Advanced Palladium-Catalyzed Cyanation for High-Purity Aryl Nitrile Manufacturing
Advanced Palladium-Catalyzed Cyanation for High-Purity Aryl Nitrile Manufacturing
The synthesis of aryl nitriles represents a cornerstone capability in modern fine chemical and pharmaceutical manufacturing, serving as a critical gateway to a vast array of functional groups including carboxylic acids, amides, and amines. Patent CN116332794A introduces a transformative preparation method for aryl nitrile compounds (Compound II) that addresses long-standing safety and efficiency challenges in the industry. By leveraging a palladium-catalyzed system utilizing potassium ferrocyanide trihydrate (K4[Fe(CN)6]·3H2O) as a non-toxic cyanide source, this technology enables the production of high-purity intermediates under remarkably mild conditions. The core innovation lies in the precise orchestration of palladium salts, specialized phosphine ligands, and biphasic solvent systems to achieve superior intermediate process control (IPC) purity.
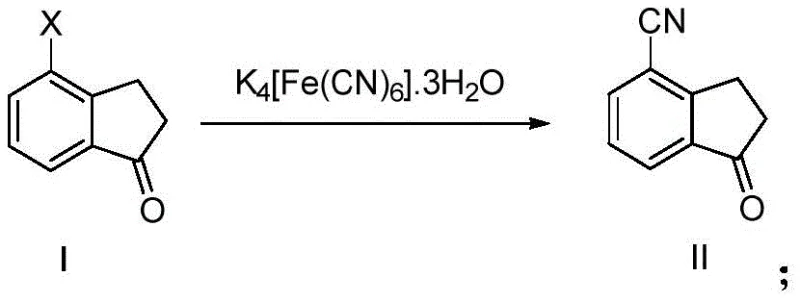
This methodology stands in stark contrast to legacy processes that rely on hazardous reagents and extreme operating parameters. For procurement and supply chain leaders, the adoption of this protocol signifies a strategic shift towards greener, safer, and more cost-effective manufacturing pathways for complex aromatic nitriles. The ability to utilize water in conjunction with organic solvents not only reduces the environmental footprint but also simplifies downstream processing, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aryl nitriles has been plagued by significant safety hazards and operational inefficiencies inherent to traditional chemistries. The classic Sandmeyer reaction and Rosenmund-von Braun reaction, while chemically robust, necessitate the use of stoichiometric amounts of copper(I) cyanide (CuCN), a reagent known for its severe toxicity and tendency to generate substantial heavy metal waste. Furthermore, these reactions often require harsh conditions that limit substrate tolerance and result in difficult purification profiles due to the formation of copper complexes.
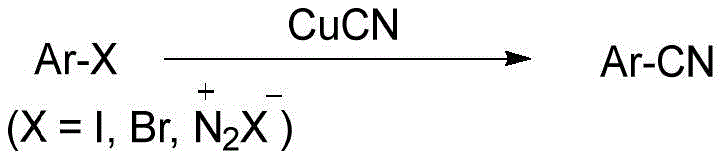
Alternatively, the ammoxidation process, which converts toluene derivatives directly to nitriles using ammonia and oxygen, demands extremely high temperatures and pressures. This imposes rigorous equipment specifications, escalating capital expenditure (CAPEX) and increasing the risk profile of the operation. Additionally, the substrate scope for ammoxidation is narrow, restricting its utility for synthesizing diverse, functionalized aryl nitriles required in modern drug discovery. These conventional methods collectively represent a bottleneck in cost reduction in API manufacturing, driving the urgent need for catalytic alternatives that decouple yield from hazard.
The Novel Approach
The novel approach detailed in the patent data circumvents these pitfalls by employing a transition metal-catalyzed cyanation strategy that prioritizes atom economy and operator safety. Instead of free cyanide ions or toxic copper salts, the process utilizes potassium ferrocyanide, a stable and low-toxicity coordination complex that releases cyanide slowly and controllably within the catalytic cycle. This "slow-release" mechanism effectively mitigates the risk of catalyst poisoning by excess cyanide, a common failure mode in palladium-catalyzed cyanations.
