Advanced Iron-Catalyzed Synthesis of 2-Isopropyl-3-Amino-4-Methylpyridine for KRAS Inhibitor Production
Advanced Iron-Catalyzed Synthesis of 2-Isopropyl-3-Amino-4-Methylpyridine for KRAS Inhibitor Production
The pharmaceutical landscape has been revolutionized by the emergence of KRAS inhibitors, particularly following the breakthrough clinical results of AMG510 presented at the 2019 ASCO annual meeting. Central to the synthesis of these life-saving small molecules is the heterocyclic intermediate known as 2-isopropyl-3-amino-4-methylpyridine, designated as Compound I in patent CN112479993A. This specific chemical entity serves as an indispensable fragment in the construction of the drug molecule, necessitating a robust, scalable, and cost-effective manufacturing process. The patent discloses a novel synthetic methodology that addresses the critical bottlenecks of prior art, specifically focusing on the replacement of precious metal catalysts with abundant iron species and the strategic implementation of a Hofmann rearrangement. For global procurement teams and R&D directors, understanding the nuances of this patented route is essential for securing a reliable KRAS inhibitor intermediate supplier capable of meeting the rigorous demands of modern oncology drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridine core found in Compound I relied heavily on transition metal-catalyzed cross-coupling reactions that posed significant economic and operational challenges. As illustrated in the prior art referenced within the patent documentation, traditional routes often employed palladium catalysts such as Pd(dppf)Cl2 in conjunction with organozinc reagents like iPrZnBr to install the isopropyl group. This approach is inherently flawed for large-scale production due to the exorbitant cost of palladium and the sensitivity of the reaction to functional group tolerance. Furthermore, the precursor required for this coupling, often a brominated pyridine derivative like Compound XI, is not a commercially available commodity and suffers from poor synthetic efficiency, with literature reports indicating yields as low as 46% for its preparation. The reliance on such inefficient upstream steps creates a fragile supply chain, while the presence of amino groups in early intermediates can poison the expensive catalyst, leading to inconsistent batch quality and increased waste generation.
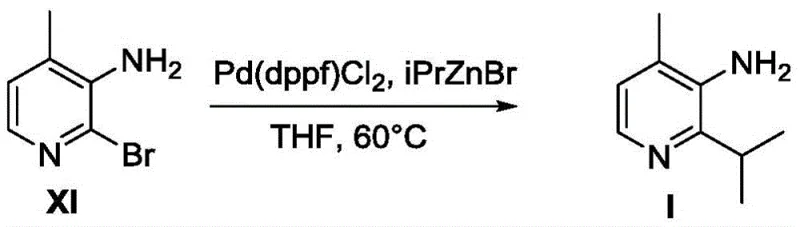
The Novel Approach
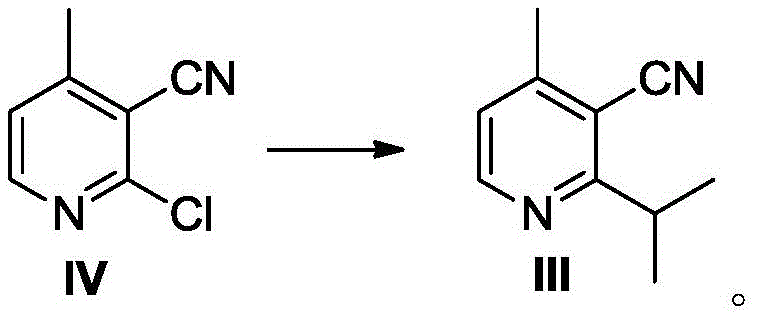
In stark contrast to the legacy methodologies, the innovative process described in CN112479993A redesigns the synthetic trajectory to bypass these economic and technical hurdles entirely. The core of this new strategy involves an iron-catalyzed cross-coupling reaction, often referred to in the art as a Panda coupling or iron-mediated Kumada-type coupling, which utilizes ferric triacetylacetate as a cheap and abundant catalyst source. By reacting 2-chloro-3-cyano-4-methylpyridine (Compound IV) with an isopropyl Grignard reagent in the presence of N-methyl pyrrolidone, the process achieves the installation of the isopropyl moiety with high efficiency. This shift from palladium to iron represents a paradigm shift in cost reduction in API manufacturing, as it eliminates the need for stringent heavy metal scavenging steps later in the process. Additionally, the route strategically delays the formation of the sensitive amino group until the final step, thereby preventing catalyst deactivation and ensuring a smoother reaction profile that is conducive to industrial scale-up.
Mechanistic Insights into Iron-Catalyzed Coupling and Hofmann Rearrangement
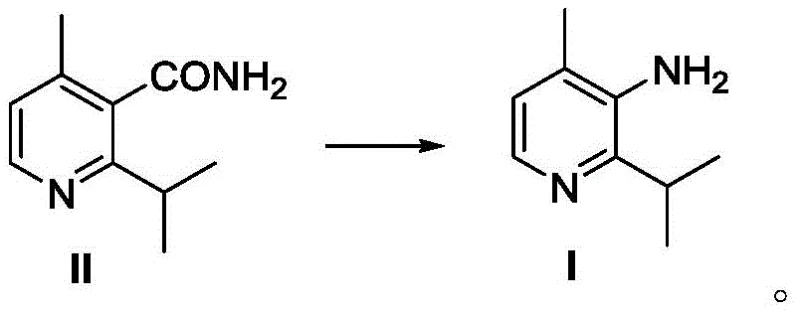
The mechanistic elegance of this synthesis lies in its two distinct phases: the construction of the carbon skeleton via iron catalysis and the functional group interconversion via rearrangement. In the initial coupling phase, the iron catalyst facilitates the transmetallation between the Grignard reagent and the chloropyridine substrate. Unlike palladium cycles which are prone to oxidative addition barriers with electron-deficient heterocycles, the iron system operates effectively under mild conditions (0-10°C), minimizing side reactions such as homocoupling or reduction. Following the successful formation of the nitrile intermediate (Compound III), the process employs a classic yet highly effective hydrolysis using concentrated sulfuric acid to generate the primary amide (Compound II). This sets the stage for the final transformation, where the amide undergoes a Hofmann rearrangement in the presence of sodium hypobromite (NaOBr). This rearrangement mechanism involves the migration of the alkyl group from the carbonyl carbon to the nitrogen atom with the loss of the carbonyl group as carbon dioxide, directly yielding the desired primary amine (Compound I) with high regioselectivity.
