactions. The use of sodium dihydrogen phosphate as a buffer helps maintain a stable pH environment throughout the prolonged heating period, preventing the decomposition of sensitive functional groups such as the trifluoromethyl moiety. Analytical data from the patent examples indicates high purity levels achievable through standard workup procedures like filtration and silica gel chromatography, without the need for preparative HPLC in many cases. For R&D directors, this means that the high-purity 1,2,4-triazole derivatives produced via this route are well-suited for direct use in subsequent coupling reactions or formulation studies. The robustness of the mechanism ensures that scaling the reaction does not disproportionately increase impurity levels, a common pitfall in process chemistry.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
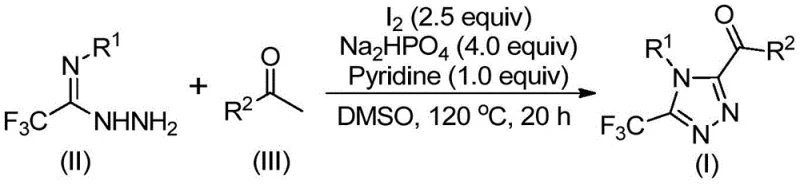
Implementing this synthesis route in a production environment requires careful attention to the stoichiometry and thermal profile outlined in the patent data to ensure optimal conversion and yield. The process begins with the precise mixing of aryl ethanone and elemental iodine in DMSO, followed by a controlled heating phase to generate the oxidized intermediate before introducing the hydrazide component. Detailed standard operating procedures regarding addition rates, temperature ramping, and quenching protocols are essential to replicate the success seen in the laboratory examples consistently. Operators must be trained to monitor the reaction progress, potentially using TLC or HPLC, to determine the exact endpoint of the cyclization step which typically occurs between 12 to 20 hours. The following section outlines the standardized steps derived directly from the intellectual property to guide your technical team in adopting this efficient methodology.
- Mix aryl ethanone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate iodination.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
The economic and logistical benefits of adopting this iodine-promoted synthesis route are substantial, offering a clear value proposition for stakeholders focused on margin improvement and supply security. By removing the dependency on precious metal catalysts, manufacturers can achieve significant cost savings not only in raw material procurement but also in waste disposal and environmental compliance fees associated with heavy metal effluent. The use of commodity solvents like DMSO and readily available aryl ketones ensures that the supply chain remains resilient against market fluctuations that often affect specialized reagents. Furthermore, the simplified workup procedure reduces the consumption of silica gel and eluents during purification, contributing to a lower overall cost of goods sold (COGS). For supply chain heads, the ability to source starting materials from multiple global vendors reduces the risk of single-source bottlenecks, ensuring enhanced supply chain reliability even during periods of geopolitical instability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper removes a major cost driver from the bill of materials, while simultaneously reducing the need for costly metal scavenging resins during purification. The reaction conditions allow for the use of technical grade solvents rather than anhydrous grades, further lowering input costs without compromising reaction efficiency. Additionally, the high atom economy of the cyclization step minimizes waste generation, leading to reduced disposal costs and a smaller environmental footprint. These cumulative effects result in a markedly more economical process that improves the overall profitability of the final API.
- Enhanced Supply Chain Reliability: The starting materials, including aryl ethanones and elemental iodine, are bulk chemicals produced by numerous suppliers worldwide, ensuring a stable and continuous supply flow. Unlike specialized ligands or catalysts that may have long lead times, these commodities can often be sourced with reduced lead time for high-purity pharmaceutical intermediates, allowing for just-in-time manufacturing strategies. The robustness of the reaction to ambient conditions means that storage and handling requirements are less stringent, reducing the risk of material degradation during transit. This reliability is critical for maintaining production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The protocol is explicitly designed for commercial scale-up of complex heterocyclic compounds, having been demonstrated to work efficiently from gram to multi-kilogram scales without loss of performance. The absence of toxic heavy metals simplifies the regulatory approval process for the manufacturing site, as there is no need for extensive validation of metal clearance steps. Waste streams are easier to treat since they primarily contain organic residues and iodine salts, which are less hazardous than organometallic waste. This alignment with green chemistry principles enhances the sustainability profile of the manufacturing operation, appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of answers based on the specific data points and claims found within the patent documentation. These responses clarify the operational parameters, safety considerations, and scalability potential of the iodine-promoted route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production pipelines. The following questions cover the most critical aspects of the process, from catalyst loading to purification strategies, providing a comprehensive overview for decision-makers.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in CN113105402B utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly reducing raw material costs and simplifying post-reaction metal removal processes.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) without strict anhydrous or oxygen-free conditions. It involves a two-stage heating process: initially at 90-110°C for iodination, followed by 110-130°C for cyclization.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states that the method can be easily expanded from gram-level laboratory synthesis to industrial scale production due to the use of cheap, readily available starting materials and simple operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate innovative patent methodologies like CN113105402B into reliable commercial supply chains for our global partners. Our engineering team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications across all our manufacturing lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the 3,4,5-trisubstituted 1,2,4-triazole intermediates we supply meet the exacting requirements of modern drug development programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements and budget constraints. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this metal-free protocol for your API manufacturing needs. We encourage you to contact us today to obtain specific COA data for our available triazole derivatives and to discuss route feasibility assessments tailored to your target molecules. Let us partner with you to drive innovation and efficiency in your pharmaceutical supply chain.
