Advanced Manufacturing of Rasagiline Intermediates via Chiral Auxiliary Resolution
Introduction to Next-Generation Rasagiline Manufacturing
The pharmaceutical landscape for Parkinson's disease treatment demands robust, scalable, and cost-effective synthesis routes for key active ingredients like Rasagiline. Patent CN101260048A introduces a transformative methodology that shifts the paradigm from traditional racemic resolution to a more efficient chiral auxiliary-driven synthesis. This approach leverages 1-indone as a readily available starting scaffold, reacting it with specific chiral amines to establish stereochemistry early in the process. By integrating asymmetric hydrogenation and streamlined alkylation steps, this technology addresses critical pain points in API manufacturing, specifically targeting high optical purity and reduced environmental footprint. For procurement leaders and R&D directors, understanding this pathway is essential for securing a reliable rasagiline intermediate supplier capable of meeting stringent global quality standards.
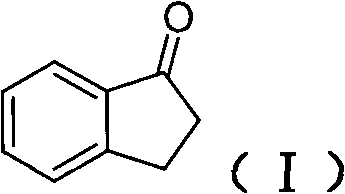
The strategic selection of 1-indone as the foundational raw material underscores the economic viability of this process. Unlike complex chiral pool starting materials that may suffer from supply chain volatility, 1-indone is a commodity chemical with stable pricing and widespread availability. The initial condensation reaction forms a chiral imine intermediate, setting the stage for highly selective reduction. This front-loading of chirality ensures that downstream processing does not require energy-intensive separation of enantiomers, thereby drastically simplifying the production workflow. Furthermore, the use of common industrial solvents such as ethyl acetate and toluene throughout the sequence enhances the safety profile and reduces the complexity of solvent recovery systems, making it an ideal candidate for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chiral amines like Rasagiline has relied heavily on the resolution of racemic mixtures, a process inherently flawed by its maximum theoretical yield of only 50%. Traditional methods often involve the formation of diastereomeric salts followed by multiple recrystallizations to achieve acceptable enantiomeric excess, resulting in significant material waste and increased processing time. Additionally, many legacy routes depend on precious metal catalysts such as Palladium on Carbon or Rhodium complexes, which not only inflate raw material costs but also introduce stringent requirements for heavy metal removal to meet ICH Q3D guidelines. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and a fragile supply chain susceptible to bottlenecks during the purification stages.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a chiral auxiliary strategy that bypasses the yield ceiling of racemic resolution. By reacting 1-indone with a chiral amine to form an imine, followed by asymmetric hydrogenation, the process achieves high stereocontrol without discarding half of the produced material. The substitution of expensive noble metals with Raney Nickel for the reduction step represents a significant cost reduction in Parkinson's drug manufacturing, as Raney Nickel is substantially cheaper and easier to handle on a multi-ton scale. Moreover, the synthetic route is designed to minimize unit operations; the transition from the reduced amine to the final propargylated product involves straightforward acid-base workups and filtrations rather than complex chromatographic separations. This streamlined approach not only accelerates the production cycle but also enhances the overall sustainability of the manufacturing process by reducing solvent consumption and waste generation.
Mechanistic Insights into Chiral Auxiliary Induction and Hydrogenation
The core of this synthetic innovation lies in the precise control of stereochemistry during the hydrogenation of the chiral imine intermediate. The reaction begins with the condensation of 1-indone and a chiral amine, such as (S)-phenylethylamine, to form a Schiff base. This imine serves as a temporary chiral mask that directs the facial selectivity of the subsequent hydrogen addition. When subjected to hydrogen pressure in the presence of Raney Nickel, the catalyst preferentially reduces the C=N bond from the less hindered face, dictated by the steric bulk of the chiral auxiliary. This mechanism ensures that the newly formed chiral center at the 1-position of the indane ring possesses the desired configuration with high fidelity. The robustness of this mechanistic pathway is evidenced by the ability to use relatively mild reaction conditions, typically between 0-50°C and moderate hydrogen pressures, which preserves the integrity of the sensitive indane skeleton.
