Advanced Recrystallization Technology for High Purity Mirabegron Manufacturing and Supply
The pharmaceutical landscape for treating overactive bladder (OAB) demands exceptionally high standards of chemical purity, particularly for beta-3 adrenergic agonists like Mirabegron. As detailed in patent CN111072589A, a significant technological breakthrough has been achieved in the post-synthetic purification of this critical active pharmaceutical ingredient. The core innovation lies in a sophisticated recrystallization methodology that specifically targets the removal of the persistent and structurally analogous impurity known as imA. Traditional synthesis pathways often struggle with this contaminant due to its nearly identical polarity to the parent compound, creating bottlenecks in manufacturing efficiency. This new approach leverages precise solvent engineering to manipulate crystal polymorphism, converting the less desirable beta-form into the highly pure alpha-form while leaving impurities in solution. For global procurement teams and R&D directors, this represents a pivotal shift towards more robust and scalable production capabilities for high-purity pharmaceutical intermediates.
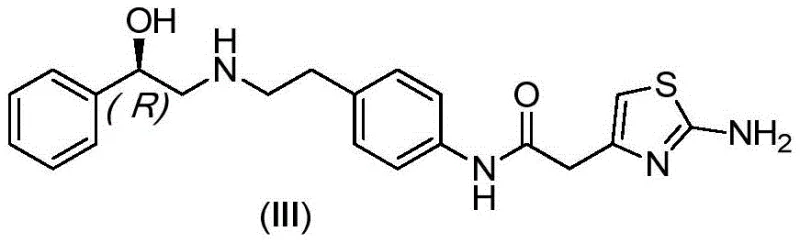
Mirabegron, chemically defined as (R)-2-(2-aminothiazol-4-yl)-N-[4-[2-[(2-hydroxy-2-phenylethyl)amino]ethyl]phenyl]acetamide, serves as a vital therapeutic agent. The molecular architecture, as depicted in the structural formula, contains multiple hydrogen bond donors and acceptors which complicate purification. The presence of the chiral center at the hydroxyethyl group further necessitates stringent control over stereochemical integrity during processing. The ability to consistently deliver this molecule with impurity profiles below regulatory thresholds is a defining capability for any reliable API supplier. The patented method addresses the specific challenge of the imA byproduct, which arises from double condensation reactions during the upstream synthesis, ensuring that the final drug substance meets the rigorous safety standards required by agencies like the FDA and EMA.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mirabegron has relied on routes involving the condensation of chiral precursors with nitrophenylethylamines followed by reduction steps. A pervasive issue in these conventional schemes, whether starting from styrene oxide or mandelic acid derivatives, is the formation of the bis-acylated impurity imA. This byproduct is generated when primary amines resulting from nitro reduction or amide reduction participate in secondary condensation reactions with excess acylating agents. The chemical structure of impurity imA is remarkably similar to Mirabegron, differing primarily by the addition of another thiazole-acetic acid moiety. This structural homology results in nearly overlapping retention times in chromatographic systems and very similar solubility characteristics in standard organic solvents. Consequently, removing imA typically requires extensive column chromatography or multiple iterative recrystallizations, which drastically increases production costs, extends lead times, and reduces overall process yield. For supply chain managers, these inefficiencies translate into higher raw material consumption and unpredictable delivery schedules.
The Novel Approach
The innovative recrystallization technique described in the patent data fundamentally alters the purification paradigm by exploiting subtle differences in crystal lattice energy and solvation dynamics. Instead of attempting to separate the impurity through complex chromatographic means, this method utilizes a binary solvent system comprising an alcohol, specifically methanol, and dichloromethane. By carefully tuning the volume ratio of these solvents, typically between 1:10 and 1:40, the process creates an environment where the solubility of the target Mirabegron is significantly depressed relative to the impurity. Crucially, the solvent mixture induces a polymorphic transition. The crude material, often existing as the beta-crystal form, dissolves and rapidly converts to the thermodynamically more stable alpha-crystal form upon cooling. This phase transition is accompanied by a sharp decrease in solubility for the alpha-form, causing it to precipitate selectively. Meanwhile, the impurity imA remains highly soluble in this specific methanol-dichloromethane matrix, effectively staying in the mother liquor. This elegant physical separation mechanism bypasses the need for expensive stationary phases and simplifies the downstream processing workflow considerably.
