Advanced Synthetic Route for 2-Isopropyl-3-Amino-4-Methylpyridine: Enhancing Purity and Scalability
Introduction to Patent CN113666865A
The pharmaceutical industry's relentless pursuit of effective oncology treatments has placed significant focus on KRAS G12C inhibitors, a class of drugs showing immense promise for treating pancreatic, colorectal, and lung cancers. Central to the manufacturing of these life-saving therapeutics is the high-quality synthesis of key intermediates, specifically 2-isopropyl-3-amino-4-methylpyridine. A groundbreaking patent, CN113666865A, filed in late 2021, introduces a revolutionary synthetic methodology that addresses critical bottlenecks in existing production routes. This innovation shifts the paradigm from hazardous, low-yield coupling reactions to a streamlined condensation and cyclization strategy. By leveraging inexpensive starting materials like ethyl isobutyrate and employing a novel L-alanine catalytic system, the disclosed method achieves exceptional purity levels exceeding 99.7%. For global procurement teams and R&D directors, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while adhering to stringent environmental standards.
The technical significance of this patent lies not merely in the final product but in the robustness of the entire process architecture. Traditional methods often struggle with impurity profiles that complicate downstream purification, leading to substantial material loss. The approach detailed in CN113666865A mitigates these risks through careful control of reaction conditions, such as maintaining specific pH levels during condensation and utilizing mild alkaline conditions for the final transformation. This ensures that the resulting 2-isopropyl-3-amino-4-methylpyridine is suitable for immediate use in sensitive coupling reactions required for API synthesis. Furthermore, the scalability of this route has been rigorously validated, demonstrating that the transition from laboratory benchtop to commercial kilogram-scale production can be achieved without compromising safety or efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-isopropyl-4-methylpyridin-3-amine was fraught with significant technical and economic challenges. Existing literature, such as WO2020/81636 and US2019/374542, describes routes relying on the bromination of 3-amino-4-methylpyridine followed by coupling with organometallic reagents like isopropyl zinc bromide or isopropyl magnesium chloride. These Grignard-type reactions are inherently hazardous due to the high reactivity of the reagents and require strict anhydrous conditions, increasing operational complexity and cost. Moreover, alternative pathways involving palladium-catalyzed coupling with boronic acid esters introduce the burden of removing trace heavy metals, a critical quality attribute for pharmaceutical ingredients. Another referenced method, described in WO2021097207, attempted to utilize ethyl isobutyrate but failed to optimize the catalytic system.
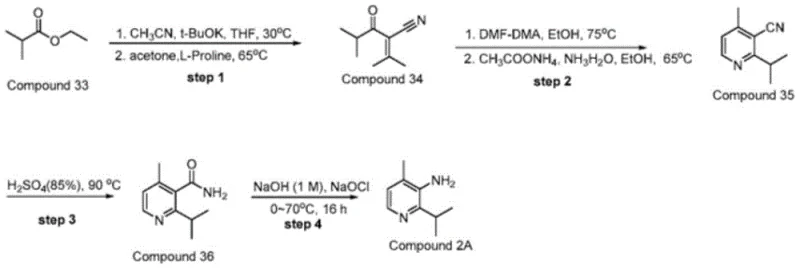
A critical analysis of the WO2021097207 route reveals severe deficiencies that hinder industrial adoption. Specifically, the use of L-proline as a catalyst for the condensation step proved ineffective, often stalling the reaction unless large quantities of molecular sieves were added, yet still resulting in suboptimal yields. Furthermore, the subsequent hydrolysis step in this prior art relied on harsh acidolysis using 85% sulfuric acid at 90°C. This aggressive condition frequently caused carbonization of the intermediate, turning the reaction mixture black and generating intractable tars that reduced overall yield to unacceptable levels. The neutralization of such large volumes of strong acid also generates massive amounts of saline wastewater, posing a significant environmental liability for manufacturers aiming for green chemistry compliance.
