Advanced Supramolecular Purification of Resveratrol for Pharmaceutical Applications
Advanced Supramolecular Purification of Resveratrol for Pharmaceutical Applications
The pharmaceutical and nutraceutical industries constantly face the challenge of removing trace structural analogs from bioactive compounds, a difficulty vividly addressed in patent CN109516899B. This groundbreaking technology introduces a novel preparation method for high-purity resveratrol, utilizing a specially synthesized amphiphilic sulfonated calixarene, designated as RS002, to achieve unprecedented separation efficiency. Traditional purification struggles with homologous polyphenols that share nearly identical chemical properties with the target molecule, often leading to toxic side effects in biological applications due to residual impurities like 3,4'-dihydroxystilbene. By leveraging the precise molecular recognition capabilities of supramolecular chemistry, this invention offers a pathway to resveratrol with purity levels exceeding 99.9%, meeting the stringent requirements for bioactive substance chemicals used in sensitive therapeutic and cosmetic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the refinement of resveratrol has relied heavily on techniques such as column chromatography, repeated recrystallization, and acid-base precipitation, all of which suffer from significant operational and environmental drawbacks. Chromatography, while effective, is notoriously resource-intensive, consuming vast quantities of organic solvents and generating substantial hazardous waste that requires costly disposal protocols. Furthermore, the scalability of chromatographic processes is often limited by column capacity and flow rates, creating bottlenecks for manufacturers aiming to produce metric ton quantities of active pharmaceutical ingredients. Recrystallization methods frequently fail to adequately separate structural isomers like 3,4'-dihydroxystilbene due to their similar solubility profiles, resulting in products that may still contain biologically active impurities capable of causing adverse reactions such as diarrhea in consumers. These legacy methods also struggle with yield optimization, as multiple processing steps inevitably lead to product loss and increased overall production costs.
The Novel Approach
In stark contrast, the methodology described in CN109516899B employs a sophisticated host-guest recognition strategy that fundamentally changes the purification paradigm. The core of this innovation is the calixarene RS002, a macrocyclic compound engineered through a multi-step synthesis involving aldol condensation, Friedel-Crafts reaction, etherification, and sulfonation to create a specific binding cavity. This custom-designed host molecule selectively identifies and encapsulates the 3,4'-dihydroxystilbene impurity based on subtle differences in molecular geometry and electronic distribution, effectively pulling the contaminant out of the solution while leaving the desired resveratrol behind. 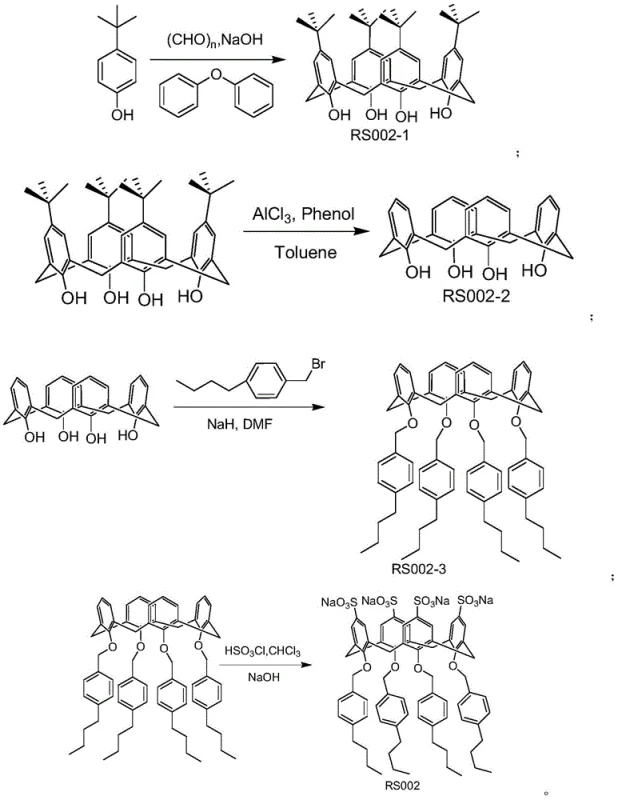 The process operates under mild conditions, typically at room temperature, and utilizes common aprotic polar solvents that can be easily recovered and recycled. This approach not only simplifies the workflow by replacing complex column setups with straightforward filtration but also aligns perfectly with the principles of green chemistry by minimizing waste generation and maximizing atomic economy.
