Advanced Synthetic Route for High-Purity Resveratrol Manufacturing
The pharmaceutical and nutraceutical industries have long recognized the immense therapeutic potential of Resveratrol (CAS 501-36-0), a potent polyphenolic phytoalexin known for its antioxidant and anti-aging properties. However, securing a reliable supply of high-purity Resveratrol has historically been constrained by the limitations of natural extraction and complex synthetic pathways. Patent CN102126931B introduces a transformative preparation method that addresses these bottlenecks through a streamlined four-step synthetic sequence. This innovation leverages the Horner-Wadsworth-Emmons (HWE) reaction to construct the critical stilbene double bond, offering a robust alternative to traditional palladium-catalyzed couplings. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more economically viable and environmentally sustainable manufacturing protocols for this high-value API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of Resveratrol has relied heavily on methodologies that introduce significant operational and economic burdens. Traditional routes, such as the Perkin reaction or early Wittig variations, often suffer from poor stereoselectivity, yielding mixtures of cis and trans isomers that require tedious separation processes to isolate the biologically active trans-form. Furthermore, the widely cited Heck reaction, while effective for forming carbon-carbon bonds, imposes stringent requirements for anhydrous and oxygen-free conditions. The reliance on palladium catalysts in Heck coupling not only escalates raw material costs due to the precious metal's price volatility but also introduces severe downstream purification challenges. Removing trace palladium residues to meet stringent pharmaceutical impurity specifications often necessitates expensive scavenging technologies, thereby inflating the overall cost of goods sold and complicating the waste management profile for large-scale facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN102126931B circumvents these hurdles by employing a phosphonate-based strategy that is inherently more stable and easier to control. The core innovation lies in the conversion of 3,5-dimethoxybenzyl alcohol into a diethyl phosphonate intermediate, which then undergoes condensation with p-methoxybenzaldehyde. This approach ensures high stereoselectivity for the desired trans-isomer without the need for complex isomerization steps. Moreover, the final demethylation step utilizes aluminum trichloride in pyridine, a classic yet highly effective Lewis acid system that avoids the heavy metal contamination associated with transition metal catalysis. By simplifying the reaction conditions to standard heating and stirring protocols, this novel approach drastically reduces the technical barrier for industrial implementation.
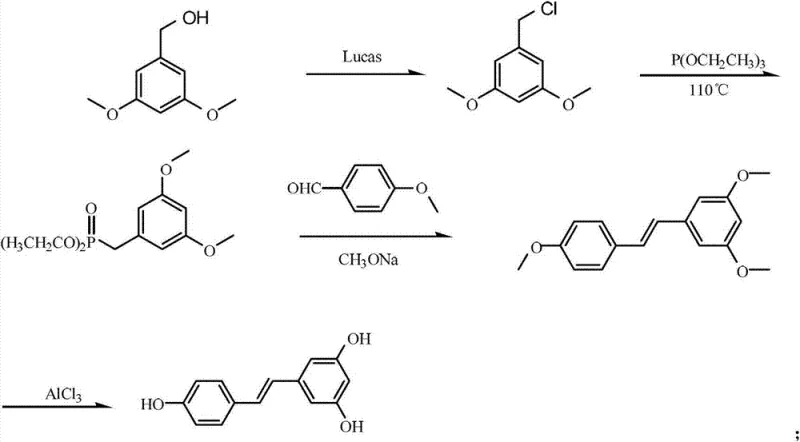
Mechanistic Insights into Horner-Wadsworth-Emmons Olefination
The heart of this synthetic strategy is the Horner-Wadsworth-Emmons reaction, which serves as the linchpin for constructing the central ethylene bridge of the stilbene scaffold. Mechanistically, this involves the generation of a stabilized carbanion from the 3,5-dimethoxybenzyl diethyl phosphonate upon treatment with a strong base like sodium methoxide. This nucleophilic species attacks the carbonyl carbon of p-methoxybenzaldehyde, forming a betaine intermediate that subsequently collapses to eliminate a phosphate byproduct. Unlike the Wittig reaction which produces stoichiometric amounts of triphenylphosphine oxide that can be difficult to remove, the HWE reaction generates water-soluble phosphate salts. This fundamental difference in byproduct solubility greatly simplifies the workup procedure, allowing for efficient separation of the organic product through simple aqueous washing and filtration, thereby enhancing the overall purity profile of the crude intermediate before recrystallization.
