Industrial Scale-Up of Trans-Resveratrol via Novel Ligand-Free Heck Cross-Coupling Technology
Industrial Scale-Up of Trans-Resveratrol via Novel Ligand-Free Heck Cross-Coupling Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective routes for synthesizing high-value bioactive molecules like trans-resveratrol. Patent CN102276426A introduces a groundbreaking approach that addresses many of the historical bottlenecks associated with the production of 3,4,5-trihydroxystilbene. This patent discloses an environmentally friendly, ligand-free Heck cross-coupling reaction method that utilizes a transition metal supported on an inorganic carrier. This innovation represents a significant departure from traditional homogeneous catalysis systems, offering a pathway that is not only simpler in route design but also boasts a high overall yield and remarkably convenient after-treatment procedures. The structural integrity and biological potential of the target molecule, as depicted in the chemical structure below, make it a prime candidate for cardiovascular and anticancer therapeutic applications, driving the need for such robust manufacturing technologies.
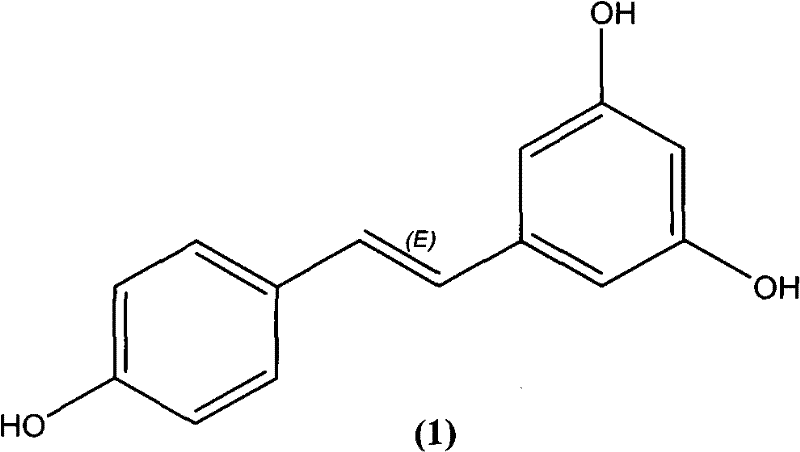
The significance of this patented technology lies in its ability to bypass the complex and often hazardous steps associated with classical synthesis methods. By employing a heterogeneous catalytic system, the process mitigates the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), a critical concern for regulatory compliance. Furthermore, the method eliminates the need for expensive and difficult-to-remove phosphine ligands, which are typically required in standard Heck couplings. This strategic shift not only reduces the raw material costs but also simplifies the downstream processing, making it an highly attractive option for large-scale industrial production where efficiency and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans-resveratrol has relied heavily on methods such as the Wittig-Horner reaction or Grubbs-catalyzed olefin metathesis, both of which present substantial challenges for industrial scale-up. The Wittig-Horner route, for instance, often necessitates the use of toxic trialkyl phosphites and involves an Arbuzov rearrangement that requires harsh pyrolysis conditions, leading to safety concerns and significant energy consumption. Additionally, these traditional pathways frequently suffer from poor atom economy and generate considerable amounts of chemical waste, complicating disposal and increasing the environmental footprint of the manufacturing process. Another critical drawback is the difficulty in controlling stereochemistry; conventional methods often produce mixtures of (E) and (Z) isomers, necessitating rigorous and costly purification steps to isolate the biologically active (E)-trans-resveratrol, which severely impacts the overall process yield and economic viability.
The Novel Approach
In stark contrast, the novel method described in the patent leverages a ligand-free Heck cross-coupling reaction that fundamentally alters the economic and operational landscape of resveratrol synthesis. As illustrated in the reaction scheme below, the process couples a diacyloxybenzoyl chloride derivative with a p-acyloxystyrene derivative under mild conditions. This approach abandons the classical reliance on expensive homogeneous catalysts and complex ligand systems, opting instead for a transition metal supported on an inexpensive inorganic carrier. The result is a highly stereoselective formation of the (E)-configuration double bond, effectively solving the isomer separation issue at the source. The simplicity of the route, combined with the use of commercially available starting materials, positions this technology as a superior alternative for manufacturers aiming to optimize their production lines for both cost and quality.
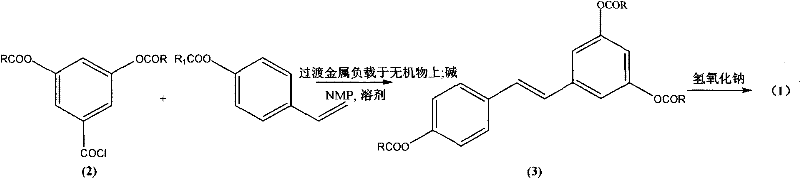
Mechanistic Insights into Ligand-Free Heck Cross-Coupling
The core of this technological advancement is the utilization of a transition metal catalyst, such as palladium, platinum, rhodium, or ruthenium, which is heterogeneously supported on materials like silica gel, activated carbon, or alumina. This specific configuration allows the catalytic cycle to proceed efficiently without the need for additional phosphine ligands, which are often air-sensitive, toxic, and difficult to remove from the final product. The general reaction formula demonstrates how the acyl chloride and styrene derivatives interact in the presence of this supported catalyst and a base. The absence of ligands simplifies the coordination sphere around the metal center, potentially enhancing the turnover frequency and reducing the catalyst loading required for the reaction to reach completion. This mechanistic simplicity translates directly to operational robustness, as the catalyst system is less sensitive to impurities and easier to handle in a standard industrial reactor setup.
