Scalable Electrochemical Micro-Flow Synthesis of Trifluoromethylated Isoindolinone Intermediates
Introduction to Next-Generation Electrochemical Manufacturing
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient technologies, a transition exemplified by the groundbreaking methodology disclosed in patent CN113549939B. This intellectual property introduces a novel continuous electrochemical synthesis route for producing trifluoromethylated isoindolinones, a critical structural motif found in numerous bioactive pharmaceutical compounds and advanced materials. By leveraging the precision of micro-reaction technology combined with organic electro-synthesis, this process circumvents the traditional reliance on stoichiometric oxidants and transition metal catalysts. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational safety, offering a robust pathway to access complex heterocyclic scaffolds that were previously difficult to manufacture sustainably. The integration of flow chemistry principles ensures that the reaction parameters, such as residence time and current density, are tightly controlled, leading to superior reproducibility and product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the trifluoromethyl functionalized isoindolinone skeleton has relied heavily on transition metal catalysis, particularly copper-mediated intramolecular aminotrifluoromethylation of olefins. While effective on a small laboratory scale, these conventional batch processes suffer from inherent drawbacks that hinder their industrial viability. The requirement for excessive amounts of metal catalysts not only drives up raw material costs but also necessitates rigorous and expensive downstream purification steps to remove trace metal residues to meet pharmaceutical standards. Furthermore, these reactions typically demand harsh oxidants to regenerate the active catalytic species, introducing significant safety hazards related to thermal runaway and the handling of unstable reagents. The accumulation of metal waste and the use of aggressive oxidizing agents create a substantial environmental burden, complicating waste disposal and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes electricity as a clean reagent to drive the intramolecular free radical cascade reaction, effectively replacing the need for chemical oxidants and metal catalysts. By employing a microchannel reaction device, the system achieves exceptional mass and heat transfer efficiency, allowing for the safe generation and immediate consumption of reactive trifluoromethyl radicals. This continuous flow setup minimizes the holdup volume of hazardous intermediates, drastically reducing the risk of explosion or uncontrolled exothermic events. The result is a streamlined, green synthesis protocol that operates under mild conditions, typically at room temperature, while delivering high selectivity for the target isoindolinone structure. This approach not only simplifies the workflow but also aligns perfectly with modern principles of sustainable chemistry and process intensification.
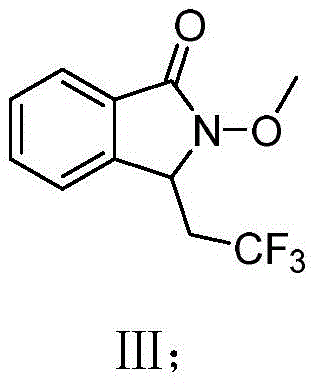
Mechanistic Insights into Electro-Oxidative Radical Cyclization
The core of this technological advancement lies in the anodic oxidation mechanism facilitated by the micro-flow environment. In this system, the sodium trifluoromethanesulfinate serves as the trifluoromethyl source, which undergoes single-electron oxidation at the anode surface to generate the highly reactive trifluoromethyl radical species. Unlike batch systems where radical concentration can fluctuate wildly, the laminar flow within the microchannels ensures a consistent flux of radicals to the substrate, N-methoxy-2-vinylbenzamide. This precise control promotes the desired intermolecular addition of the trifluoromethyl radical to the vinyl group, followed rapidly by an intramolecular cyclization involving the amide nitrogen. The subsequent oxidation and deprotonation steps finalize the formation of the isoindolinone ring system. The use of tetrabutylammonium acetate as a supporting electrolyte further stabilizes the reaction medium and facilitates ion transport without interfering with the radical pathway, ensuring high faradaic efficiency.
From an impurity control perspective, the electrochemical flow method offers distinct advantages over traditional thermal radical initiators. In batch reactors, localized hot spots can lead to the decomposition of sensitive intermediates or the formation of polymerization byproducts. However, the high surface-to-volume ratio of the microreactor allows for instantaneous heat dissipation, maintaining a uniform temperature profile throughout the reaction zone. This thermal homogeneity suppresses side reactions such as over-oxidation or non-selective radical coupling, resulting in a cleaner crude reaction mixture. Consequently, the burden on downstream purification is significantly reduced, as the impurity profile is much simpler and more predictable. For quality assurance teams, this translates to more consistent batch-to-batch purity and a lower risk of encountering difficult-to-remove genotoxic impurities often associated with metal-catalyzed processes.
