Revolutionizing Thioamide Production: A Green Commercial Scale-Up Strategy
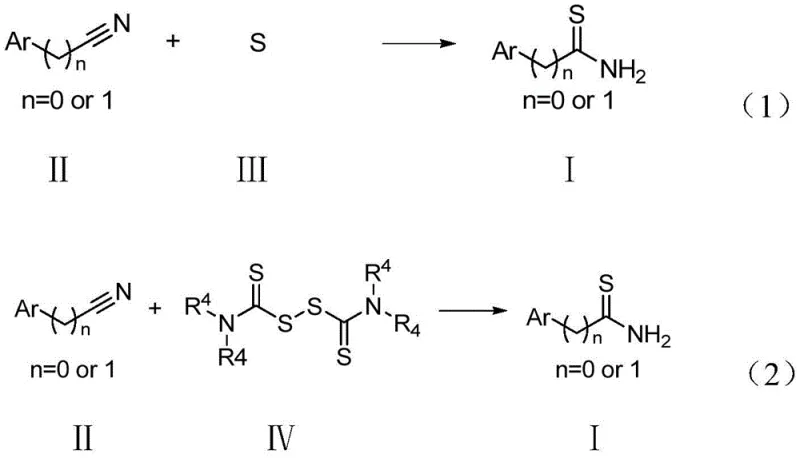
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, particularly in the synthesis of high-value nitrogen-sulfur containing scaffolds. Patent CN110981850A introduces a groundbreaking green method for preparing thioamide compounds, addressing critical bottlenecks in both pharmaceutical and agrochemical intermediate production. This technology leverages a novel oxidative thioamidation strategy that replaces hazardous traditional sulfuring agents with benign elemental sulfur or thiuram. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize supply chains while adhering to increasingly stringent environmental regulations. The core innovation lies in the use of an iodine reagent and oxidant system to activate the sulfur source, enabling a direct one-step conversion of nitriles to thioamides under mild conditions. This report analyzes the technical feasibility and commercial implications of adopting this protocol for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamides has relied heavily on sulfurization reagents such as Lawesson's reagent or phosphorus pentasulfide (P2S5), which pose severe operational and environmental challenges. These conventional reagents are not only expensive to procure but also generate substantial quantities of phosphorus-containing wastewater that requires costly treatment before disposal. Furthermore, alternative methods using hydrogen sulfide (H2S) gas involve significant safety risks due to its high toxicity and flammability, necessitating specialized closed-system equipment that increases capital expenditure. The low atomic utilization of sulfur in these traditional pathways often leads to poor yields and complex purification processes, resulting in higher overall production costs. For supply chain heads, the reliance on such hazardous materials introduces volatility in sourcing and potential regulatory shutdowns, making long-term planning difficult. Consequently, there is an urgent industrial demand for a safer, more cost-effective alternative that does not compromise on yield or purity standards.
The Novel Approach
The methodology disclosed in CN110981850A offers a transformative solution by utilizing elemental sulfur or thiuram as the sulfur source, which are abundant, non-toxic, and economically superior raw materials. This approach operates under mild reaction conditions, typically between 70°C and 140°C, using common organic solvents mixed with water, thereby drastically simplifying the reaction setup. The introduction of an iodine reagent and oxidant catalyst system activates the inert sulfur, allowing for efficient conversion without the need for extreme temperatures or pressures. This green chemistry protocol significantly reduces the environmental footprint by eliminating phosphorus waste and avoiding toxic gases, aligning perfectly with modern sustainability goals. From a commercial perspective, the simplicity of the operation translates to reduced processing time and lower energy consumption, providing a robust foundation for cost reduction in fine chemical manufacturing. The broad substrate scope ensures that this method can be applied to a wide range of cyano-containing compounds, enhancing its versatility for diverse product portfolios.
Mechanistic Insights into Oxidative Thioamidation
The core of this technological advancement lies in the synergistic catalytic cycle driven by the iodine-oxidant system, which facilitates the nucleophilic attack of sulfur species on the cyano group. Mechanistically, the oxidant, such as potassium persulfate, generates reactive radical species or activates the iodine reagent to form an electrophilic sulfur intermediate in situ. This activated sulfur species then reacts selectively with the sp-hybridized carbon of the nitrile group, initiating the formation of the carbon-sulfur bond essential for thioamide structure. The presence of water in the solvent system plays a crucial role in stabilizing intermediates and facilitating proton transfer steps, ensuring high conversion rates even for sterically hindered substrates. Understanding this mechanism is vital for R&D teams aiming to adapt this process for new molecule entities, as it highlights the tolerance for various functional groups including halogens and esters. The reaction pathway avoids the formation of heavy metal byproducts, which is a common issue in transition-metal catalyzed sulfurations, thus simplifying the downstream purification workflow.