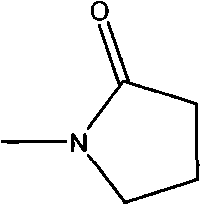
A critical component of this reaction mechanism is the role of N-Methyl-2-pyrrolidone (NMP) as a promoter or co-solvent. Experimental findings within the patent reveal that the addition of NMP is indispensable for achieving high conversion rates; without it, the yield of the coupling reaction drops precipitously. NMP likely acts by stabilizing the catalytic species or facilitating the solubility of the inorganic base and reactants, thereby accelerating the oxidative addition and reductive elimination steps of the Heck cycle. The structure of NMP, shown below, highlights its polar aprotic nature, which is ideal for dissolving a wide range of organic and inorganic species while maintaining thermal stability under the reaction conditions of 40 to 140 degrees Celsius. This synergistic effect between the supported metal and the NMP additive ensures that the reaction proceeds with high selectivity and minimal byproduct formation.
How to Synthesize Trans-Resveratrol Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating high-purity trans-resveratrol suitable for pharmaceutical applications. The process begins with the Heck coupling of protected precursors to form a triacyloxy stilbene intermediate, followed by a straightforward deprotection step. This two-stage sequence is designed to maximize yield while minimizing the complexity of purification, relying on crystallization and filtration rather than column chromatography. For detailed operational parameters, including specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide provided below, which encapsulates the critical steps for successful implementation of this novel chemistry.
- Perform a ligand-free Heck cross-coupling reaction between 3,5-diacyloxybenzoyl chloride and p-acyloxystyrene using a transition metal catalyst supported on an inorganic carrier in the presence of NMP.
- Isolate the intermediate triacyloxy stilbene derivative through simple filtration and washing, avoiding complex chromatographic purification.
- Conduct a saponification reaction on the intermediate using an alkali solution, followed by acidification and recrystallization to obtain the final trans-resveratrol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand-free Heck coupling technology offers tangible benefits that extend beyond mere chemical elegance. The shift from homogeneous to heterogeneous catalysis fundamentally changes the cost structure of the manufacturing process by eliminating the need for expensive ligand procurement and complex metal scavenging operations. Furthermore, the ability to recover and potentially reuse the supported catalyst through simple filtration significantly reduces the consumption of precious metals, leading to substantial long-term savings in raw material expenditures. The simplified workflow also means reduced labor hours and lower utility costs associated with extended purification processes, making the overall cost of goods sold (COGS) much more competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of costly phosphine ligands and the reduction in precious metal usage due to the efficiency of the supported catalyst system directly lower the variable costs of production. By avoiding the need for specialized equipment to handle toxic reagents or remove trace metals via chromatography, the capital expenditure and operational overhead are significantly decreased. This streamlined approach allows for a more lean manufacturing model where resources are focused on value-added steps rather than waste management and complex purification.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as acyl chlorides and styrenes, ensures a robust supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents. The simplicity of the reaction conditions, which do not require extreme temperatures or pressures, further enhances operational reliability and reduces the risk of batch failures due to equipment limitations. This stability translates to more predictable lead times and a consistent supply of high-quality intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst and the absence of toxic phosphorus byproducts make this process inherently scalable and environmentally compliant. The ease of catalyst separation via filtration facilitates large-scale batch processing without the bottlenecks associated with homogeneous catalyst removal. Additionally, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the liability and disposal costs associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's feasibility. Understanding these nuances is essential for R&D teams planning to integrate this method into their existing production workflows or for quality assurance teams assessing the purity profile of the resulting material.
Q: What are the advantages of using a supported transition metal catalyst over homogeneous catalysts in this synthesis?
A: Using a transition metal supported on an inorganic carrier allows for heterogeneous catalysis, which simplifies the separation process significantly. The catalyst can be removed via simple filtration, eliminating the need for complex purification steps to remove toxic metal residues from the final product, thereby ensuring higher purity and reducing environmental waste.
Q: Why is NMP (N-Methyl-2-pyrrolidone) critical in this Heck coupling reaction?
A: Experimental data indicates that the addition of NMP is essential for achieving high reaction yields. Without NMP, the yield of the transition metal-catalyzed coupling is very low, but its presence drastically promotes the reaction efficiency, allowing for milder conditions and shorter reaction times compared to traditional methods.
Q: How does this method compare to the traditional Wittig-Horner reaction for resveratrol synthesis?
A: Unlike the Wittig-Horner reaction, which often requires toxic phosphites, harsh conditions, and generates significant waste, this novel Heck coupling method utilizes easily available raw materials and avoids the use of expensive ligands. It offers a simpler operational path with high stereoselectivity for the (E)-configuration, resulting in a more environmentally friendly and cost-effective process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ligand-free Heck coupling described in CN102276426A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of trans-resveratrol meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this technology can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable, high-quality supply of this critical pharmaceutical intermediate.
