Revolutionizing 8-Aminocaprylic Acid Production: A Safe, Scalable Route for Global API Supply Chains
Revolutionizing 8-Aminocaprylic Acid Production: A Safe, Scalable Route for Global API Supply Chains
The pharmaceutical industry's relentless pursuit of effective oral delivery systems for polypeptide drugs has placed a spotlight on permeation enhancers, specifically N-(8-[2-hydroxybenzoyl]amino)caprylate, widely known as SNAC. As the demand for oral semaglutide and similar therapies surges, the supply chain for its critical precursor, 8-aminocaprylic acid, faces unprecedented pressure. Patent CN115557846A introduces a transformative synthetic methodology that addresses the longstanding bottlenecks of safety, cost, and environmental compliance associated with this valuable pharmaceutical intermediate. By shifting away from hazardous azide chemistry and heavy metal oxidants, this innovation offers a robust pathway for commercial scale-up of complex amino acids, ensuring a stable supply for global API manufacturers.
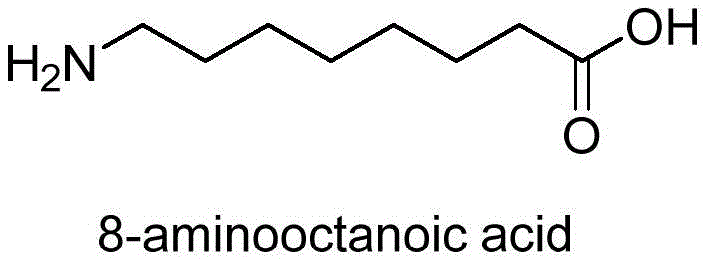
8-Aminocaprylic acid serves as the foundational backbone for SNAC, a molecule that facilitates the transcellular transport of therapeutic peptides by altering their configuration to expose hydrophobic regions. The structural integrity and purity of this fatty acid derivative are paramount, as impurities can compromise the efficacy and safety of the final drug product. The patent details a novel intermediate, represented by Formula I, which acts as a protected precursor, allowing for precise control over the synthesis trajectory. This strategic approach not only simplifies the purification process but also aligns with the rigorous quality standards required by regulatory bodies for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
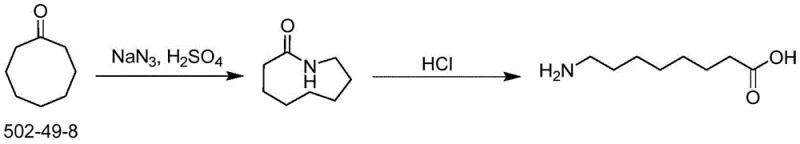
Historically, the synthesis of 8-aminocaprylic acid has been plagued by significant safety and environmental hazards that hinder large-scale production. One prominent legacy method involves the rearrangement of cyclooctanone using sodium azide and concentrated sulfuric acid. While chemically feasible, this route presents a catastrophic safety profile due to the potential formation of virulent hydrazoic acid and the inherent explosivity of organic azides. Such risks render this method unsuitable for multi-ton manufacturing, restricting it to small laboratory batches and creating a fragile supply chain vulnerable to shutdowns.
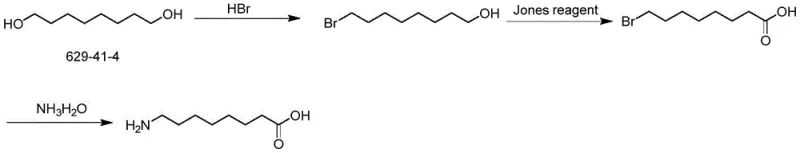
Another conventional pathway utilizes 1,8-octanediol as a starting material, employing the Jones reagent for oxidation. This approach introduces severe environmental liabilities due to the generation of chromium-containing waste solids. The disposal of hexavalent chromium is costly and heavily regulated, driving up the operational expenditure for manufacturers. Furthermore, the starting material, octanediol, is relatively expensive compared to alternative feedstocks. These factors combined result in a process that is neither economically viable nor environmentally sustainable for the growing demands of the pharmaceutical intermediates market.
The Novel Approach
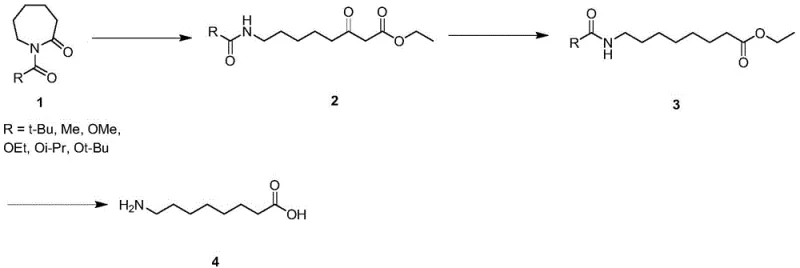
In stark contrast, the methodology disclosed in Patent CN115557846A leverages a substituted caprolactam derivative as the primary feedstock, initiating a sequence that is inherently safer and more cost-effective. The core innovation lies in the ring-opening reaction of the lactam with ethyl acetate under basic conditions, followed by a mild reductive amination. This strategy completely bypasses the need for explosive azides and toxic chromium oxidants. The use of readily available and inexpensive raw materials significantly lowers the barrier to entry for production, facilitating cost reduction in API manufacturing while maintaining a superior safety profile for plant operators.
