Advanced Copper-Catalyzed Cyanation for Scalable Pharmaceutical Intermediate Production
Advanced Copper-Catalyzed Cyanation for Scalable Pharmaceutical Intermediate Production
The synthesis of aryl nitriles represents a cornerstone transformation in modern medicinal chemistry, serving as a critical gateway to a vast array of bioactive scaffolds including tetrazoles, amines, and carboxylic acids. As the demand for complex pharmaceutical intermediates continues to surge, the industry faces increasing pressure to adopt methodologies that balance high efficiency with rigorous safety standards. Patent CN102875275A, published in January 2013, introduces a transformative approach to this challenge by detailing a robust copper-catalyzed cyanation protocol. This technology replaces hazardous traditional cyanating agents with benzyl cyanide, offering a paradigm shift in how we approach the construction of the carbon-nitrogen triple bond. By leveraging inexpensive copper salts and ambient oxidative conditions, this method not only mitigates significant safety risks but also streamlines the purification workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
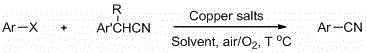
This proprietary methodology addresses the longstanding dichotomy between reactivity and safety in nucleophilic aromatic substitution. Traditional routes often rely on toxic reagents that require specialized containment infrastructure, whereas this novel copper-mediated pathway utilizes bench-stable solids and common organic solvents. For R&D teams focused on process intensification, the ability to conduct these reactions under air or oxygen at moderate temperatures (80 to 130°C) removes the necessity for inert atmosphere gloveboxes or high-pressure autoclaves. This accessibility translates directly into operational flexibility, allowing for rapid iteration during route scouting and seamless transition to pilot plant operations. The versatility of the system is further evidenced by its tolerance for diverse functional groups, ensuring that sensitive motifs present in advanced drug candidates remain intact throughout the cyanation process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aryl nitriles from aryl halides has been dominated by classical transformations such as the Sandmeyer reaction and the Rosenmund-von Braun reaction. While these methods established the foundational chemistry for nitrile synthesis, they are plagued by severe limitations that render them increasingly obsolete in a modern GMP environment. The primary concern is the reliance on stoichiometric amounts of highly toxic metal cyanides, such as potassium cyanide (KCN) or sodium cyanide (NaCN). Handling these reagents requires extreme caution, specialized waste treatment protocols, and extensive personnel training, all of which drive up the operational expenditure significantly. Furthermore, these traditional processes often necessitate harsh reaction conditions, including high temperatures and strong bases, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles that are difficult to separate.
Beyond safety and selectivity issues, conventional metal-catalyzed cyanation methods frequently employ expensive palladium catalysts coupled with sophisticated ligand systems. While effective, the cost of these precious metals and the difficulty in removing trace metal residues from the final API pose significant hurdles for procurement and quality control departments. The need for rigorous metal scavenging steps adds time and cost to the manufacturing process, extending the overall production timeline. Additionally, many of these legacy methods suffer from limited substrate scope, often failing to react efficiently with electron-rich aryl bromides or sterically hindered substrates. This lack of generality forces process chemists to develop custom solutions for each new molecule, hindering the standardization of manufacturing platforms and delaying time-to-market for critical therapies.
The Novel Approach
In stark contrast to these legacy challenges, the method disclosed in patent CN102875275A offers a streamlined and economically viable alternative by utilizing benzyl cyanide as the cyanide source. This organic nitrile acts as a safe, solid surrogate for toxic inorganic cyanides, effectively decoupling the synthetic utility of the cyano group from its inherent hazard profile. The reaction proceeds smoothly in common polar aprotic solvents such as DMF, DMSO, or NMP, utilizing inexpensive copper salts like cuprous oxide (Cu2O) or cuprous iodide (CuI) as catalysts. This shift from precious metals to base metals results in a drastic reduction in raw material costs, while the simplicity of the catalyst system facilitates easier removal during workup. The operational simplicity is further enhanced by the use of air or oxygen as the terminal oxidant, eliminating the need for exotic oxidizing agents.
The practical advantages of this novel approach extend deeply into the realm of process scalability and environmental compliance. By avoiding the generation of cyanide-containing wastewater streams, facilities can significantly reduce their environmental footprint and lower the costs associated with hazardous waste disposal. The reaction conditions are mild enough to preserve sensitive functionalities, as demonstrated by the successful conversion of substrates containing esters, amides, and ethers without significant side reactions. This functional group tolerance minimizes the need for protecting group strategies, thereby shortening the overall synthetic sequence. For supply chain managers, the availability of benzyl cyanide as a commodity chemical ensures a stable and reliable supply of the key reagent, mitigating the risks associated with the procurement of controlled or highly regulated toxic substances.
Mechanistic Insights into Copper-Catalyzed Cyanation
The mechanistic underpinning of this transformation involves a copper-catalyzed cross-coupling cycle that diverges from traditional palladium-mediated pathways. The reaction initiates with the activation of the aryl halide by the copper species, likely forming an aryl-copper intermediate through an oxidative addition or single-electron transfer process. Unlike palladium cycles that often require phosphine ligands to stabilize the metal center, this copper system appears to operate efficiently with simple salt precursors, suggesting a robust catalytic manifold that is less sensitive to ligand degradation. The benzyl cyanide then participates in a transmetallation-like step, transferring the cyano group to the copper center. This step is facilitated by the relatively acidic benzylic protons, which can be deprotonated under the reaction conditions to generate a nucleophilic species capable of interacting with the metal complex.
