Scalable Synthesis of L-Alanyl-L-Glutamine via Formyl Protection for Parenteral Nutrition
The landscape of parenteral nutrition has been revolutionized by the advent of stable amino acid dipeptides, specifically L-alanyl-L-glutamine, which serves as a critical source of glutamine for patients under metabolic stress. As detailed in patent CN101519428A, a novel synthetic methodology has emerged that addresses the longstanding challenges of toxicity, cost, and complexity associated with traditional dipeptide manufacturing. This technical insight report analyzes the strategic advantages of this formyl-protection route, offering a compelling value proposition for R&D directors seeking high-purity intermediates and procurement managers aiming for sustainable cost structures. The patent outlines a robust two-step process that utilizes N-formyl-L-alanine and isobutyl chloroformate to create a mixed anhydride, which subsequently couples with L-glutamine. This approach not only mitigates the risks of racemization but also simplifies the downstream purification process, making it an ideal candidate for commercial scale-up in the competitive pharmaceutical intermediates market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-alanyl-L-glutamine has been plagued by significant technical and economic hurdles that hinder efficient commercial production. One prevalent method involves the use of Z-Ala (N-benzyloxycarbonyl-L-alanine) coupled with N-hydroxy-succinimide (HOSu) in the presence of N,N'-dicyclohexylcarbodiimide (DCC). While chemically effective, this route generates dicyclohexylurea (DCU) as a stoichiometric byproduct, which is notoriously difficult to remove completely from the reaction mixture, often requiring complex filtration and washing steps that reduce overall yield. Furthermore, the Z-protecting group requires hydrogenation for removal, necessitating the use of precious metal catalysts and high-pressure equipment, which introduces safety risks and capital expenditure burdens. Another historical approach utilizes phosgene to generate mixed anhydrides; however, phosgene is a Schedule 1 chemical warfare agent precursor with extreme toxicity, imposing rigorous regulatory compliance costs and severe safety hazards for plant operators. These conventional pathways result in higher production costs, lower purity profiles, and significant environmental liabilities due to hazardous waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN101519428A introduces a streamlined and safer synthetic pathway that leverages the N-formyl protecting group and mixed anhydride activation. By reacting N-formyl-L-alanine with isobutyl chloroformate in the presence of N-methylmorpholine at controlled low temperatures (-18°C to -15°C), the process generates a reactive mixed anhydride intermediate in situ. This intermediate then couples efficiently with L-glutamine to form N-formyl-L-alanyl-L-glutamine. The brilliance of this approach lies in its simplicity and safety profile; isobutyl chloroformate is a manageable liquid reagent, eliminating the need for toxic phosgene gas. Moreover, the subsequent deprotection step utilizes mild hydrochloric acid hydrolysis at 40°C, avoiding the need for explosive hydrogenation processes. This novel route ensures that the chiral center of the alanine residue remains intact, preventing racemization and guaranteeing the biological efficacy of the final dipeptide product.
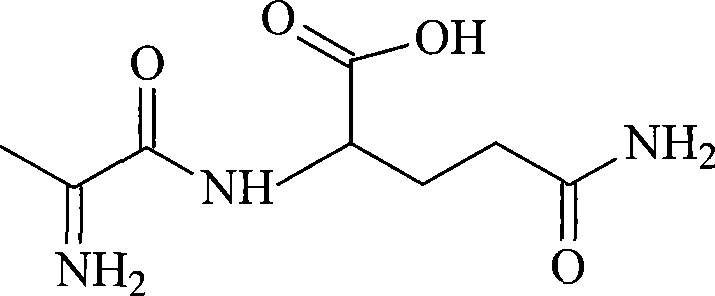
Mechanistic Insights into Formyl-Protection and Mixed Anhydride Coupling
The core chemical innovation of this process revolves around the strategic selection of the formyl group as the N-terminal protecting moiety and the utilization of the mixed anhydride method for peptide bond formation. Mechanistically, the reaction begins with the activation of the carboxylic acid of N-formyl-L-alanine by isobutyl chloroformate. In the presence of a tertiary amine base like N-methylmorpholine, the chloroformate reacts with the carboxylate anion to form a highly electrophilic mixed anhydride species. This species is particularly advantageous because it is less prone to racemization compared to other activated esters, primarily due to the electron-withdrawing nature of the formyl group which stabilizes the alpha-proton against base-catalyzed enolization. When L-glutamine is introduced to the reaction mixture, its alpha-amino group acts as a nucleophile, attacking the carbonyl carbon of the alanine-derived anhydride. The reaction is conducted at sub-zero temperatures (-18°C) to further suppress any potential side reactions or epimerization, ensuring that the stereochemical integrity of the amino acid building blocks is preserved throughout the coupling event.
Following the coupling, the removal of the formyl protecting group is achieved through acid-catalyzed hydrolysis, a mechanism that offers superior control over product quality. The N-formyl-L-alanyl-L-glutamine intermediate is suspended in 1M hydrochloric acid and heated to a moderate temperature of 40°C. Under these specific conditions, the amide bond of the formyl group is cleaved selectively without hydrolyzing the newly formed peptide bond between alanine and glutamine. This selectivity is critical; harsher acidic conditions or higher temperatures could lead to the degradation of the dipeptide back into free amino acids, drastically reducing yield. The resulting L-alanyl-L-glutamine is then isolated by adjusting the pH to neutrality and utilizing isobutyl acetate for extraction. This solvent choice is mechanistically sound as it effectively partitions the organic product away from inorganic salts and water-soluble impurities, facilitating a clean crystallization process that yields a product with optical purity exceeding 99.8%.
