Advanced Synthetic Route for Rasagiline Mesylate Intermediates and Commercial Scale-Up
Advanced Synthetic Route for Rasagiline Mesylate Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks more efficient pathways for producing critical neuroprotective agents like Rasagiline, particularly for the treatment of Parkinson's disease. A significant technological breakthrough in this domain is detailed in patent CN102464589A, which discloses a novel preparation method for Rasagiline and its mesylate salt. Unlike conventional approaches that rely on the resolution of racemic mixtures—thereby discarding half of the synthesized material as waste—this innovative strategy employs a chiral pool synthesis starting from readily available (R)-(+)-1-indanamine. The core of this methodology involves a strategic protection-deprotection sequence using a trifluoroacetyl group, which effectively suppresses side reactions and ensures high optical purity throughout the synthesis. By integrating this advanced chemistry into our manufacturing capabilities, we offer a reliable rasagiline intermediate supplier solution that aligns with modern green chemistry principles and cost-efficiency goals.
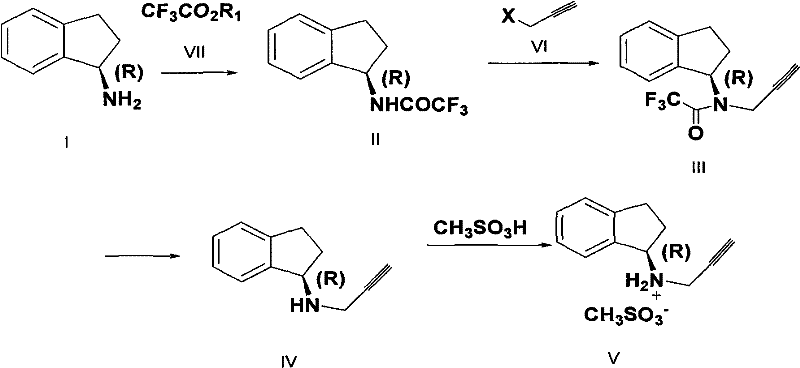
This comprehensive reaction scheme illustrates the seamless transition from the chiral starting material to the final active pharmaceutical ingredient salt. The process begins with the aminolysis of (R)-(+)-1-indanamine with a trifluoroacetate ester to form a stable amide intermediate. This intermediate then undergoes a highly selective N-alkylation with a halo-propyne derivative, followed by a mild hydrolysis step to reveal the free amine functionality of Rasagiline. Finally, salt formation with methanesulfonic acid yields the stable mesylate form. This linear progression minimizes unit operations and maximizes overall yield, addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing while maintaining stringent quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Rasagiline has been plagued by inherent inefficiencies associated with racemic synthesis and subsequent optical resolution. Traditional routes, such as those reported by Allegrini or Luo, typically involve the synthesis of racemic N-propargyl-1-indanamine followed by resolution using chiral acids like tartaric acid. This approach suffers from a fundamental theoretical yield limit of 50%, as the unwanted (S)-enantiomer is often discarded as chemical waste, creating significant environmental burdens and inflating raw material costs. Furthermore, alternative methods involving the reduction of imines or oximes frequently require hazardous reagents such as sodium borohydride or Raney nickel, generating substantial quantities of boron-containing waste or requiring complex catalyst removal steps. Another persistent challenge in direct alkylation strategies is the formation of N,N-di-propargyl impurities, which possess physical properties strikingly similar to the target product, making their removal via standard crystallization or distillation extremely difficult and costly.
The Novel Approach
The methodology presented in patent CN102464589A offers a transformative solution by bypassing the resolution step entirely through the use of enantiomerically pure starting materials. By initiating the synthesis with (R)-(+)-1-indanamine, the process inherently guarantees the stereochemical integrity of the final product without the need for wasteful separation techniques. The introduction of the trifluoroacetyl protecting group is a masterstroke of process chemistry; it temporarily reduces the nucleophilicity of the amine nitrogen, thereby kinetically favoring mono-alkylation over di-alkylation. This electronic modulation effectively eliminates the formation of the troublesome N,N-di-propargyl byproduct, streamlining the purification process. Moreover, the deprotection step utilizes mild basic hydrolysis, avoiding the harsh conditions or specialized reagents like thiophenol required in other protective group strategies (e.g., nitrobenzenesulfonyl groups), thus enhancing operator safety and environmental compatibility.
Mechanistic Insights into Trifluoroacetyl-Mediated Selective Alkylation
The success of this synthetic route hinges on the precise electronic control exerted by the trifluoroacetyl moiety during the alkylation phase. In a standard nucleophilic substitution reaction involving a free primary amine and a propargyl halide, the high nucleophilicity of the nitrogen often leads to poly-alkylation, where the mono-alkylated product reacts further to form a tertiary amine. However, when the amine is converted into a trifluoroacetamide, the strong electron-withdrawing nature of the trifluoromethyl group significantly delocalizes the lone pair electrons on the nitrogen atom into the carbonyl system. This reduced electron density renders the nitrogen a weaker nucleophile, sufficient to react with the electrophilic propargyl halide under basic conditions but insufficient to undergo a second alkylation event rapidly. This kinetic differentiation is crucial for achieving high chemical purity without resorting to complex chromatographic separations.