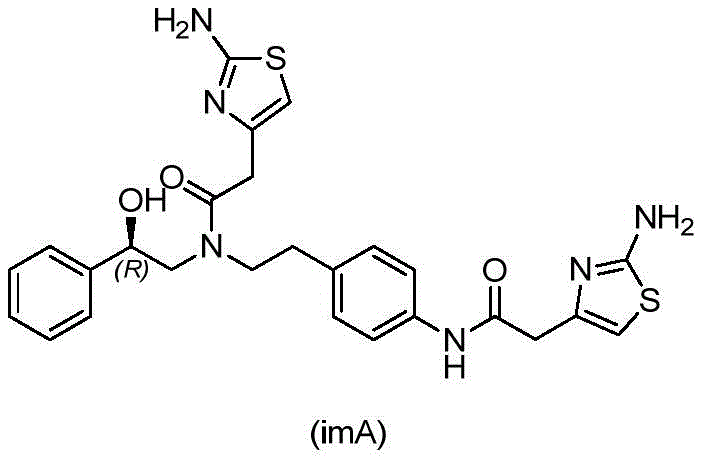
Mechanistic Insights into Solvent-Mediated Polymorphic Transformation
The success of this purification strategy hinges on the intricate interplay between solvent polarity, hydrogen bonding networks, and crystal nucleation kinetics. Methanol acts as a protic solvent capable of disrupting the existing hydrogen bonds in the beta-crystal lattice of the crude Mirabegron. When heated to reflux temperatures, typically around 30 to 40 degrees Celsius given the boiling point of the mixture, the crude solid fully dissolves. At this stage, the molecules are free to reorganize. Upon the introduction of dichloromethane, a non-polar aprotic solvent, the overall dielectric constant of the medium changes. This specific solvent blend destabilizes the solvation shell around the alpha-polymorph less than it does for the impurity. As the solution cools naturally to room temperature, the supersaturation point for the alpha-form of Mirabegron is reached first. The nucleation of alpha-crystals is energetically favorable in this environment, leading to the growth of large, well-defined crystals. The impurity imA, possessing different steric bulk and electronic distribution due to the extra thiazole ring, interacts differently with the solvent molecules. Its solubility curve does not intersect with the precipitation conditions of the alpha-Mirabegron in this specific ratio, allowing it to remain in solution even as the product crystallizes out. This differential solubility is the key driver for the high purity levels observed, often exceeding 99.9 percent.
Furthermore, the control of the cooling rate and stirring speed plays a vital role in determining the final particle size distribution and purity. Rapid cooling might trap impurities within the crystal lattice or induce oiling out, whereas the patented method advocates for natural cooling with moderate stirring, such as 150 rpm. This gentle approach allows for the orderly assembly of the crystal lattice, rejecting impurity molecules that do not fit the geometric constraints of the alpha-form unit cell. The result is a solid product where the internal crystal structure is highly ordered and free from the inclusion of the imA byproduct. Analytical data confirms that this method consistently reduces imA content to levels as low as 0.02 percent, a significant improvement over standard techniques. For R&D teams, understanding this mechanism allows for better scale-up predictions, as the thermodynamics of the polymorphic transition are reproducible across different batch sizes provided the solvent ratios and thermal profiles are maintained.
How to Synthesize High Purity Mirabegron Efficiently
Implementing this recrystallization protocol requires precise adherence to solvent ratios and thermal conditions to ensure the successful conversion from beta to alpha polymorphs. The process begins with the preparation of the binary solvent system, followed by the dissolution of the crude feedstock under reflux. The subsequent cooling phase is critical for maximizing yield while maintaining the exclusion of impurities. Detailed operational parameters regarding solvent volumes, stirring rates, and drying conditions are essential for reproducibility in a GMP environment. The following guide outlines the standardized steps derived from the patent examples to achieve optimal results in a manufacturing setting.
- Prepare a mixed solvent system comprising methanol and dichloromethane in a volume ratio ranging from 1: 10 to 1:40, preferably heating to a reflux temperature of 30 to 40 degrees Celsius.
- Dissolve the crude Mirabegron material into the heated solvent mixture under mechanical stirring, ensuring complete solvation to trigger the polymorphic transformation from beta to alpha crystal forms.