The Novel Approach
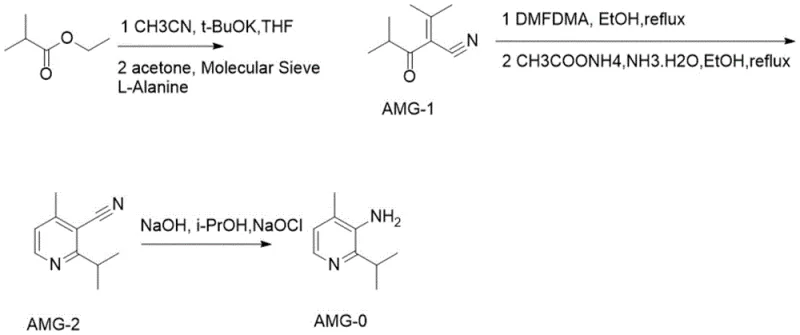
The methodology presented in CN113666865A offers a decisive break from these inefficiencies by introducing a completely redesigned synthetic logic. The most striking improvement is the substitution of L-proline with L-alanine in the initial condensation phase. This seemingly minor change results in a dramatic enhancement of catalytic activity, allowing the reaction to proceed smoothly to form 2-isobutyryl-3-methyl-2-butenenitrile with high selectivity. The process eliminates the need for excessive dehydrating agents and operates under milder conditions, thereby preserving the integrity of the sensitive nitrile functionality. Additionally, the cyclization step utilizes ammonium acetate as a nitrogen source, which is far more cost-effective and easier to handle than ammonia gas or complex amine sources, facilitating safer commercial scale-up of complex pharmaceutical intermediates.
Perhaps the most impactful innovation is the replacement of acid hydrolysis with a base-mediated pathway. By employing sodium hydroxide for the hydrolysis of the nitrile group, the process avoids the carbonization issues entirely. The resulting amide intermediate can then be seamlessly subjected to Hofmann degradation in a one-pot or stepwise manner using sodium hypochlorite. This alkaline environment is not only gentler on the molecular structure but also simplifies the workup procedure, as it negates the need for extensive acid neutralization and the associated waste treatment. Consequently, this novel approach delivers a cleaner product profile with significantly reduced impurity loads, making it an ideal solution for cost reduction in API manufacturing where purification costs often dominate the budget.
Mechanistic Insights into L-Alanine Catalyzed Condensation and Cyclization
The core of this synthetic breakthrough lies in the mechanistic efficiency of the L-alanine catalyzed aldol-type condensation. In the first stage, ethyl isobutyrate reacts with acetonitrile in the presence of potassium tert-butoxide to form a beta-keto nitrile intermediate. Unlike the sluggish performance observed with L-proline, L-alanine appears to facilitate a more favorable enolization and subsequent nucleophilic attack on acetone. The presence of molecular sieves in this step serves to sequester water generated during the condensation, driving the equilibrium towards the desired 2-isobutyryl-3-methyl-2-butenenitrile product. This precise control over the reaction equilibrium is crucial for preventing the formation of polymeric byproducts that typically plague such condensations. The resulting enone-nitrile structure is perfectly poised for the subsequent ring-closing reaction, demonstrating a high degree of atom economy.
Following the formation of the linear precursor, the construction of the pyridine ring proceeds via a methylene condensation using DMF-DMA (N,N-Dimethylformamide dimethyl acetal). This reagent effectively converts the active methylene group into an enamine intermediate, which then undergoes cyclocondensation with ammonium acetate. The mechanism involves the nucleophilic attack of ammonia on the electrophilic carbons, followed by dehydration and aromatization to yield the stable pyridine core of 2-isopropyl-4-methyl-3-cyanopyridine. The final transformation involves the hydrolysis of the nitrile to an amide, followed by the Hofmann rearrangement. In this critical step, the amide nitrogen is brominated or chlorinated in situ by sodium hypochlorite under basic conditions, leading to the migration of the isopropyl-substituted carbon group and the expulsion of carbonate, ultimately furnishing the primary amine with retention of configuration at the chiral center if applicable, although here the isopropyl group provides steric bulk that stabilizes the transition state.

How to Synthesize 2-Isopropyl-3-Amino-4-Methylpyridine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters disclosed in the patent to ensure maximum yield and purity. The process begins with the preparation of the key enone-nitrile intermediate, where temperature control during the addition of acetonitrile is vital to prevent exothermic runaway. Following the isolation of this intermediate, the cyclization step must be conducted under reflux in ethanol to ensure complete conversion of the DMF-DMA adduct. The final Hofmann degradation is particularly sensitive to temperature; the addition of sodium hypochlorite must be performed below 5°C to prevent over-oxidation, followed by a controlled warm-up to 70°C to drive the rearrangement to completion. Detailed standard operating procedures for each unit operation are essential for reproducibility.