The process operates under mild conditions, typically at room temperature, and utilizes common aprotic polar solvents that can be easily recovered and recycled. This approach not only simplifies the workflow by replacing complex column setups with straightforward filtration but also aligns perfectly with the principles of green chemistry by minimizing waste generation and maximizing atomic economy.
Mechanistic Insights into Supramolecular Host-Guest Recognition
The efficacy of this purification system lies in the precise structural complementarity between the sulfonated calixarene RS002 and the target impurity, 3,4'-dihydroxystilbene. The calixarene framework provides a rigid, cup-shaped architecture that acts as a molecular receptor, where the sulfonate groups enhance solubility in polar media and participate in electrostatic interactions with the guest molecule. When crude resveratrol containing the impurity is introduced to the system, the RS002 molecules selectively bind to the 3,4'-dihydroxystilbene through non-covalent interactions such as hydrogen bonding, pi-pi stacking, and hydrophobic effects, forming a stable supermolecular complex. This binding event alters the physical properties of the impurity, specifically its solubility in the presence of a non-polar anti-solvent like n-hexane, causing the complex to precipitate out of the solution while the unbound resveratrol remains dissolved. 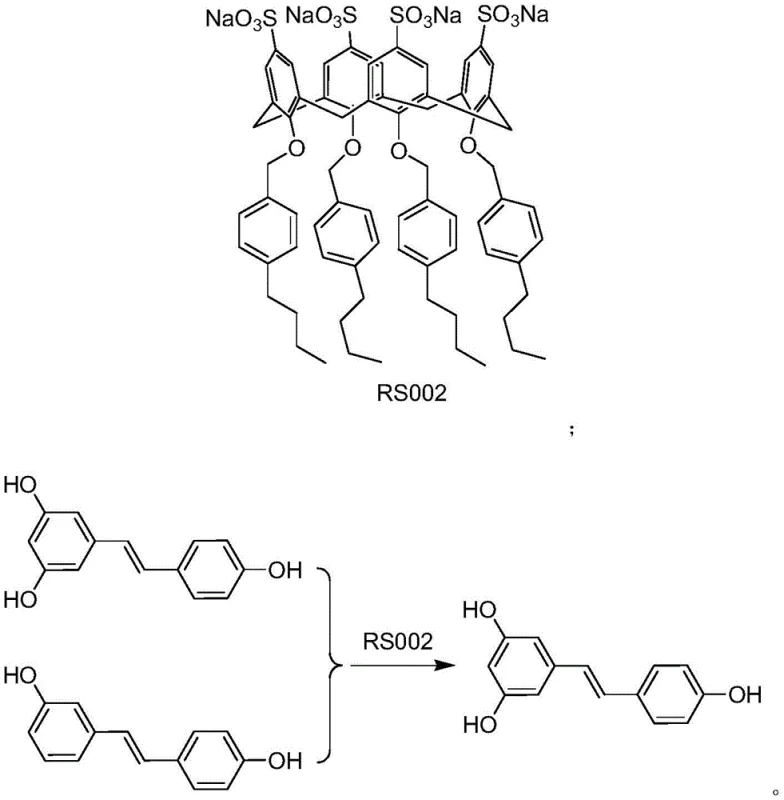 The selectivity is driven by the specific spatial arrangement of hydroxyl groups on the stilbene backbone; the 3,4'-substitution pattern of the impurity fits the calixarene cavity more favorably than the 3,5,4'-pattern of resveratrol, ensuring high-fidelity separation.
The selectivity is driven by the specific spatial arrangement of hydroxyl groups on the stilbene backbone; the 3,4'-substitution pattern of the impurity fits the calixarene cavity more favorably than the 3,5,4'-pattern of resveratrol, ensuring high-fidelity separation.
From an impurity control perspective, this mechanism offers a robust solution to the problem of homologous polyphenol contamination which has long plagued synthetic resveratrol production. Unlike chromatography which separates based on general polarity, this method separates based on specific molecular recognition, meaning that even trace amounts of the structurally similar impurity can be captured with high efficiency. The process parameters, such as the molar ratio of host to guest (ranging from 100:1 to 100:2) and the temperature control during precipitation (-5°C), are optimized to maximize the formation of the insoluble complex. This ensures that the final filtrate, upon solvent removal, yields resveratrol with a purity profile that surpasses 99.9%, effectively eliminating the risk of toxicity associated with the byproduct. Furthermore, the reversibility of the host-guest interaction allows for the regeneration of the expensive calixarene host from the filter cake, adding a layer of economic sustainability to the technical process.