The new route is designed with scalability in mind, utilizing standard unit operations such as low-temperature addition, reflux, and acidic hydrolysis. The intermediate compounds generated along the pathway are stable and易于 handle, reducing the risk of runaway reactions. By establishing a linear and predictable synthesis, this method ensures supply chain reliability, allowing manufacturers to ramp up production volumes from pilot scales to hundreds of tons annually without the technical hurdles associated with legacy chemistries. This represents a paradigm shift towards green chemistry principles in the production of critical peptide delivery excipients.
Mechanistic Insights into Lactam Ring-Opening and Reductive Amination
The chemical elegance of this synthesis begins with the nucleophilic attack on the lactam ring. In the presence of a strong non-nucleophilic base such as potassium hexamethyldisilazide (KHMDS) or lithium diisopropylamide (LDA), ethyl acetate is deprotonated to form an enolate. This enolate attacks the carbonyl carbon of the caprolactam derivative at cryogenic temperatures ranging from -80°C to -70°C. This low-temperature control is crucial for regioselectivity, ensuring the ring opens cleanly to form the desired beta-keto ester intermediate (Compound 2) without side reactions or polymerization. The precision of this step dictates the overall yield and purity of the downstream process.
Following the ring opening, the ketone functionality is converted to an amine via a hydrazone intermediate. The reaction with p-toluenesulfonyl hydrazide in toluene at elevated temperatures (100-110°C) forms the hydrazone, which is subsequently reduced in situ using sodium borohydride acetate. This reductive amination is highly selective, avoiding the over-reduction of the ester group, which is a common pitfall in similar transformations. The final deprotection step involves acidic hydrolysis using concentrated hydrochloric acid, followed by neutralization with propylene oxide. This specific choice of base for salt decomposition minimizes the introduction of inorganic salts, simplifying the final isolation and drying steps to achieve the reported 99.9% purity.
How to Synthesize 8-Aminocaprylic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing 8-aminocaprylic acid with industrial viability. The process is divided into three distinct stages: the formation of the beta-keto ester, the reductive amination to the protected amine, and the final deprotection. Each stage has been optimized for yield and safety, with specific attention paid to temperature control and reagent stoichiometry. For R&D teams looking to implement this technology, the following guide summarizes the critical operational parameters derived from the patent examples.
- Perform a ring-opening reaction on a substituted caprolactam derivative using ethyl acetate and a strong base like KHMDS at cryogenic temperatures (-80 to -70°C) to form a beta-keto ester intermediate.
- Execute a reductive amination using p-toluenesulfonyl hydrazide and sodium borohydride acetate in toluene at elevated temperatures (100-110°C) to reduce the ketone and install the amine functionality.
- Conduct acidic hydrolysis using concentrated hydrochloric acid followed by salt decomposition with propylene oxide to isolate the final 8-aminocaprylic acid product with 99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of hazardous reagents translates directly into lower insurance premiums, reduced safety infrastructure costs, and minimized risk of production stoppages due to safety incidents. Furthermore, the avoidance of heavy metals simplifies waste management protocols, leading to substantial cost savings in environmental compliance and waste disposal. These factors collectively enhance the economic attractiveness of sourcing 8-aminocaprylic acid produced via this method.
- Cost Reduction in Manufacturing: The replacement of expensive starting materials like octanediol and cyclooctanone with cheaper lactam derivatives significantly lowers the raw material cost base. Additionally, the removal of the Jones oxidation step eliminates the need for costly chromium waste treatment facilities. The streamlined three-step process also reduces utility consumption and labor hours per kilogram of product, driving down the overall cost of goods sold (COGS) and allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: By utilizing stable intermediates and avoiding reagents with strict transportation restrictions (such as sodium azide), the logistics of raw material procurement become far more resilient. The process is less susceptible to regulatory crackdowns on hazardous chemical transport, ensuring a continuous flow of materials into the production facility. This stability is critical for maintaining long-term supply contracts with major pharmaceutical clients who require guaranteed delivery schedules for their API production lines.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively, with examples showing successful translation from gram-scale to multi-kilogram batches without loss of efficiency. The absence of heavy metal catalysts and explosive precursors aligns perfectly with modern ESG (Environmental, Social, and Governance) goals. This makes the resulting product highly attractive to multinational corporations seeking to reduce the carbon footprint and environmental toxicity of their supply chains, future-proofing the procurement strategy against tightening global regulations.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the implementation and benefits of this new synthetic route. These insights are derived directly from the technical specifications and comparative data provided in Patent CN115557846A, offering clarity on safety, purity, and operational feasibility for potential partners and licensees.
Q: How does this new synthesis method improve safety compared to traditional azide routes?
A: Traditional methods often utilize sodium azide and concentrated sulfuric acid, which pose severe explosion risks and generate toxic hydrazoic acid. The patented method replaces these hazardous reagents with stable lactam derivatives and standard reducing agents, eliminating the risk of explosive decomposition during scale-up.
Q: What are the environmental advantages over the Jones oxidation method?
A: Conventional synthesis using octanediol requires Jones reagent, which contains hexavalent chromium, leading to heavy metal waste disposal issues. This novel route avoids heavy metal oxidants entirely, significantly reducing environmental compliance costs and waste treatment complexity.
Q: What purity levels can be achieved with this industrial process?
A: The process is designed for high-purity output, consistently achieving 99.9% purity (ELSD) in the final 8-aminocaprylic acid product. This high quality is critical for its application as a key intermediate in peptide drug permeation enhancers like SNAC.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Aminocaprylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of next-generation peptide therapeutics. Our team of expert chemists has thoroughly analyzed the patented route for 8-aminocaprylic acid and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 8-aminocaprylic acid meets the exacting standards required for SNAC production.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and security into your supply chain.