- Condense ethyl isobutyrate with acetonitrile using t-BuOK, followed by aldol condensation with acetone catalyzed by L-alanine and molecular sieves to form 2-isobutyryl-3-methyl-2-butenenitrile.
- Perform methylene condensation using DMF-DMA, followed by cyclization with ammonium acetate in ethanol to construct the pyridine ring, yielding 2-isopropyl-4-methyl-3-cyanopyridine.
- Execute alkaline hydrolysis using sodium hydroxide followed by one-pot Hofmann degradation with sodium hypochlorite to convert the nitrile group directly to the primary amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond simple technical metrics. The primary advantage is the drastic simplification of the raw material portfolio. By shifting away from specialized organometallic reagents like isopropyl zinc bromide and noble metal catalysts, manufacturers can source all inputs from bulk chemical suppliers. This diversification of the supply base significantly mitigates the risk of shortages and price volatility associated with niche reagents. Furthermore, the elimination of palladium catalysts removes the necessity for expensive scavenging resins and rigorous heavy metal testing, which are costly and time-consuming steps in the quality control workflow. This streamlining directly translates to a leaner manufacturing process with reduced operational overhead.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the replacement of high-cost reagents with commodity chemicals. Ethyl isobutyrate, acetone, and ammonium acetate are produced on a massive global scale, ensuring stable pricing and availability. Additionally, the switch from L-proline to L-alanine reduces catalyst costs, while the avoidance of sulfuric acid carbonization prevents the loss of valuable intermediates, thereby improving the overall mass balance. The one-pot nature of the final hydrolysis and degradation steps further reduces solvent consumption and energy usage, contributing to substantial cost savings in utility and waste disposal budgets without compromising output quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance of the L-alanine catalytic system and the mild alkaline hydrolysis means that the process is less susceptible to minor fluctuations in raw material quality or environmental conditions. This reliability ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted API production schedules. Moreover, the simplified workup procedures reduce the turnaround time between batches, allowing facilities to increase throughput and respond more agilely to market demand surges for KRAS inhibitor therapies.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern green chemistry principles. The absence of heavy metals and the reduction of acidic wastewater generation simplify the effluent treatment process, lowering the environmental footprint of the manufacturing site. The high selectivity of the reaction minimizes the formation of hazardous byproducts, reducing the volume of chemical waste requiring incineration. These factors make the technology highly scalable, as regulatory hurdles for expansion are lower when the process is inherently cleaner and safer, facilitating rapid capacity increases to meet global pharmaceutical needs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these nuances is essential for process chemists and engineers tasked with technology transfer. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why is L-alanine preferred over L-proline for the condensation step?
A: Experimental data indicates that L-proline exhibits very poor catalytic activity for this specific substrate, often requiring molecular sieves and still resulting in low conversion rates. In contrast, L-alanine significantly improves reaction yield and selectivity without the need for excessive additives.
Q: What are the advantages of using alkaline hydrolysis over acid hydrolysis?
A: Traditional acid hydrolysis using concentrated sulfuric acid at high temperatures (90°C) leads to severe carbonization of intermediates, turning the reaction mixture black and drastically reducing yield. The alkaline method avoids this degradation, simplifies neutralization, and allows for a direct one-pot transition to Hofmann degradation.
Q: How does this route improve supply chain stability for KRAS inhibitors?
A: By eliminating the need for expensive and hazardous reagents like isopropyl zinc bromide, Grignard reagents, and noble metal catalysts, this route relies on commodity chemicals. This reduces raw material volatility and removes complex heavy metal removal steps, ensuring a more robust and continuous supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Isopropyl-3-Amino-4-Methylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation oncology drugs depends on the availability of high-quality intermediates. Our technical team has thoroughly analyzed the route disclosed in CN113666865A and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities that could affect downstream coupling reactions. We are committed to delivering 2-isopropyl-3-amino-4-methylpyridine that meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize your supply chain for KRAS G12C inhibitor projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this advanced synthetic route can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us partner with you to accelerate the delivery of life-saving therapies to patients worldwide through superior chemical manufacturing excellence.