How to Synthesize Trifluoromethylated Isoindolinone Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot setting, operators must adhere to precise fluidic and electrical parameters to maximize yield and safety. The process begins with the preparation of a homogeneous feed solution containing the substrate, the trifluoromethyl source, and the electrolyte in a mixed solvent system of acetonitrile and water. This solution is then continuously pumped through the microchannel reactor where the electrochemical transformation occurs. The detailed standardized synthesis steps, including specific flow rates, current densities, and electrode configurations validated by the patent data, are outlined below to ensure successful replication of the high-yielding protocol.
- Prepare a homogeneous solution by dissolving N-methoxy-2-vinylbenzamide, sodium trifluoromethanesulfinate, and tetrabutylammonium acetate electrolyte in an acetonitrile/water solvent mixture.
- Pump the homogeneous solution into a microchannel reaction device equipped with a graphite anode and platinum cathode at a controlled flow rate of 225 μL/min.
- Apply a constant current of 10mA to initiate the electrolytic reaction at room temperature, collecting the effluent containing the target isoindolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical micro-flow technology presents a compelling value proposition centered on cost optimization and supply security. By eliminating the dependency on expensive transition metal catalysts like copper and avoiding the procurement of hazardous chemical oxidants, the raw material cost structure is fundamentally improved. The removal of metal catalysts also eradicates the need for specialized scavenging resins or complex filtration units, simplifying the equipment train and reducing capital expenditure. Furthermore, the continuous nature of the process allows for a smaller physical footprint compared to large batch reactors, enabling production facilities to generate higher throughput within existing infrastructure constraints. This intensification of manufacturing capacity directly addresses the growing demand for complex fluorinated intermediates in the pharmaceutical sector without requiring massive facility expansions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the reduction of auxiliary material costs. Since the reaction does not require stoichiometric oxidants or metal catalysts, the bill of materials is significantly leaner, and the costs associated with waste treatment for heavy metals are completely avoided. Additionally, the high selectivity of the electrochemical method reduces solvent consumption during purification, as less chromatography or recrystallization is needed to achieve target purity specifications. These factors combine to deliver substantial cost savings per kilogram of finished product, enhancing the overall margin profile for commercial manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials that do not require special storage conditions or hazardous shipping protocols. The continuous flow setup ensures a steady, on-demand production capability, reducing the lead times associated with long batch cycles and cleaning validation between runs. This agility allows manufacturers to respond more quickly to fluctuations in market demand, ensuring a consistent supply of high-purity intermediates to downstream API producers. The robustness of the electrochemical cell design also minimizes unplanned downtime, further securing the continuity of supply for critical pharmaceutical projects.
- Scalability and Environmental Compliance: Scaling this process is inherently safer and more straightforward than scaling batch radical reactions, as the risks are managed by the micro-channel geometry rather than by massive containment vessels. The green chemistry credentials of the process, characterized by the absence of toxic metal waste and the use of electricity as a renewable energy input, align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and potential fines associated with hazardous waste disposal, making the supply chain more sustainable and future-proof against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the feasibility and advantages of adopting this technology for industrial applications. Understanding these nuances is essential for stakeholders evaluating the transition from traditional batch chemistry to continuous flow electro-synthesis.
Q: What are the primary advantages of this electrochemical method over traditional copper-catalyzed synthesis?
A: Unlike traditional methods that require harsh oxidants and excessive copper catalysts which create heavy metal waste, this electrochemical approach uses electricity as the reagent. This eliminates the need for expensive metal removal steps and significantly reduces environmental impact while maintaining high efficiency.
Q: How does the microchannel reactor improve the safety of trifluoromethylation reactions?
A: The microchannel reactor offers a high surface-area-to-volume ratio, allowing for precise temperature control and instantaneous mixing. This prevents the accumulation of reactive radical intermediates and avoids thermal hotspots, making the handling of trifluoromethyl radicals much safer compared to large batch reactors.
Q: Is this process suitable for industrial scale-up of pharmaceutical intermediates?
A: Yes, the continuous flow nature of the microchannel system facilitates easy scale-up through numbering up or increasing run time. The mild reaction conditions and absence of hazardous oxidants make it highly compatible with GMP manufacturing requirements for complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical micro-flow synthesis in the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced flow chemistry modules and rigorous QC labs capable of meeting the most stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity isoindolinone derivatives that empower your drug development pipelines with reliable quality and consistent supply.
We invite you to collaborate with our technical team to explore how this green synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this electrochemical method for your supply chain. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique molecular targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