Following the formation of the trimethoxystilbene intermediate, the process employs a Lewis acid-mediated demethylation to reveal the phenolic hydroxyl groups essential for biological activity. The use of anhydrous aluminum trichloride (AlCl3) in anhydrous pyridine creates a potent complex capable of cleaving the aryl methyl ethers. The reaction mechanism involves the coordination of the aluminum center to the ether oxygen, followed by nucleophilic attack by chloride ions or pyridine complexes to sever the methyl-oxygen bond. Controlling the temperature gradient from 110°C during catalyst addition to 155°C for the main reaction phase is critical to driving this equilibrium to completion. This specific thermal profile ensures that all three methoxy groups are successfully converted to hydroxyl groups while minimizing potential side reactions such as polymerization or degradation of the sensitive stilbene double bond, resulting in the characteristic white needle-like crystals of high-purity Resveratrol.
How to Synthesize Resveratrol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise stoichiometric control and thermal management. The process begins with the activation of the benzylic position followed by phosphonation, setting the stage for the key coupling event. Operators must pay close attention to the vacuum distillation steps used to recover excess triethyl phosphite, as this not only improves atom economy but also prevents interference in subsequent steps. The final crystallization from ethyl acetate serves as a crucial purification checkpoint, ensuring that the final API intermediate meets the rigorous quality standards required for downstream formulation.
- Convert 3,5-dimethoxybenzyl alcohol to 3,5-dimethoxybenzyl chloride using Lucas reagent at room temperature.
- React the benzyl chloride with triethyl phosphite at 90-150°C to form the phosphonate ester.
- Perform Horner-Wadsworth-Emmons condensation with p-methoxybenzaldehyde using sodium methoxide to form the stilbene intermediate.
- Execute Lewis acid-mediated demethylation using anhydrous aluminum trichloride in pyridine at elevated temperatures to yield Resveratrol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of palladium catalysts represents a significant opportunity for cost reduction in Resveratrol manufacturing, as it removes the dependency on volatile precious metal markets and the capital expenditure associated with metal recovery systems. Furthermore, the use of commodity chemicals such as 3,5-dimethoxybenzyl alcohol and p-methoxybenzaldehyde ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty synthesis. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, translates directly into enhanced supply chain reliability and reduced energy consumption per kilogram of output.
- Cost Reduction in Manufacturing: The process architecture is designed to minimize operational expenditures by utilizing inexpensive reagents and avoiding costly transition metals. The ability to recover and recycle triethyl phosphite through vacuum distillation further enhances the economic efficiency of the process. Additionally, the simplified workup procedures reduce the demand for specialized chromatography resins or extensive solvent exchanges, leading to substantial cost savings in utilities and waste disposal. These factors collectively contribute to a lower cost of goods sold, enabling more competitive pricing strategies in the global nutraceutical and pharmaceutical markets.
- Enhanced Supply Chain Reliability: By relying on a linear synthesis with high-yielding steps, the method reduces the cumulative loss of material often seen in convergent syntheses with low individual step yields. The starting materials are widely available from bulk chemical suppliers, ensuring that production schedules are not held hostage by niche precursor availability. The tolerance of the reaction to standard industrial equipment means that manufacturing can be easily transferred between facilities or scaled up without requiring bespoke reactor designs, thereby securing continuity of supply even during periods of high market demand.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly lighter than traditional methods due to the absence of heavy metal waste streams. The aqueous solubility of the phosphate byproducts facilitates easier wastewater treatment, aligning with increasingly stringent global environmental regulations. The high selectivity of the HWE reaction minimizes the formation of difficult-to-separate isomers, reducing the volume of organic solvents required for purification. This green chemistry profile not only simplifies regulatory compliance but also enhances the sustainability credentials of the final product, a key differentiator for eco-conscious brand partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about technology transfer and capacity planning.
Q: Why is the phosphonate route preferred over Heck coupling for Resveratrol?
A: The phosphonate route avoids the use of expensive palladium catalysts, eliminating heavy metal contamination risks and reducing purification costs significantly compared to Heck coupling methods.
Q: What are the critical control points in the demethylation step?
A: Temperature control is critical; the reaction requires gradual heating to 110°C for catalyst addition and maintenance at 155°C for 4-5 hours to ensure complete removal of methyl groups without degrading the stilbene backbone.
Q: How does this method impact industrial scalability?
A: The process utilizes readily available raw materials and standard unit operations like vacuum distillation and recrystallization, making it highly suitable for multi-ton commercial scale-up without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this phosphonate route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Resveratrol we deliver meets the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this catalyst-free route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless integration of high-quality Resveratrol into your product pipeline.