Impurity control is another critical aspect where this mechanism excels, as the selective nature of the oxidative system minimizes side reactions such as over-oxidation or polymerization. Traditional methods often suffer from the formation of disulfides or other sulfur-containing impurities that are difficult to separate from the target thioamide. In contrast, the controlled release of active sulfur species in this patent ensures that the reaction proceeds cleanly towards the desired mono-thioamide product. High-performance liquid chromatography (HPLC) data from the patent examples consistently show purity levels exceeding 99%, indicating a very clean reaction profile. This high level of purity is essential for pharmaceutical applications where strict impurity thresholds must be met to ensure patient safety and regulatory approval. By minimizing the generation of hard-to-remove impurities, manufacturers can reduce the number of purification steps, leading to significant improvements in overall process efficiency and yield retention.
How to Synthesize Thioamide Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric ratios of the oxidant and iodine reagent to ensure optimal catalytic activity. The patent specifies a molar ratio range for the compound to sulfurizing agent of 1:1 to 1:4, allowing flexibility based on the specific reactivity of the substrate. Operators should maintain the reaction temperature within the specified 70°C to 140°C window to balance reaction rate and selectivity, with 120°C often yielding the best results for aromatic nitriles. Post-reaction workup involves standard extraction and silica gel column chromatography, techniques that are well-established in most manufacturing facilities, ensuring easy technology transfer. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the cyano-containing substrate with elemental sulfur or thiuram in a solvent system such as DMF and water.
- Add the catalytic system consisting of an oxidant like potassium persulfate and an iodine reagent such as potassium iodide to the vessel.
- Heat the reaction mixture to a temperature between 70°C and 140°C, stir for approximately 12 hours, and purify the resulting solid via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this green synthesis method offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain, as elemental sulfur and thiuram are commodity chemicals with stable pricing and high availability globally. Unlike specialized phosphorus reagents that may face supply constraints or price volatility, these sulfur sources ensure a reliable agrochemical intermediate supplier base that can support long-term production contracts. The elimination of toxic gas handling also reduces the need for expensive safety infrastructure and insurance premiums, contributing to substantial cost savings in facility operations. Furthermore, the high atom economy of the reaction means less waste generation, which directly lowers the costs associated with waste disposal and environmental compliance monitoring.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous sulfurizing agents with cheap elemental sulfur fundamentally alters the cost structure of thioamide production. By removing the need for complex reagent synthesis and reducing the quantity of waste generated, the overall variable cost per kilogram of product is significantly lowered. This economic efficiency allows companies to maintain competitive pricing margins even in fluctuating market conditions. Additionally, the simplified purification process reduces solvent consumption and labor hours required for chromatography, further enhancing the economic viability of the process. These factors combine to create a robust business case for switching to this greener methodology without compromising on product quality.
- Enhanced Supply Chain Reliability: Utilizing widely available raw materials mitigates the risk of supply disruptions that often plague specialty chemical manufacturing. Since elemental sulfur and common oxidants like potassium persulfate are produced on a massive industrial scale, securing consistent supply is far easier than sourcing niche reagents. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for global clients. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, adding another layer of security to the supply chain. Consequently, manufacturers can offer more reliable lead times and build stronger trust with their downstream partners.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gases make this process inherently safer and easier to scale from pilot plant to full commercial production. Facilities do not require specialized containment systems for hazardous gases, reducing the capital investment needed for scale-up. Moreover, the green nature of the process aligns with increasingly strict environmental regulations, future-proofing the manufacturing site against potential legislative changes. This compliance advantage is particularly valuable for companies operating in regions with rigorous environmental oversight, ensuring uninterrupted operations. The ability to scale up complex polymer additives or pharmaceutical intermediates using this method provides a strategic edge in the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green thioamide synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They are intended to provide clarity on the process capabilities and its suitability for various industrial applications. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing lines.
Q: What are the primary advantages of this thioamide synthesis method over conventional Lawesson's reagent protocols?
A: This method utilizes elemental sulfur or thiuram, which are significantly cheaper and less toxic than Lawesson's reagent or P2S5. It eliminates the generation of large amounts of phosphorus-containing wastewater, aligning with green chemistry principles and reducing environmental compliance costs.
Q: Is this process suitable for complex heterocyclic substrates found in agrochemical intermediates?
A: Yes, the patent data demonstrates broad substrate scope, successfully converting various cyano-containing heterocycles including pyridines, pyrimidines, and indoles. The mild reaction conditions preserve sensitive functional groups, making it ideal for complex agrochemical intermediate manufacturing.
Q: How does the iodine-oxidant system impact the purity profile of the final thioamide product?
A: The catalytic system facilitates a clean conversion with high atom utilization. Experimental data indicates purity levels exceeding 99% HPLC after standard purification, minimizing the need for extensive recrystallization and ensuring a cleaner impurity profile for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthesis route for the production of high-purity thioamide derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from lab bench to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical and agrochemical companies. We are committed to leveraging this innovative technology to deliver cost-effective and environmentally responsible solutions for our global clientele. Our team of experts is ready to assist in optimizing the reaction parameters to suit your specific substrate requirements.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio and reduce your manufacturing footprint. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the viability of this process for your target molecules. Let us help you engineer a more sustainable and profitable supply chain together.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