From an impurity control perspective, this sequence offers superior manageability compared to direct amination strategies. By maintaining the nitrogen functionality as a nitrile and subsequently an amide during the harsh coupling and hydrolysis conditions, the synthesis avoids the formation of complex polymeric byproducts that often arise from free anilines. The hydrolysis step is carefully monitored to ensure complete conversion of the nitrile without degrading the pyridine ring, while the final rearrangement is conducted in a biphasic or aqueous system that allows for easy extraction and purification. The use of silica gel column chromatography with ethyl acetate and n-heptane eluents, as detailed in the examples, ensures that the final high-purity heterocyclic intermediates meet the stringent specifications required for pharmaceutical applications. This meticulous control over the reaction pathway ensures that the impurity profile remains simple and predictable, a critical factor for regulatory approval.
How to Synthesize 2-Isopropyl-3-Amino-4-Methylpyridine Efficiently
The practical execution of this synthesis is designed for reproducibility and safety, utilizing standard laboratory and plant equipment. The process begins with the preparation of the iron catalyst mixture in tetrahydrofuran, followed by the controlled addition of the Grignard reagent to maintain thermal stability. Subsequent steps involve straightforward acidic hydrolysis and a base-mediated rearrangement, both of which are well-understood unit operations in fine chemical manufacturing. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures, are outlined below to guide process chemists in replicating this efficient route.
- Perform an iron-catalyzed coupling reaction between 2-chloro-3-cyano-4-methylpyridine (Compound IV) and isopropyl Grignard reagent to form Compound III.
- Hydrolyze the nitrile group of Compound III using concentrated sulfuric acid to obtain the amide intermediate, Compound II.
- Execute a Hofmann rearrangement on Compound II using NaOBr to convert the amide to the primary amine, yielding the final target Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary benefit is the drastic simplification of the raw material portfolio; by utilizing iron salts instead of palladium complexes and avoiding exotic organozinc reagents, the process leverages commodities that are available in bulk quantities globally. This shift significantly mitigates the risk of supply disruptions caused by the geopolitical volatility often associated with precious metal mining and refining. Furthermore, the elimination of expensive catalysts translates directly into substantial cost savings, allowing for more competitive pricing models in the long term without compromising on the quality of the final intermediate. The robustness of the chemistry also implies a higher success rate in production batches, reducing the financial impact of failed runs and wasted materials.
- Cost Reduction in Manufacturing: The substitution of palladium with iron catalysts removes one of the most significant cost drivers in heterogeneous catalysis. Since iron is orders of magnitude cheaper than palladium and does not require the same level of recovery infrastructure, the overall cost of goods sold (COGS) is significantly reduced. Additionally, the avoidance of zinc reagents, which can be hazardous and costly to handle, further streamlines the operational expenditure. The process design inherently lowers the barrier to entry for manufacturing, enabling facilities to produce high-purity intermediates without investing in specialized heavy metal removal systems, thus optimizing capital expenditure.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as 2-chloro-3-cyano-4-methylpyridine and isopropyl magnesium chloride, are established industrial chemicals with mature supply chains. This availability ensures that production schedules are not held hostage by the lead times of custom-synthesized precursors. By decoupling the synthesis from rare earth or precious metal dependencies, the supply chain becomes more resilient to market fluctuations. The ability to source reagents from multiple vendors enhances negotiation power and secures continuity of supply, which is paramount for maintaining the production timelines of critical oncology therapies.
- Scalability and Environmental Compliance: The reaction conditions described, ranging from 0°C to 105°C, are well within the operational limits of standard stainless steel reactors, facilitating seamless commercial scale-up of complex pyridine derivatives. The use of common solvents like tetrahydrofuran, ethyl acetate, and toluene simplifies solvent recovery and recycling processes, aligning with green chemistry principles. Moreover, the reduction in heavy metal usage lessens the environmental burden of waste disposal, ensuring compliance with increasingly strict environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of this KRAS inhibitor intermediate. These answers are derived directly from the technical disclosures and experimental data provided in the patent literature, offering clarity on the feasibility and benefits of the described methodology.
Q: Why is the iron-catalyzed route preferred over palladium coupling for this intermediate?
A: The iron-catalyzed route eliminates the need for expensive palladium catalysts and zinc reagents, significantly reducing raw material costs and simplifying the removal of heavy metal residues, which is critical for API compliance.
Q: How does the Hofmann rearrangement improve the synthesis compared to direct amination?
A: The Hofmann rearrangement allows for the introduction of the amino group at a late stage from a stable amide precursor, avoiding the catalyst poisoning issues associated with free amino groups during earlier coupling steps.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials, operates under relatively mild conditions, and avoids complex purification steps, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Isopropyl-3-Amino-4-Methylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation cancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. Our expertise in handling sensitive heterocyclic chemistry allows us to navigate the complexities of iron-catalyzed reactions and rearrangements with precision, guaranteeing a consistent supply of material for your clinical and commercial needs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your KRAS inhibitor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity pharmaceutical intermediates.