Moreover, the reaction operates at moderate temperatures ranging from 80°C to 120°C, eliminating the need for high-pressure autoclaves required by ammoxidation. The versatility of the system is further enhanced by its compatibility with a wide range of leaving groups, including bromides, chlorides, iodides, and sulfonates, as illustrated in the substrate scope definitions. This broad applicability ensures that the method can be seamlessly integrated into the synthesis of diverse chemical libraries, providing a reliable aryl nitrile supplier with the flexibility to handle complex molecular architectures without compromising on safety or yield.
Mechanistic Insights into Palladium-Catalyzed Cyanation
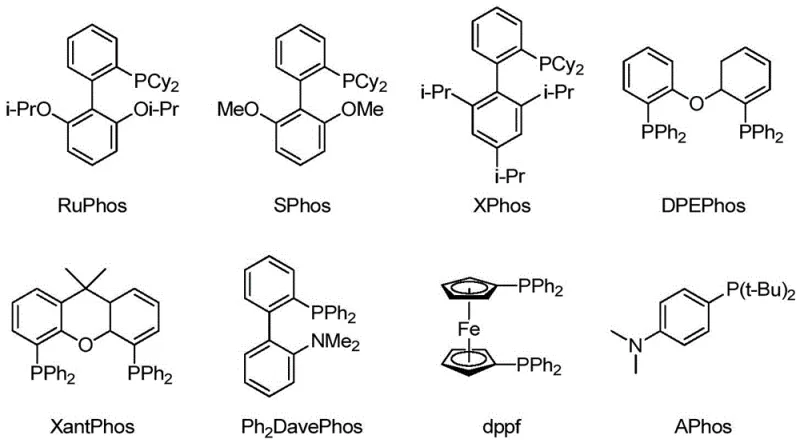
At the heart of this technological advancement is a sophisticated palladium catalytic cycle that balances oxidative addition, ligand exchange, and reductive elimination with high precision. The reaction initiates with the oxidative addition of the aryl halide (Compound I) to the active Pd(0) species, a step that is critically dependent on the electronic and steric properties of the supporting phosphine ligand. The patent data highlights that the choice of ligand is not merely incidental but is the primary determinant of reaction success, dictating the stability of the catalytic intermediate and the rate of turnover.
Following oxidative addition, the cyanide transfer occurs via ligand exchange with the ferrocyanide anion. Unlike free cyanide salts which can flood the coordination sphere and deactivate the metal center, the ferrocyanide complex provides a buffered source of CN⁻ ions. This controlled delivery prevents the formation of inactive Pd-CN clusters, thereby maintaining high catalytic activity throughout the extended reaction times of 18 to 24 hours. The final reductive elimination step releases the desired aryl nitrile product (Compound II) and regenerates the Pd(0) catalyst, closing the loop. The inclusion of water in the solvent system plays a dual role: it facilitates the solubility of the inorganic cyanide source and may assist in stabilizing charged intermediates within the catalytic cycle.
Impurity control is another critical aspect where this mechanism excels. By avoiding stoichiometric copper reagents, the process eliminates the formation of tenacious copper-organic complexes that are notoriously difficult to remove to ppm levels. The use of specific ligand-solvent pairings, such as Ph2DavePhos in DMAc or dppf in t-BuOH, further suppresses side reactions like homocoupling or hydrodehalogenation. This results in a crude reaction profile with significantly higher purity, reducing the burden on downstream purification units and ensuring that the final material meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Aryl Nitrile Compounds Efficiently
The practical implementation of this synthesis route requires careful attention to the specific pairing of solvents and ligands to maximize efficiency. As demonstrated in the experimental data, the reaction is not a "one-size-fits-all" process; rather, it relies on optimized conditions where the solvent polarity and ligand bite angle are matched to the specific substrate electronics. For instance, when using DMAc as the solvent, ligands like Ph2DavePhos or RuPhos drive the reaction to near-quantitative conversion, whereas in 1,4-dioxane, S-Phos or dppf proves superior. This nuanced understanding allows process chemists to tailor the reaction environment for optimal performance.