- Allow the solution to cool naturally to room temperature and stir for 2 to 4 hours to facilitate slow crystallization, then filter and dry the solids to obtain high-purity Mirabegron with minimized imA content.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced recrystallization technology offers substantial strategic benefits beyond mere chemical purity. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the reliance on preparative chromatography or multi-step recrystallizations with varying solvent systems, manufacturers can significantly reduce the consumption of expensive stationary phases and large volumes of diverse solvents. This streamlining directly translates to cost reduction in API manufacturing, as the operational expenditure associated with waste disposal, solvent recovery, and labor hours is markedly decreased. The ability to achieve high purity in a single crystallization step enhances the overall throughput of the production facility, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps removes a major cost center from the production budget. Chromatography resins are expensive consumables that require frequent replacement and generate significant hazardous waste. By shifting to a crystallization-based purification, the process relies on commodity solvents like methanol and dichloromethane, which are readily available and cost-effective. Furthermore, the high yield associated with this method ensures that valuable raw materials are not lost to inefficient separation processes, thereby optimizing the cost of goods sold. The reduction in processing time also lowers utility costs related to heating, cooling, and extended equipment occupancy, contributing to a leaner and more economically viable manufacturing model.
- Enhanced Supply Chain Reliability: Simplifying the synthesis and purification route inherently reduces the number of potential failure points in the supply chain. Complex multi-step purifications increase the risk of batch failures due to column channeling, solvent quality issues, or operational errors. A robust recrystallization process is easier to control and validate, leading to higher batch success rates and more consistent delivery schedules. Additionally, the solvents used are standard industrial chemicals with stable global supply chains, mitigating the risk of raw material shortages. This reliability is crucial for pharmaceutical companies that require uninterrupted supply to meet regulatory commitments and patient needs, ensuring that the reliable Mirabegron supplier can maintain continuous production without unexpected interruptions.
- Scalability and Environmental Compliance: The transition from chromatography to crystallization significantly improves the environmental footprint of the manufacturing process. Chromatographic methods generate large volumes of mixed solvent waste that are difficult and costly to treat. In contrast, the binary solvent system used in this recrystallization method is easier to separate and recycle, aligning with green chemistry principles. The process is also inherently more scalable; crystallization tanks can be easily enlarged to accommodate commercial scale-up of complex pharmaceutical intermediates without the engineering challenges associated with scaling up chromatographic columns. This scalability ensures that as demand for Mirabegron grows, production capacity can be expanded efficiently while maintaining strict adherence to environmental regulations regarding waste discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recrystallization technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, offering clarity on process robustness and quality outcomes. Understanding these details helps stakeholders make informed decisions regarding technology transfer and vendor qualification.
Q: Why is impurity imA difficult to remove from Mirabegron using standard methods?
A: Impurity imA is a bis-condensation byproduct formed during synthesis that possesses chemical polarity extremely similar to the target Mirabegron molecule. This similarity makes traditional separation techniques like chromatography inefficient and costly, necessitating specialized recrystallization strategies.
Q: How does the methanol and dichloromethane solvent system improve purity?
A: This specific binary solvent system uniquely facilitates the rapid conversion of Mirabegron from its beta crystal form to the more stable alpha form. The solubility profile of this mixture allows the target alpha-crystal Mirabegron to precipitate slowly while keeping the structurally similar imA impurity dissolved in the mother liquor.
Q: What represents the optimal solvent ratio for maximum yield and purity?
A: Experimental data indicates that a volume ratio of methanol to dichloromethane between 1:20 and 1:30 offers the best balance. This range ensures sufficient solubility for dissolution at reflux while maximizing the precipitation of pure alpha-crystals upon cooling, effectively reducing imA content to below 0.1 percent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirabegron Supplier
The technological advancements in Mirabegron purification underscore the importance of partnering with a manufacturer that possesses deep process chemistry expertise. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Mirabegron meets the highest international standards. We understand that the transition from laboratory scale to commercial manufacturing requires not just chemical knowledge but also engineering excellence. Our facilities are equipped to handle the specific solvent systems and thermal controls required for this advanced recrystallization process, guaranteeing a consistent supply of high-quality material for your pharmaceutical formulations.
We invite you to collaborate with us to optimize your supply chain for this critical OAB medication. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By engaging with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of our manufacturing capabilities. Let us help you secure a stable, cost-effective, and high-purity supply of Mirabegron, ensuring your products reach the market with confidence and compliance.