How to Synthesize High-Purity Resveratrol Efficiently
The implementation of this technology involves a streamlined sequence of mixing, precipitation, and filtration that is highly amenable to industrial scale-up. The process begins by dissolving crude resveratrol and the calixarene RS002 in a suitable aprotic polar solvent such as tetrahydrofuran or dioxane, followed by a stirring period to allow the supramolecular assembly to reach equilibrium. Subsequent addition of n-hexane acts as an anti-solvent to induce the crystallization of the impurity-host complex, which is then removed via simple filtration. The detailed standardized synthesis steps, including specific reagent quantities and reaction times derived from the patent examples, are outlined in the guide below.
- Mix crude resveratrol with calixarene RS002 in an aprotic polar solvent like tetrahydrofuran at room temperature for 6-8 hours to form the host-guest complex.
- Add n-hexane to the reaction system and cool the mixture to -5°C to precipitate the impurity-host complex while keeping pure resveratrol in solution.
- Filter the mixture to separate the solid complex, then distill the filtrate to recover solvents and obtain refined resveratrol with purity exceeding 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this supramolecular purification technology represents a significant opportunity to optimize cost structures and mitigate operational risks associated with traditional refining methods. The elimination of silica gel chromatography removes a major variable cost center, as the recurring expense of stationary phases and the logistical burden of hazardous waste disposal are drastically reduced. Moreover, the ability to recover and reuse the calixarene host molecule RS002 from the filter cake creates a closed-loop material flow, diminishing the dependency on continuous raw material inputs for the purification agent itself. This circular economy approach not only lowers the direct cost of goods sold but also insulates the supply chain from volatility in the pricing of specialized purification media.
- Cost Reduction in Manufacturing: The transition from chromatography to selective precipitation fundamentally alters the cost basis of resveratrol production by removing the need for expensive column packing materials and reducing solvent consumption volumes. Since the process relies on simple mixing and filtration equipment rather than complex chromatographic systems, capital expenditure for facility upgrades is minimized, and maintenance costs are significantly lower. The high yield reported in the patent examples, consistently approaching theoretical maximums, ensures that valuable raw material is not lost to tailing fractions or column dead volumes, directly improving the overall mass balance and profitability of the manufacturing line.
- Enhanced Supply Chain Reliability: By simplifying the purification workflow to a few batch operations, manufacturers can achieve faster turnaround times and more predictable production schedules compared to the variable run-times of column chromatography. The use of common industrial solvents like tetrahydrofuran and n-hexane ensures that raw material sourcing remains stable and unaffected by the supply constraints often seen with specialized chromatographic grades. Additionally, the robustness of the supramolecular recognition mechanism means that variations in crude feedstock quality can be accommodated without compromising the final purity specification, providing a buffer against upstream supply fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the geometric limitations of chromatography columns, allowing for straightforward expansion from pilot batches to multi-ton commercial production using standard reactor vessels. From a regulatory and sustainability standpoint, the absence of wastewater, waste gas, and solid residue generation simplifies environmental compliance and reduces the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the operation against tightening environmental regulations but also enhances the brand value of the final product in markets that prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this supramolecular purification technology, based on the specific data and mechanisms disclosed in the patent literature. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this method into existing production lines and for quality assurance personnel defining new specification standards. The answers provided reflect the proven capabilities of the RS002 calixarene system in achieving high-purity outcomes while maintaining process efficiency.
Q: How does the RS002 calixarene distinguish between resveratrol and its impurities?
A: The RS002 calixarene utilizes a supramolecular host-guest recognition mechanism based on molecular size and shape differences. It selectively forms a stable inclusion complex with the impurity 3,4'-dihydroxystilbene, allowing the target resveratrol molecule to remain in the solution phase for easy separation.
Q: What are the environmental benefits of this purification method compared to chromatography?
A: Unlike traditional column chromatography which generates significant solvent waste and silica gel residue, this supramolecular method operates with recyclable solvents and a reusable host molecule. It eliminates wastewater, waste gas, and solid residue, aligning with green chemistry principles and atomic economy.
Q: Can the calixarene host molecule RS002 be recovered and reused?
A: Yes, the process is designed for circularity. The filter cake containing the impurity-host complex can be refluxed in ethanol to recover the calixarene RS002, which can then be reused in subsequent purification cycles, significantly reducing material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and safety in the production of bioactive compounds like resveratrol, and we possess the technical expertise to translate complex supramolecular pathways into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully adapted for large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of trace impurities like 3,4'-dihydroxystilbene, guaranteeing that every batch meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced purification technology can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this green chemistry approach. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering high-quality, cost-effective solutions for the global market.