- Prepare the reaction mixture by dissolving the aryl halide substrate (Compound I), palladium salt (e.g., PdCl2), and the specific phosphine ligand in a suitable organic solvent such as DMAc, t-BuOH, or 1,4-dioxane.
- Add an aqueous solution of potassium ferrocyanide trihydrate (K4[Fe(CN)6]·3H2O) to the reaction vessel, ensuring the molar ratio of substrate to cyanide source is optimized (typically 1: 0.5).
- Heat the sealed reaction mixture to a temperature between 80°C and 120°C (optimally around 100°C) and stir for 18 to 24 hours to ensure complete conversion before workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this palladium-catalyzed ferrocyanide method offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By replacing highly toxic KCN or NaCN with the benign K4[Fe(CN)6]·3H2O, facilities can significantly reduce the costs associated with hazardous material handling, storage, and emergency response protocols. This shift directly contributes to cost reduction in fine chemical manufacturing by lowering the regulatory compliance burden and insurance premiums associated with toxic reagent usage.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal reagents like CuCN removes the need for expensive and complex heavy metal scavenging steps during workup. In traditional processes, removing residual copper to meet regulatory limits often requires multiple filtration stages or specialized resin treatments, which consume time and materials. By utilizing a catalytic amount of palladium and a non-toxic cyanide source, the new method streamlines the purification workflow, leading to substantial cost savings in both consumables and labor. Furthermore, the high conversion rates observed (often exceeding 90% in optimized examples) minimize the loss of valuable starting materials, enhancing overall material efficiency.
- Enhanced Supply Chain Reliability: The reagents employed in this protocol, including palladium salts, phosphine ligands, and potassium ferrocyanide, are commercially available commodities with robust global supply chains. Unlike specialized or restricted reagents that may face export controls or availability fluctuations, these materials ensure a stable and continuous supply for long-term production campaigns. The mild reaction conditions also mean that the process can be executed in standard glass-lined or stainless steel reactors without the need for exotic high-pressure equipment, increasing the number of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a significant leap forward in sustainability. The generation of hazardous waste is minimized due to the absence of copper sludge and free cyanide in the effluent. The use of water as a co-solvent further aligns with green chemistry principles, reducing the volume of organic solvent waste that requires incineration or recovery. This improved environmental profile facilitates easier permitting for new production lines and supports corporate sustainability goals, making the supply of these intermediates more resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyanation technology. These answers are derived directly from the experimental findings and comparative data presented in the patent literature, providing a clear picture of the method's capabilities and limitations in a real-world manufacturing context.
Q: Why is potassium ferrocyanide preferred over traditional cyanide sources like KCN or CuCN?
A: Potassium ferrocyanide (K4[Fe(CN)6]·3H2O) is significantly less toxic than alkali metal cyanides (KCN, NaCN) and avoids the heavy metal pollution associated with stoichiometric copper cyanide (CuCN). It acts as a slow-release cyanide source, improving operational safety and reducing hazardous waste generation.
Q: What represents the key innovation in patent CN116332794A regarding ligand selection?
A: The patent identifies specific phosphine ligands (such as Ph2DavePhos, RuPhos, DPEPhos, and dppf) that are uniquely compatible with different solvent systems (DMAc, t-BuOH, 1,4-dioxane). This tailored ligand-solvent matching is critical for achieving high conversion rates and process control purity, overcoming the catalyst deactivation often seen with excess cyanide ions.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes mild reaction conditions (80-120°C) and commercially available reagents. The use of water as a co-solvent and the avoidance of highly toxic gases make the process inherently safer and more amenable to large-scale commercial manufacturing compared to traditional high-pressure ammoxidation or Sandmeyer reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Nitrile Supplier
The technological breakthroughs encapsulated in patent CN116332794A underscore the evolving landscape of aryl nitrile synthesis, moving decisively towards safer and more efficient catalytic methodologies. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these advanced laboratory protocols to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, ensuring that every batch of aryl nitrile intermediate we deliver meets the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this specific palladium-catalyzed route can be adapted to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this non-toxic cyanation method can optimize your bill of materials and reduce your overall environmental footprint. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for your next project, ensuring a seamless transition from development to commercial supply.