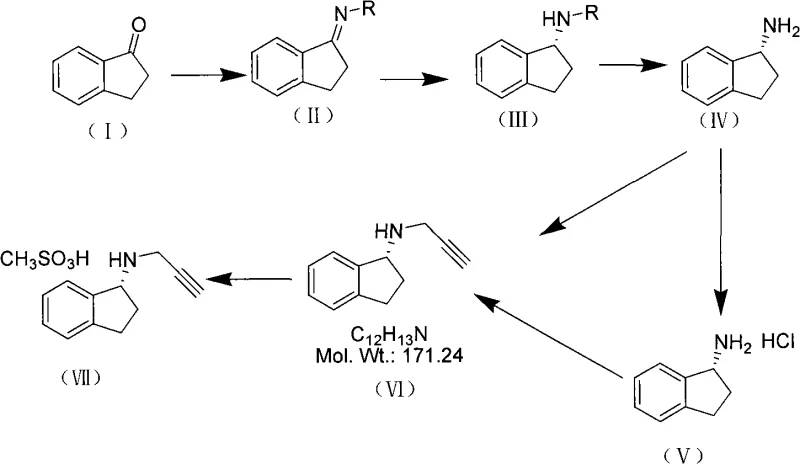
Following the establishment of the chiral center, the auxiliary must be removed to liberate the free amine for further functionalization. This is achieved through a carefully controlled hydrolysis step involving acid and base treatments, which cleaves the C-N bond of the auxiliary without racemizing the product. The resulting chiral amine is then converted into its hydrochloride salt to facilitate handling and purification. The final critical transformation involves the N-alkylation with 3-propargyl bromide. This step requires precise control of basicity and temperature to prevent over-alkylation or polymerization of the alkyne moiety. The use of alkali bases like potassium carbonate or triethylamine in polar aprotic solvents ensures clean conversion to the secondary amine. Finally, the formation of the mesylate salt is performed by treating the free base with methanesulfonic acid, yielding the thermodynamically stable crystalline form of Rasagiline suitable for pharmaceutical formulation. This comprehensive mechanistic understanding allows for tight control over impurity profiles, ensuring the final API meets rigorous regulatory specifications.
How to Synthesize Rasagiline Efficiently
The execution of this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure reproducibility and high yield. The process is divided into distinct operational phases: chiral imine formation, catalytic reduction, de-protection, alkylation, and salt formation. Each step utilizes common laboratory and plant equipment, such as standard reactors equipped with reflux condensers and hydrogenation autoclaves, making the technology transfer to manufacturing sites straightforward. The detailed标准化 synthesis steps见下方的指南 provide a granular view of the stoichiometry, solvent choices, and temperature profiles required to replicate the success of the pilot examples. Operators must pay particular attention to the moisture control during the imine formation and the hydrogen pressure regulation during the reduction phase to maximize efficiency.
- Condensation of 1-indone with a chiral auxiliary reagent to form the chiral imine intermediate.
- Catalytic hydrogenation of the imine using Raney Nickel to establish the chiral center.
- Hydrolysis and subsequent N-alkylation with propargyl bromide followed by salt formation with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings driven by the elimination of expensive chiral resolving agents and precious metal catalysts. By utilizing Raney Nickel and commodity chiral amines, the raw material bill of materials (BOM) is significantly optimized. Furthermore, the high yield reported in the patent examples suggests a drastic reduction in the amount of starting material required per kilogram of final API, directly impacting the bottom line. The simplicity of the workup procedures, which rely on filtration and extraction rather than column chromatography, translates to shorter batch cycles and higher throughput in existing manufacturing facilities. This efficiency gain allows suppliers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with Raney Nickel eliminates the need for costly metal recovery processes and reduces the risk of heavy metal contamination in the final product. Additionally, the use of inexpensive solvents like ethyl acetate and toluene, which are easily recycled, further lowers the operational expenditure. The high atom economy of the chiral auxiliary method means less waste disposal cost compared to resolution methods that discard 50% of the material. These factors combine to create a leaner, more cost-effective production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on 1-indone and common chiral amines ensures a stable supply of starting materials, as these are produced by multiple global vendors. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, reduces the risk of batch failures due to minor process deviations. This reliability is crucial for maintaining continuous supply to downstream formulation partners. Moreover, the scalability of the hydrogenation step using standard autoclaves means that production capacity can be easily ramped up from pilot scale to multi-ton commercial production without requiring specialized or bespoke equipment.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions and spent catalyst that can be treated or recycled according to standard environmental protocols. The absence of chlorinated solvents in the major reaction steps (except for optional extraction) aligns with green chemistry principles and simplifies regulatory compliance regarding VOC emissions. The ability to crystallize the final product directly from the reaction mixture or simple solvent swaps reduces solvent usage and energy consumption associated with distillation. This environmental friendliness not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rasagiline synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios. The answers reflect the balance between technical rigor and practical manufacturability that defines modern API production.
Q: What are the primary advantages of this chiral auxiliary method over traditional resolution?
A: This method avoids the theoretical 50% yield loss associated with racemic resolution by inducing chirality early in the synthesis via a chiral amine auxiliary, significantly improving overall atom economy.
Q: Which catalyst is utilized for the asymmetric reduction step?
A: The process employs Raney Nickel under moderate hydrogen pressure, which offers a cost-effective alternative to expensive noble metal catalysts like Palladium or Platinum while maintaining high stereoselectivity.
Q: How is the final purity of Rasagiline Mesylate ensured?
A: High optical purity is achieved through the initial chiral induction step, and final purification is facilitated by simple recrystallization from ether/alcohol mixtures, eliminating the need for complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rasagiline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value neurological therapeutics. Our team of expert chemists has extensively analyzed the chiral auxiliary methodology described in CN101260048A and possesses the technical capability to optimize this pathway for commercial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with advanced hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess. We are committed to delivering high-purity Rasagiline intermediates and API that comply with international pharmacopoeia standards.
We invite potential partners to engage with our technical sales team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this manufacturing process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming development programs. Let us collaborate to accelerate the delivery of affordable and high-quality Parkinson's treatments to patients worldwide.