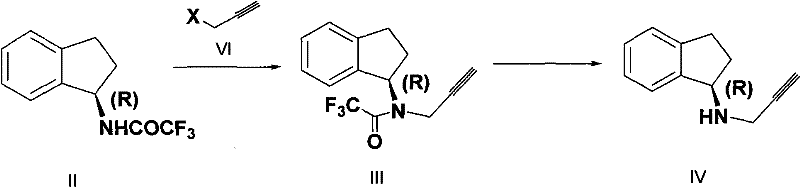
Following the alkylation, the restoration of the free amine is achieved through a base-catalyzed hydrolysis mechanism. The trifluoroacetyl group, while stable enough to withstand the alkylation conditions, is sufficiently labile to be cleaved by hydroxide ions in an alcoholic solvent. The mechanism involves the nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide, forming a tetrahedral intermediate that collapses to release the trifluoroacetate anion and the free Rasagiline amine. This step is remarkably clean, producing water-soluble trifluoroacetate salts that are easily separated from the organic product during the workup phase. The entire sequence demonstrates a sophisticated balance between stability and reactivity, ensuring that the chiral center at the 1-position of the indane ring remains untouched and racemization-free throughout the process.
How to Synthesize Rasagiline Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and optical purity. The process is divided into three distinct operational stages: protection, alkylation, and deprotection. Each stage utilizes common industrial solvents and reagents, facilitating easy technology transfer from laboratory to pilot plant. The following guide outlines the critical operational phases derived from the patent examples, providing a roadmap for process chemists aiming to replicate this high-efficiency route.
- Protect (R)-(+)-1-indanamine by reacting it with a trifluoroacetate ester (such as ethyl trifluoroacetate) to form the trifluoroacetamide intermediate.
- Perform N-alkylation on the protected intermediate using a halo-propyne (chloro- or bromo-propyne) in the presence of a mineral base and polar aprotic solvent.
- Hydrolyze the resulting N-propargyl-trifluoroacetamide using an aqueous base in an alcohol solvent to remove the protecting group and yield pure Rasagiline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of the optical resolution step fundamentally alters the cost structure of the API production. By utilizing 100% of the chiral starting material rather than discarding half of it, the effective cost of goods sold is drastically reduced. Furthermore, the avoidance of expensive chiral resolving agents, such as tartaric acid derivatives, and the removal of heavy metal catalysts like palladium or nickel from the process flow significantly lower raw material expenditure and waste disposal costs. The simplified purification train, driven by the high selectivity of the trifluoroacetyl protection, reduces the number of processing cycles and solvent consumption, leading to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The process eliminates the need for costly resolution agents and the associated loss of 50% of the material mass. By preventing the formation of hard-to-remove di-alkylated impurities, the need for expensive preparative chromatography or multiple recrystallizations is removed. The use of inexpensive mineral bases like sodium hydroxide or potassium carbonate instead of specialized organic bases further drives down the variable cost per kilogram, ensuring a highly competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: The starting material, (R)-(+)-1-indanamine, is a commercially available chiral building block with a stable supply chain, reducing dependency on custom synthesis of complex precursors. The reagents used, such as halo-propynes and trifluoroacetate esters, are commodity chemicals with multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent grades, ensures consistent production output even when facing minor fluctuations in raw material quality.
- Scalability and Environmental Compliance: The reaction operates under mild thermal conditions (20-100°C) and atmospheric pressure, removing the need for specialized high-pressure reactors or cryogenic cooling systems. This simplicity facilitates rapid scale-up from kilogram to multi-ton production scales without significant engineering hurdles. Additionally, the absence of heavy metals and the generation of water-soluble trifluoroacetate byproducts simplify wastewater treatment, ensuring easier compliance with increasingly stringent environmental regulations regarding industrial effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Rasagiline synthesis. These answers are derived directly from the experimental data and technical specifications provided in the source documentation, offering clarity on process feasibility and quality outcomes.
Q: How does this synthetic route eliminate the 50% waste associated with traditional resolution methods?
A: Traditional methods often synthesize racemic mixtures and discard the unwanted (S)-enantiomer. This patented process starts directly with commercially available chiral (R)-(+)-1-indanamine, utilizing 100% of the starting material's chirality and avoiding the generation of optical isomer waste entirely.
Q: What specific impurity profile challenges does the trifluoroacetyl group address?
A: Direct alkylation of free amines often leads to over-alkylation, creating difficult-to-remove N,N-di-propargyl impurities. The electron-withdrawing trifluoroacetyl group moderates the nucleophilicity of the nitrogen, ensuring mono-alkylation selectivity and significantly simplifying downstream purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes robust reaction conditions (20-100°C), common industrial solvents like DMF or alcohols, and inexpensive mineral bases. It avoids cryogenic temperatures, hazardous reducing agents like sodium borohydride, and complex chromatographic separations, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rasagiline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purities demonstrated in the laboratory are faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to monitor every batch for optical purity and trace impurities, guaranteeing that our Rasagiline intermediates meet the exacting standards of global pharmaceutical regulators.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this resolution-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your Parkinson's disease therapeutic portfolio.