How to Synthesize L-Alanyl-L-Glutamine Efficiently
The synthesis of L-alanyl-L-glutamine via this patented route represents a significant optimization of process chemistry, balancing reaction kinetics with operational safety. The procedure is designed to be robust, allowing for the direct use of the intermediate in the subsequent step without extensive purification, which is a key factor in improving overall throughput. The detailed standardized synthesis steps, including precise molar ratios, temperature ramping rates, and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Activate N-formyl-L-alanine with isobutyl chloroformate and N-methylmorpholine in THF at -18°C to form a mixed anhydride intermediate.
- React the mixed anhydride with L-glutamine under controlled low temperatures to form N-formyl-L-alanyl-L-glutamine.
- Hydrolyze the formyl protecting group using 1M hydrochloric acid at 40°C, followed by pH adjustment and recrystallization to obtain the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this formyl-protection synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike methods requiring specialized coupling reagents like HOSu or hazardous gases like phosgene, this process relies on commodity chemicals such as isobutyl chloroformate and formic acid derivatives, which are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks and price volatility, ensuring a more stable and predictable procurement environment. Furthermore, the elimination of heavy metal catalysts (used in hydrogenation steps of other methods) removes the need for expensive and time-consuming metal scavenging processes, directly translating to lower operational expenditures and faster batch cycle times.
- Cost Reduction in Manufacturing: The economic impact of this synthesis method is profound, driven primarily by the substitution of expensive protecting groups and coupling agents with cost-effective alternatives. The use of the formyl group, which is derived from inexpensive formic acid sources, replaces the costly Z-group (benzyloxycarbonyl) that requires benzyl chloride and phosgene for its own synthesis. Additionally, the avoidance of DCC eliminates the loss of material associated with DCU byproduct formation and removal. The process also utilizes isobutyl acetate, a solvent that can be efficiently recovered and recycled through distillation, significantly reducing solvent purchase and waste disposal costs. These cumulative factors result in a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the global amino acid dipeptide market without compromising margin.
- Enhanced Supply Chain Reliability: From a logistics and continuity perspective, this method enhances reliability by removing dependencies on highly regulated and dangerous substances. The absence of phosgene means that facilities do not require specialized containment systems or emergency response protocols associated with toxic gas leaks, thereby reducing insurance premiums and regulatory inspection frequencies. The raw materials, including N-formyl-L-alanine and L-glutamine, are stable solids that are easy to store and transport, minimizing the risks associated with shelf-life degradation or hazardous shipping requirements. This stability ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and enabling manufacturers to respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than traditional routes, aligning with modern green chemistry principles and stringent environmental regulations. The aqueous workup and extraction steps generate waste streams that are easier to treat compared to the complex organic sludge produced by DCC couplings. The high atom economy of the mixed anhydride method, combined with the recyclability of the organic solvents, minimizes the E-factor (mass of waste per mass of product). This eco-friendly profile facilitates smoother regulatory approvals for new manufacturing sites and supports corporate sustainability goals. Moreover, the mild reaction conditions (near ambient pressure and moderate temperatures) make the process inherently safer and easier to scale from pilot plant to multi-ton commercial production, ensuring that supply can grow in tandem with the expanding demand for parenteral nutrition solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of L-alanyl-L-glutamine synthesized via this advanced method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on purity standards, process safety, and scalability for potential partners and stakeholders evaluating this technology for their supply chains.
Q: Why is the N-formyl protecting group preferred over Z-group or phosgene methods?
A: The N-formyl group is significantly cheaper and easier to introduce than the Z-group (benzyloxycarbonyl). Unlike phosgene methods which involve highly toxic gases, the formyl method uses safer reagents like isobutyl chloroformate. Furthermore, the formyl group can be removed under mild acidic conditions (40°C, 1M HCl) without risking racemization or peptide bond hydrolysis, ensuring high optical purity.
Q: How does this synthesis method address impurity control in dipeptide production?
A: Traditional methods using DCC often leave behind difficult-to-remove dicyclohexylurea (DCU) byproducts. The mixed anhydride method described in patent CN101519428A generates soluble byproducts that are easily removed during the aqueous workup and extraction phases. The specific use of isobutyl acetate as an extraction solvent allows for efficient separation of the organic phase, leading to a final product purity exceeding 99.8% after recrystallization.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It avoids hazardous reagents like phosgene and expensive coupling agents like HOSu/DCC. The solvents used, such as tetrahydrofuran and isobutyl acetate, are recyclable, which drastically reduces waste disposal costs. The reaction conditions (-18°C to 40°C) are easily manageable in standard industrial reactors without requiring extreme cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Alanyl-L-Glutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex peptides like L-alanyl-L-glutamine requires more than just a laboratory recipe; it demands a partner with deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We understand the critical nature of stringent purity specifications in the pharmaceutical sector, which is why our rigorous QC labs employ state-of-the-art analytical techniques to verify optical purity, residual solvent levels, and impurity profiles, guaranteeing that every batch meets the highest international standards for parenteral nutrition ingredients.
We invite you to collaborate with us to leverage this innovative synthesis technology for your product portfolio. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this formyl-protection route for your operations. We encourage you to reach out today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you optimize your supply chain, reduce manufacturing costs, and secure a reliable source of high-quality L-alanyl-L-glutamine for the global market.