A critical feature of this mechanism is the role of molecular oxygen or air in sustaining the catalytic turnover. The copper catalyst, potentially cycling between Cu(I) and Cu(II) or higher oxidation states, requires re-oxidation to maintain activity throughout the prolonged heating period (8 to 16 hours). The presence of oxygen ensures that the active copper species is regenerated, preventing catalyst deactivation and driving the equilibrium towards product formation. This oxidative regime also helps in managing the fate of the benzyl byproduct, ensuring that the reaction proceeds cleanly to the desired aryl nitrile. Understanding this redox balance is crucial for R&D directors aiming to optimize reaction parameters, as controlling the oxygen flow rate can directly influence reaction kinetics and impurity formation.
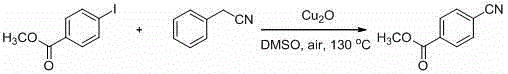
Impurity control in this system is inherently superior due to the chemoselectivity of the copper-benzyl cyanide interaction. Traditional cyanation methods often suffer from homocoupling of the aryl halide or hydrodehalogenation side reactions, which generate difficult-to-remove impurities. In this novel protocol, the specific coordination environment created by the solvent and the copper salt favors the cross-coupling pathway. For instance, as shown in the conversion of methyl 4-iodobenzoate to methyl 4-cyanobenzoate, the ester functionality remains completely intact, demonstrating the mildness of the conditions. The absence of strong bases or nucleophilic metal cyanides prevents saponification or other nucleophilic attacks on the ester group. This high level of selectivity simplifies downstream purification, often allowing for crystallization or simple chromatographic separation to achieve the high purity specifications required for pharmaceutical applications.
How to Synthesize Aryl Nitriles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to maximize yield and safety. The general procedure involves charging a reactor with the aryl halide substrate and benzyl cyanide in a precise molar ratio of 1:1.5, ensuring an excess of the cyanide source to drive the reaction to completion. Following the addition of the copper catalyst (typically 1 equivalent relative to the substrate) and the chosen high-boiling solvent, the mixture is heated to temperatures ranging between 80°C and 130°C. Maintaining an atmosphere of air or oxygen throughout the reaction duration of 8 to 16 hours is essential for catalyst turnover. Detailed standardized operating procedures for specific substrates, including exact workup and purification protocols, are provided in the technical guide below.
- Charge the reactor with aryl halide and benzyl cyanide in a 1: 1.5 molar ratio.
- Add 1 equivalent of copper salt catalyst (e.g., Cu2O, CuI) and organic solvent (e.g., DMF, DMSO).
- Heat the mixture to 80-130°C under air or oxygen atmosphere for 8-16 hours to complete the reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed cyanation technology offers substantial strategic benefits that extend beyond simple reaction yield. The primary value driver is the significant reduction in raw material costs achieved by substituting expensive palladium catalysts and toxic metal cyanides with commodity-grade copper salts and benzyl cyanide. This switch not only lowers the direct cost of goods sold (COGS) but also reduces the regulatory burden associated with the storage and handling of scheduled toxic substances. The simplified supply chain for reagents means fewer bottlenecks and a more resilient manufacturing process, capable of adapting quickly to fluctuations in market demand without being constrained by the availability of specialized catalysts.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive copper salts drastically lower the catalyst cost per kilogram of product. Furthermore, the avoidance of toxic metal cyanides removes the need for costly specialized waste treatment and disposal services, leading to substantial operational savings. The simplified workup procedure, which often avoids complex metal scavenging steps, reduces solvent consumption and processing time, contributing to a leaner and more cost-efficient manufacturing process overall.
- Enhanced Supply Chain Reliability: Benzyl cyanide and copper salts are widely available commodity chemicals with stable global supply chains, unlike specialized ligands or controlled cyanide salts which may face regulatory shipping restrictions. This accessibility ensures consistent production schedules and reduces the risk of supply disruptions. The robustness of the reaction conditions also allows for manufacturing in a broader range of facilities, providing greater flexibility in site selection and capacity planning for large-scale production campaigns.
- Scalability and Environmental Compliance: The use of air or oxygen as an oxidant eliminates the need for storing and handling hazardous chemical oxidants, improving site safety and reducing environmental liabilities. The process generates less hazardous waste, aligning with green chemistry principles and facilitating easier regulatory approval for new manufacturing sites. The thermal stability of the reaction mixture allows for safe scale-up from gram to ton scale without the exothermic risks often associated with traditional cyanation methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyanation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the method's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of this route for specific target molecules.
Q: Why is benzyl cyanide preferred over traditional metal cyanides in this process?
A: Benzyl cyanide serves as a non-toxic, solid cyanide source that eliminates the severe safety hazards associated with handling alkali metal cyanides like KCN or NaCN, significantly reducing EHS compliance costs.
Q: What types of aryl halides are compatible with this copper-catalyzed method?
A: The method demonstrates broad substrate scope, successfully converting both aryl iodides and aryl bromides, including those with electron-donating or electron-withdrawing substituents, into the corresponding nitriles.
Q: How does the use of air or oxygen impact the catalytic cycle?
A: The presence of air or oxygen acts as a terminal oxidant, facilitating the regeneration of the active copper species and driving the catalytic cycle forward without the need for expensive stoichiometric oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Nitrile Supplier
The technological advancements detailed in patent CN102875275A underscore the potential for safer and more efficient synthesis of critical pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative methodologies for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aryl nitrile intermediate meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to evaluate the feasibility of this copper-catalyzed route for your pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for similar compounds and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes safety, cost-efficiency, and reliability, empowering your organization to bring life-saving medicines to market faster and more economically.
